Aaa Sore Throat Spray 1.5 Mg Oromucosal Spray
SUMMARY OF PRODUCT CHARACTERISTICS
1 NAME OF THE MEDICINAL PRODUCT
AAA Sore Throat Spray 1.5 mg Oromucosal Spray
2. QUALITATIVE AND QUANTITATIVE COMPOSITION
Benzocaine E.P. 1.5mg
3 PHARMACEUTICAL FORM
Oromucosal spray.
4. CLINICAL PARTICULARS
4.1. Therapeutic Indications
Symptomatic temporary relief of pain associated with sore throat pain and minor infections.
4.2. Posology and Method of Administration
Spray two metered doses every two to three hours if required (not more than sixteen doses in every 24 hours) or as directed by the physician.
Children:
Children 6 - 12 years: One metered dose every two to three hours if required (not more than 8 doses in every 24 hours) or as directed by the physician.
Not suitable for use in children under 6 years.
Route of Administration
Topical application to the mucosa of the mouth and throat by means of a metered dose aerosol.
Product should not be administered for more than 7 consecutive days. Can to be shaken before use.
4.3 Contraindications
Known hypersensitivity to benzocaine or any of the excipients Methaemoglobinaemia
4.4 Special warnings and precautions for use
This preparation should not be administered to children under 6 years or used for more than seven consecutive days unless directed by a physician.
If the sore throat is severe, persistent or accompanied by fever or headache, a physician should be consulted before the use of this product. Avoid spraying into eyes.
Caution should be exercised in the use of this product if there have been previous allergic reactions with other local anaesthetics or sunscreen products.
Avoid inhalation of the product.
To be used with caution in patients with asthma.
4.5. Interaction with other Medicinal Products and other Forms of Interaction
Benzocaine is an ester which on hydrolysis produces p-aminobenzoic acid so it should not be used in patients being treated with sulphonamides.
4.6. Pregnancy and Lactation
There is no evidence, at present, of hazard from benzocaine in pregnancy. However, only very limited data is available. Therefore it should not be used in pregnancy unless considered essential by a physician.
4.7.
Effects on Ability to Drive and Use Machines
None.
4.8 Undesirable effects
Hypersensitivity reactions to benzocaine have been reported.
Methaemoglobinaemia may occur in patients receiving high doses or repeated applications of benzocaine-containing products.
Reporting of suspected adverse reactions
Reporting suspected adverse reactions after authorisation of the medicinal product is important. It allows continued monitoring of the benefit/risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions via the Yellow Card Scheme at: www. mhra. gov. uk/yellowcard
4.9 Overdose
There have been no reports of overdosage with AAA Spray. Systemic effects are unlikely.
5. PHARMACOLOGICAL PROPERTIES
5.1. Pharmacodynamic Properties
Benzocaine is a surface anaesthetic of the ester type. It has found frequent use as lozenges or solution to treat pain arising from various throat and mouth conditions.
5.2. Pharmacokinetic Properties
Benzocaine is sparingly soluble in water with toxicity about the tenth that of cocaine. It is not readily absorbed from mucus membranes. It is an ester which on hydrolysis produces p-aminobenzoic acid.
5.3. Pre-clinical Safety Data
None stated.
6.
PHARMACEUTICAL PARTICULARS
6.1. List of Excipients
Cetylpyridinium chloride, glycerin, ethanol, clove bud oil, menthol crystals, sodium saccharin 450, peppermint Cremophor, water.
6.2. Incompatibilities
None.
6.3. Shelf-Life
24 months.
6.4. Special Precautions for Storage
Keep away from naked flame.
Store in a cool place.
6.5. Nature and Content of Container
White aluminium can with 100ml pumps, white high density polyethylene folding arm activator and white high density polyethylene cap.
6.6. Instructions for Use, Handling and Disposal
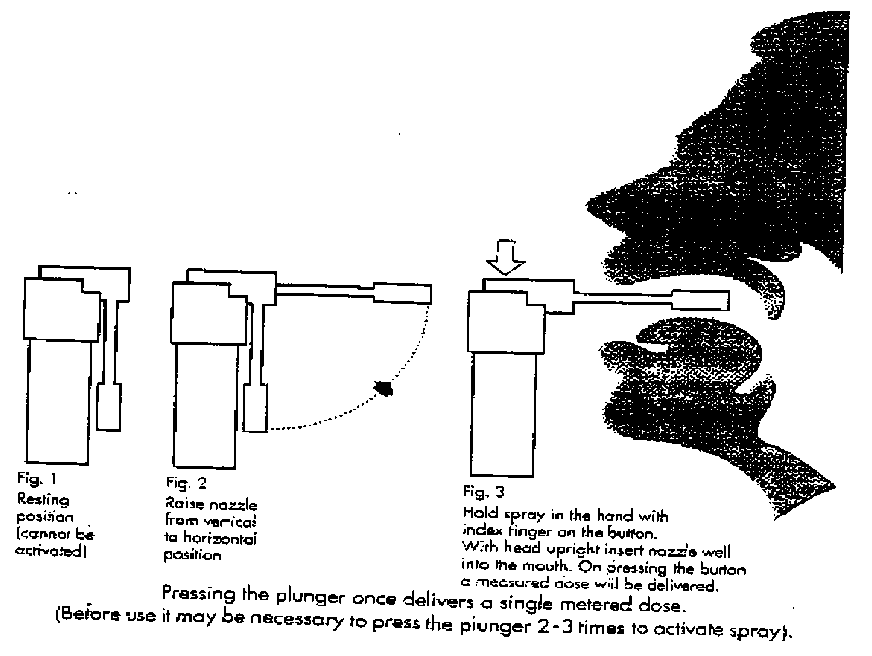
7 MARKETING AUTHORISATION HOLDER
Manx Pharma Limited Taylor Group House Wedgnock Lane Warwick CV34 5YA United Kingdom
8. MARKETING AUTHORISATION NUMBER(S)
PL 15833/0001
DATE OF FIRST AUTHORISATION/RENEWAL OF THE AUTHORISATION
1st September 1997/2nd January 2007
10
DATE OF REVISION OF THE TEXT
09/06/2015