Almuriva 9.5 Mg/24H Transdermal Patch

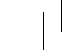
SZ00000LT000
1
What Almuriva is and what it is used for
swelling) and if it does not improve within 48 hours after removal of the transdermal patch.
If this applies to you, tell your doctor and do not apply Almuriva transdermal patches.
Almuriva 4.6 mg/24 h transdermal patch Almuriva 9.5 mg/24 h transdermal patch
rivastigmine
Read all of this leaflet carefully before you start
using this medicine.
• Keep this leaflet. You may need to read it again.
• If you have any further questions, ask your doctor or pharmacist.
• This medicine has been prescribed for you. Do not pass it on to others. It may harm them, even if their symptoms of illness are the same as yours.
• If any of the side effects gets serious, or if you notice any side effects not listed in this leaflet, please tell your doctor or pharmacist.
What is in this leaflet
1. What Almuriva is and what it is used for
2. What you need to know before you use Almuriva
3. How to use Almuriva
4. Possible side effects
5. How to store Almuriva
6. Contents of the pack and other information
The active substance of Almuriva is rivastigmine.
Rivastigmine belongs to a class of substances called cholinesterase inhibitors. In patients with Alzheimer's dementia, certain nerve cells die in the brain, resulting in low levels of the neurotransmitter acetylcholine (a substance that allows nerve cells to communicate with each other). Almuriva works by blocking the enzymes that break down acetylcholine: acetylcholinesterase and butyrylcholinesterase. By blocking these enzymes, Almuriva allows levels of acetylcholine to be increased in the brain, helping to reduce the symptoms of Alzheimer's disease.
Almuriva is used for the treatment of adult patients with mild to moderately severe Alzheimer's dementia, a progressive brain disorder that gradually affects memory, intellectual ability and behaviour.
What you need to know before you use Almuriva
Do not use Almuriva
• if you are allergic to rivastigmine (the active substance in Almuriva or any of the other ingredients of this medicine (listed in section 6).
• if you have ever had an allergic reaction to a similar type of medicine (carbamate derivatives).
• if you have a skin reaction spreading beyond the patch size, if there is a more intense local reaction (such as blisters, increasing skin inflammation,
Warnings and precautions
Talk to your doctor before using Almuriva:
• if you have, or have ever had, an irregular or slow heartbeat.
• if you have, or have ever had, an active stomach ulcer.
• if you have, or have ever had, difficulties in passing urine.
• if you have, or have ever had, seizures.
• if you have, or have ever had, asthma or a severe respiratory disease.
• if you suffer from trembling.
• if you have a low body weight.
• if you have gastrointestinal reactions such as feeling sick (nausea), being sick (vomiting) and diarrhoea. You may become dehydrated (losing too much fluid) if vomiting or diarrhoea are prolonged.
• if you have impaired liver function.
If any of these apply to you, your doctor may need to monitor you more closely while you are on this medicine.
If you have not applied a patch for more than three days, do not apply the next one before you have talked to your doctor.
Children and adolescents
There is no relevant use of Almuriva in the paediatric population in the treatment of Alzheimer's disease.
Other medicines and Almuriva
Tell your doctor or pharmacist if you are taking, have recently taken or might take any other medicines.
Almuriva might interfere with anticholinergic medicines some of which are medicines used to relieve stomach cramps or spasms (e.g. dicyclomine), to treat Parkinson's disease (e.g. amantadine) or to prevent motion sickness (e.g. diphenhydramine, scopolamine, or meclizine).
Almuriva Patch should not be given at the same time as metoclopramide (a medicine used to relieve or prevent nausea and vomiting). Taking the two medicines together could cause problems such as stiff limbs and trembling hands.
If you have to undergo surgery whilst using Almuriva transdermal patches, tell your doctor that you are using them because they may exaggerate the effects of some muscle relaxants during anaesthesia.
Caution when Almuriva Patch is given together with beta-blockers (medicines such as atenolol used to treat hypertension, angina, and other heart conditions). Taking the two medicines together could cause problems such as slowing of the heartbeat (bradycardia) leading to fainting or loss of consciousness.
Pregnancy, breast-feeding and fertility
If you are pregnant or breast-feeding, think you may be pregnant or are planning to have a baby, ask your doctor or pharmacist for advice before taking this medicine.
If you are pregnant, the benefits of using Almuriva transdermal patches must be assessed against the possible effects on your unborn child. Almuriva should not be used during pregnancy unless clearly necessary.
You should not breast-feed during treatment with Almuriva transdermal patches.
Driving and using machines
Your doctor will tell you whether your illness allows you to drive vehicles and use machines safely. Almuriva transdermal patches may cause fainting or severe confusion. If you feel faint or confused do not drive, use machines or perform any other tasks that require your attention.
3 How to use Almuriva
Always use Almuriva transdermal patches exactly
as your doctor has told you. Check with your doctor,
pharmacist or nurse if you are not sure.
IMPORTANT:
• Take off the previous patch before putting ONE new patch on.
• Only one patch per day.
• Do not cut the patch into pieces.
• Press the patch firmly in place for at least 30 seconds using the palm of the hand.
How to start treatment
Your doctor will tell you which Almuriva transdermal
patch is most suitable for you.
• Treatment usually starts with Almuriva 4.6 mg/24 h.
• The recommended usual daily dose is Almuriva 9.5 mg/24 h. If well tolerated, the treating physician may consider increasing the dose to 13.3 mg/24 h (The 13.3 mg/24 h dose strength cannot be achieved with this product. For conditions where this strength should be used, please refer to other
rivastigmine products for which transdermal patches of the 13.3 mg/24 h strength are available).
• Only wear one Almuriva patch at a time and replace the patch with a new one after 24 hours.
During the course of the treatment your doctor may adjust the dose to suit your individual needs.
If you have not applied a patch for three days, do not apply the next one before you have talked to your doctor. Transdermal patch treatment can be resumed at the same dose if treatment is not interrupted for more than three days. Otherwise your doctor will restart your treatment on Almuriva 4.6 mg/24 h.
Almuriva can be used with food, drink and alcohol.
Where to apply your Almuriva transdermal patch
• Before you apply a patch, make sure that your skin is clean, dry and hairless, free of any powder, oil, moisturiser or lotion that could keep the patch from sticking to your skin properly, free of cuts, rashes and/or irritations.
• Carefully remove any existing patch before putting on a new one. Having multiple patches on your body could expose you to an excessive amount of this medicine which could be potentially dangerous.
• Apply ONE patch per day to ONLY ONE of the possible locations, shown in the following diagrams:
• left upper arm or right upper arm
• left upper chest or right upper chest (avoid breast)
• left upper back or right upper back
• left lower back or right lower back
Every 24 hours take off the previous patch before putting on ONE new patch. The new patch should only be applied to one of the following possible locations.
|
Front __ 1 or |
or |
n |
or ~ or |
|
s |
■ I |
s / \ |
v |
|
Back ^ |
• |
• |
t |
|
or T_r |
S or |
5_ |
or ■ |
When changing the patch, you must remove the previous day’s patch before you apply the new one to a different location of skin each time (for example on the right side of your body one day, then on the left side the next day, and on your upper body one day, then on your lower body the next day). Do not apply a new patch to the same skin area twice within 14 days.
How to apply your Almuriva transdermal patch
Almuriva patches are thin, opaque, plastic patches that stick to the skin. Each patch is sealed in a sachet that protects it until you are ready to put it on. Do not open the sachet or remove a patch until just before you apply it.
Carefully remove the existing patch before putting on a new one.

For patients starting treatment for the first time and for patients restarting Almuriva after treatment interruption, please begin with the second picture.
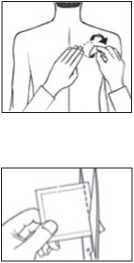
• Each patch is sealed in its own protective sachet.
You should only open the sachet when you are ready to apply the patch. Cut the sachet along the dotted line with scissors and remove the patch from the sachet.
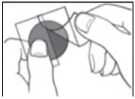
• A protective liner covers the sticky side of the patch.
Peel off one side of the protective liner and do not touch the sticky part of the patch with the fingers.
• Put the sticky side of the patch on the upper or lower back, upper arm or chest and then peel off the second side of the protective liner.
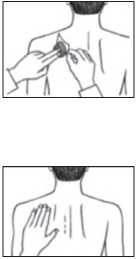
• Then press the patch firmly in place for at least 30 seconds using the palm of the hand to make sure that the edges stick well.


4 Possible side effects
5 How to store Almuriva
6
Contents of the pack and other information
If it helps you, you may write, for example, the day of the week, on the patch with a thin ball point pen.
The patch should be worn continuously until it is time to replace it with a new one. You may wish to experiment with different locations when applying a new patch, to find ones that are most comfortable for you and where clothing will not rub on the patch.
How to remove your Almuriva transdermal patch
Gently pull at one edge of the patch to remove it slowly from the skin. In case the adhesive residue is left over on your skin, gently soak the area with warm water and mild soap or use baby oil to remove it. Alcohol or other dissolving liquids (nail polish remover or other solvents) should not be used.
You should wash your hands with soap and water after removing the patch. In case of contact with eyes or if the eyes become red after handling the patch, rinse immediately with plenty of water and seek medical advice if symptoms do not resolve.
Can you wear your Almuriva transdermal patch when you are bathing, swimming, or in the sun?
• Bathing, swimming or showering should not affect the patch. Make sure the patch does not loosen during these activities.
• Do not expose the patch to any external heat sources (e.g. excessive sunlight, saunas, solarium) for long periods of time.
What to do if a patch falls off
If a patch falls off, apply a new one for the rest of the day, then replace it at the same time as usual the next day.
When and for how long to apply your Almuriva transdermal patch
• To benefit from treatment, you must apply a new patch every day, preferably at the same time of day.
• Only wear one Almuriva patch at a time and replace the patch with a new one after 24 hours.
If you use more Almuriva than you should
If you accidentally apply more than one patch, remove all the patches from your skin, then inform your doctor that you have accidentally applied more than one patch. You may require medical attention. Some people who have accidentally taken too much Almuriva have experienced feeling sick (nausea), being sick (vomiting), diarrhoea, high blood pressure and hallucinations. Slow heartbeat and fainting may also occur.
If you forget to use Almuriva
If you find you have forgotten to apply a patch, apply one immediately. You may apply the next patch at the usual time the next day. Do not apply two patches to make up for the one that you missed.
If you stop using Almuriva
Tell your doctor or pharmacist if you stop using the patch.
If you have any further questions on the use of this medicine, ask your doctor or pharmacist.
Like all medicines, Almuriva transdermal patches can cause side effects, although not everybody gets them.
You may have side effects more often when you start your medicine or when your dose is increased. Usually, the side effects will slowly go away as your body gets used to the medicine.
Take off your patch and tell your doctor immediately or go to the hospital if you experience any of the following side effects which could become serious:
Common (may affect up to 1 in 10 people)
• Urinary incontinence (inability to retain adequate urine)
Uncommon (may affect up to 1 in 100 people)
• Problems with your heartbeat such as slow heartbeat
• Seeing things that are not really there (hallucinations)
Not known (frequency cannot be estimated from the available data)
• Allergic reaction where the patch was used, such as blisters or inflamed skin
• The signs of Parkinson's disease get worse - such as tremor, stiffness and shuffling
• Inflammation of the pancreas - signs include serious upper stomach pain, often with feeling sick (nausea) or being sick (vomiting)
• Liver disorders (yellow skin, yellowing of the whites of the eyes, abnormal darkening of the urine or unexplained nausea, vomiting, tiredness and loss of appetite)
• Fast or uneven heartbeat
• Fits (seizures)
The following side effects have also been reported:
Common (may affect up to 1 in 10 people)
• Loss of appetite
• Feeling dizzy
• Feeling agitated or sleepy
Uncommon (may affect up to 1 in 100 people)
• Stomach ulcer
• Dehydration (losing too much fluid)
• Hyperactivity (high level of activity, restlessness)
• Aggression
Rare (may affect up to 1 in 1,000 people)
• Falling
Very rare (may affect up to 1 in 10,000 people)
• Stiff arms or legs
• Trembling hands
Not known (frequency cannot be estimated from the available data)
• High blood pressure
• Changes in tests which show how well the liver is working
• Feeling restless
• Nightmares
Other side effects seen with rivastigmine capsules or oral solution and which may occur with the patch:
Tell your doctor immediately or go to the hospital if you experience any of the following side effects which could become serious:
Uncommon (may affect up to 1 in 100 people)
• Uneven heart rate (e.g. fast heart rate)
Rare (may affect up to 1 in 1,000 people)
• Fits (seizures)
• Chest pain - this may be caused by heart spasm
Very rare (may affect up to 1 in 10,000 people)
• Inflammation of the pancreas - the signs include serious upper stomach pain, often with feeling sick (nausea) or being sick (vomiting)
• Some people who have been violently sick have had tearing of the tube that connects your mouth with your stomach (oesophagus)
• Bleeding in the gut - shows as blood in stools or when being sick
Other side effects with rivastigmine capsules or oral solution :
Common (may affect up to 1 in 10 people)
• Too much saliva
• Loss of appetite
• Feeling restless
• Generally feeling unwell
• Trembling or feeling confused
• Increased sweating
Uncommon (may affect up to 1 in 100 people)
• Difficulty sleeping
• Accidental falls
Rare (may affect up to 1 in 1,000 people)
• Ulcer in the intestine
Very rare (may affect up to 1 in 10,000 people)
• High blood pressure
• Seeing things that are not there (hallucinations)
Reporting of side effects
If you get any side effects, talk to your doctor, pharmacist or nurse. This includes any possible side effects not listed in this leaflet.
You can also report side effects directly via the Yellow Card Scheme: www.mhra.gov.uk/yellowcard.
By reporting side effects you can help provide more information on the safety of this medicine
• Keep this medicine out of the sight and reach of children.
• Do not use this medicine after the expiry date, which is stated on the carton and sachet. The expiry date refers to the last day of the month.
• Do not store above 25°C.
• Keep the transdermal patch in the sachet until use.
• Do not use any patch that is damaged or shows signs of tampering.
• After removing a patch, fold it in half with the sticky sides on the inside and press them together.
Return the used patch to its sachet and dispose
of it in such a way that children cannot handle it.
Do not touch your eyes with your fingers and wash your hands with soap and water after removing the patch. If your community burns domestic rubbish, you can dispose of the patch with your domestic rubbish. Otherwise, return used patches to a pharmacy, preferably in the original packaging.
What Almuriva contains
• The active substance is rivastigmine.
- Almuriva 4.6 mg/24 h transdermal patches: Each patch releases 4.6 mg of rivastigmine per 24 hours is 5 cm2 and contains 9 mg of rivastigmine.
- Almuriva 9.5 mg/24 h transdermal patches: Each patch releases 9.5 mg of rivastigmine per 24 hours is 10 cm2 and contains 18 mg of rivastigmine.
- The other ingredients are: polyethylene terephthalate film lacquered, all-rac-a Tocopherol, poly(butylmethacrylate, methyl-methacrylate) copolymer (3:1), acrylic copolymer, silicone, dimeticone, polyester film fluoropolymer-coated, resin, pigments, organic polymers/resins.
What Almuriva looks like and contents of the pack Almuriva 4.6 mg/24 h transdermal patches Each transdermal patch is a thin, matrix-type transdermal patch consisting of three layers. The outside of the backing layer is beige and labelled with “RIV”, 4.6 mg/24 h”
Almuriva 9.5 mg/24 h transdermal patches Each transdermal patch is a thin, matrix-type transdermal patch consisting of three layers. The outside of the backing layer is beige and labelled with “RIV”, 9.5mg/24 h”
One transdermal patch is sealed in one sachet. The patches are available in packs containing 7, 30, 60 (2 packs of 30), 90 (3 packs of 30) sachets. Not all pack sizes may be marketed in your country.
Marketing Authorisation Holder and Manufacturer Marketing Authorisation Holder
Sandoz Limited,
Frimley Business Park, Frimley,
Camberley, Surrey, GU16 7SR.
Manufacturer
Novartis Pharma GmbH,
Roonstrasse 25,
D-90429 Nurnberg
Germany
or
Hexal AG
Industriestrasse 25,
83607 Holzkirchen Germany
or
Salutas Pharma GmbH Otto-von-Guericke-Allee 1,
39179 Barleben Germany
or
Lek Pharmaceuticals d.d Verovskova 57,
1526 Ljubljana Slovenia.
This leaflet was last revised in 12/2015.
SZ00000LT000

|
Artwork Proof Box | |
|
Ref: V007- PSUSA - SPC & PIL | |
|
Proof no. Date prepared: 004.0 15/12/2015 |
Font size: 8pt |
|
Colours: | Black O □ □ |
Fonts: Helvetica |
|
Dimensions: 594 x 148 mm |
_/ |
