Alphagan Eye Drops
ALPHAGAN® EYE DROPS BRIMONIDINE TARTRATE 0.2% EYE DROPS
(brimonidine tartrate)
PACKAGE LEAFLET: INFORMATION FOR THE USER
Your medicine is avaliable using one of the above names but will be referred to as Alphagan throughout the leaflet.
Read all of this leaflet carefully before you start using this medicine.
■ Keep this leaflet. You may need to read it again.
■ If you have any further questions, ask your doctor or pharmacist.
■ This medicine has been prescribed for you. Do not pass it on to others. It may harm them, even if their symptoms are the same as yours.
■ If any of the side effects gets serious, or if you notice any side effects not listed in this leaflet, please tell your doctor or pharmacist.
In this leaflet:
1. What Alphagan is and what it is used for
2. Before you use Alphagan
3. How to use Alphagan
4. Possible side effects
5. How to store Alphagan
6. Further information
1. What Alphagan is and what it is used for
Alphagan is used to reduce pressure within the eye.
It can be used either alone, when beta-blocker eye drops are contraindicated, or with another eye drop, when a single medicine is not enough to lower the increased pressure in the eye, in the treatment of open angle glaucoma or ocular hypertension. The active ingredient in Alphagan is brimonidine tartrate, which works by reducing pressure within the eyeball.
2. Before you use Alphagan Do not use Alphagan
■ If you are allergic (hypersensitive) to brimonidine tartrate or any of the other ingredients of Alphagan.
■ If you are taking monoamine oxidase (MAO) inhibitors or certain antidepressants. You must inform your doctor if you are taking any antidepressant drug.
■ If you are breast-feeding.
■ In infants/babies (from birth until 2 years).
Take special care with Alphagan
Before you take this medicine, tell your doctor:
■ If you suffer or have suffered from depression, reduced mental capacity, reduced blood supply to the brain, heart problems, a disturbed blood supply of the limbs or a blood pressure disorder.
■ If you have or have had in the past kidney or liver problems.
■ If being given to a child between the age of 2 and 12 years because Alphagan is not recommended for use in this age group.
Using other medicines
Please tell your doctor or pharmacist if you are taking or have recently taken any other medicines, including medicines obtained without a prescription.
Tell your doctor if you are taking any of the following medicines:
■ pain killers, sedatives, opiates, barbiturates or are regularly consuming alcohol.
■ anaesthetics.
■ to treat a heart condition or to lower blood pressure.
■ that can affect the metabolism like chlorpromazine, methylphenidate and reserpine.
■ which work on the same receptor as Alphagan, for example isoprenaline and prazosin.
■ monoamine oxidase (MAO) inhibitors and other antidepressants.
■ medicines for any condition, even if unrelated to your eye condition.
■ or if the dose of any of your current medicines is changed. These could affect your treatment with Alphagan.
Pregnancy and breast-feeding
Ask your doctor or pharmacist for advice before taking any medicine. Tell your doctor if you are pregnant or planning to become pregnant.
Alphagan should not be used while breast-feeding.
Driving and using machines
■ Alphagan may cause blurred or abnormal vision. This effect may seem worse at night or in reduced lighting.
■ Alphagan may also cause drowsiness or tiredness in some patients.
■ If you experience any of these symptoms, do not drive or use machinery until the symptoms are cleared.
Important information about some of the ingredients of Alphagan
A preservative in Alphagan (benzalkonium chloride) may cause eye irritation and is also known to discolour soft contact lenses. Therefore, avoid contact with soft lenses. If you wear soft contact lenses take them out before using these eye drops and then wait 15 minutes after using Alphagan before inserting your lenses.
3. How to use Alphagan
Always use Alphagan exactly as your doctor has told you. You should check with your doctor or pharmacist if you are not sure.
Adults
The usual dose is one drop twice daily in the affected eye(s), approximately 12 hours apart.
Children under 12 years
Alphagan must not be used for infants below 2 years of age. Alphagan is not recommended for use in children (from 2 years until 12 years).
Instructions for use
Alphagan comes as eye drops. Always wash your hands before applying eye drops. Your prescription label tells you how many drops to use at each dose. If you use Alphagan with another eye drop, wait 5-15 minutes before applying the second eye drop. Apply your eye drops in the following way:
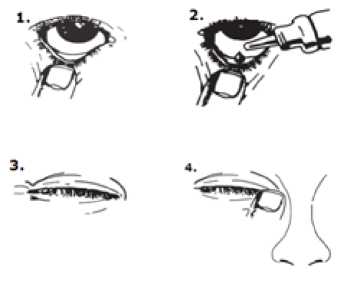
1. Tilt your head back and look at the ceiling.
2. Gently pull the lower eyelid down until there is a small pocket.
3. Squeeze the upturned dropper bottle to release a drop into your eye.
4. Whilst keeping the affected eye closed, press your finger against the corner of the closed eye (the side where the eye meets the nose) and hold for 1 minute.
Avoid touching the dropper tip against your eye or anything else. Replace and tighten the cap straight after use.
If you use more Alphagan than you should Adults
In adults who were instilling more drops than prescribed, the side effects reported were those already known to occur with Alphagan.
Adults who accidentally swallowed Alphagan experienced decrease in blood pressure, which in some patients was followed by the increase in blood pressure.
Children
Serious side effects were reported in children who accidentally swallowed Alphagan. Signs included sleepiness, floppiness, low body temperature, paleness and breathing difficulties. Should this happen, contact your doctor immediately.
Adults and Children
If Alphagan has been accidentally swallowed or if you have used more Alphagan than you should, please contact your doctor immediately.
If you forget to use Alphagan
If you forget to take a dose, apply it as soon as you remember. If, however, it is almost time for your next dose, you should omit the missed dose altogether and then follow your normal routine.
|
Very common |
Occurs in more than 1 out of 10 patients |
|
Common |
Occurs in fewer than 1 out of 10 patients |
|
Uncommon |
Occurs in fewer than 1 out of 100 patients |
|
Ra re |
Occurs in fewer than 1 out of 1,000 patients |
|
Very rare |
Occurs in fewer than 1 out of 10,000 patients |
If you stop using Alphagan
To be effective Alphagan must be used every day. Do not stop using Alphagan until your doctor tells you to. If you have any further questions on the use of this product, ask your doctor or pharmacist.
4. Possible side effects
Like all medicines, Alphagan can cause side effects, although not everybody gets them. The chance of having a side effect is described by the following categories:
The following eye side effects may be seen with Alphagan.
Affecting the eye
Very common:
■ Irritation of the eye (eye redness, burning, stinging, a feeling of something in the eye, itching, follicles or white spots on the see-through layer which covers the surface of the eye)
■ Blurred vision
■ An allergic reaction in the eye
Common:
■ Local irritation (inflammation and swelling of the eyelid, swelling of the see-through layer which covers the surface of the eye, sticky eyes, pain and tearing)
■ Sensitivity to light
■ Erosion on the surface of the eye and staining
■ Eye dryness
■ Whitening of the see-through layer which covers the surface of the eye
■ Abnormal vision
■ Inflammation of the see-through layer which covers the surface of the eye
Very rare:
■ Inflammation within the eye
■ Reduction in pupil size
Side effects where the occurrence is unknown:
■ Itching of eyelids
Affecting the body
Very common:
■ Headache
■ Dry mouth
■ Tiredness/drowsiness
Common:
■ Dizziness
■ Cold-like symptoms
■ Symptoms involving the stomach and digestion
■ Abnormal taste
■ General weakness
Uncommon:
■ Depression
■ Palpitations or changes in heart rate
■ Dry nose
■ General allergic reactions.
Rare:
■ Shortness of breath
Very rare:
■ Sleeplessness
■ Fainting
■ High blood pressure
■ Low blood pressure
Side effects where the occurrence is unknown:
■ Skin reactions including redness, face swelling, itching, rash and widening of blood vessels
Reporting of side effects
If you get any side effects, talk to your doctor or pharmacist. This
includes any possible side effects not listed in this leaflet.
You can also report side effects directly via: Yellow Card Scheme
Website: www.mhra.gov.uk/yellowcard.
By reporting side effects you can help provide more information on
the safety of this medicine.
5. How to store Alphagan
■ Keep out of the sight and reach of children.
■ Store the bottle at or below room temperature (25°C).
■ Do not use the bottle if the tamper-proof seal on the bottle is broken before first use. The product should not be used after the expiry date (This is printed on both the bottle label and on the carton the bottle is packed in). The expiry date refers to the last day of that month.
■ Throw the bottle away 28 days after opening, even if there is solution remaining.
■ If your doctor decides to stop your treatment, take any leftover medicine back to the pharmacy.
■ If your medicine appears to be discoloured or shows any other signs of deterioration, take it to your pharmacist who will advise you.
■ Medicines should not be disposed of via wastewater or household waste. Ask your pharmacist how to dispose of medicines no longer required. These measures will help to protect the environment.
6. Further information What Alphagan contains
■ The active ingredient is brimonidine tartrate 0.2% w/v (2.0mg/ml) equivalent to brimonidine base 0.13% w/v (1.3mg/ml).
One ml solution contains 2mg of brimonidine tartrate equivalent to 1.3mg brimonidine base.
■ It also contains benzalkonium chloride as a preservative, polyvinyl alcohol 1.4% (14mg/ml), sodium chloride, sodium citrate dihydrate, citric acid monohydrate, purified water, hydrochloric acid or sodium hydroxide to adjust pH.
What Alphagan looks like and contents of the pack
Alphagan is a clear eyedrop solution.
Each bottle contains 5ml of the medicine.
Manufacturer
Manufactured by: Allergan, Hormigueros, Puerto Rico.
And
Allergan Pharmaceuticals Ireland, Westport, Co. Mayo, Ireland.
Procured from within the EU and repackaged by: Doncaster Pharmaceuticals Group Ltd., Kirk Sandall, Doncaster, DN3 1QR.
Product Licence holder: BR Lewis Pharmaceuticals Ltd.,
Kirk Sandall, Doncaster, DN3 1QR.
PL No: 08929/0035 |POM|
Leaflet revision and issue date (Ref): 15.01.15
Alphagan® is a registered trademark of Allergan Inc.
For any information about this medicinal product, please contact
the local representative of the Marketing Authorisation Holder:
Allergan Ltd., The Parkway, Marlow, Bucks, SL7 1YL
United Kingdom
Tel: +44 (0)1628 494026
e-mail: uk medinfo@allergan.com
Further information about glaucoma is available from: International Glaucoma Association (IGA)
Tel: 01233 64 81 70 Fax: 01233 64 81 79 Email: info@iga.org.uk
(The IGA is an organisation which helps glaucoma patients and their relatives, and is not associated with Allergan.)
Page 2 of 2