Amorolfine Hydrochloride 5% W/V Nail Lacquer
Out of date information, search anotherPACKAGE LEAFLET: INFORMATION FOR THE USER
Amorolfine Hydrochloride 5% w/v Nail Lacquer
Read all of this leaflet carefully before you start using this medicine.
• Keep this leaflet. You may need to read it again.
• If you have any further questions, ask your doctor or pharmacist.
• This medicine has been prescribed for you. Do not pass it on to others, it may harm them, even if their symptoms are the same as yours.
• If any of the side-effects gets serious, or if you notice any side-effects not listed in this leaflet, please tell your doctor, pharmacist or nurse. See section 4.
In this leaflet:
1. What Amorolfine hydrochloride 5% w/v is and what it is used for
2. Before you use Amorolfine hydrochloride 5% w/v
3. How to use Amorolfine hydrochloride 5% w/v
4. Possible side-effects
5. How to store Amorolfine hydrochloride 5% w/v
6. Further information
1. What Amorolfine hydrochloride 5% w/v is and what it is used for
• Amorolfine hydrochloride 5% w/v is used to treat fungal infections of the nails.
• Amorolfine hydrochloride 5% w/v contains the active ingredient amorolfine (as the hydrochloride) which belongs to a group of medicines known as antifungals.
• It kills a wide variety of fungi that can cause nail infections.
2. Before you use Amorolfine hydrochloride 5% w/v
Do not use Amorolfine hydrochloride 5% w/v if you are:
• allergic (hypersensitive) to amorolfine or any of the other ingredients of Amorolfine hydrochloride 5% w/v (see section 6 for other ingredients).
Take special care with Amorolfine hydrochloride 5% w/v:
• if you suffer from diabetes
• if you are being treated because you have a weak immune system
• if you have poor circulation in your hands and feet
• if your nail is severely damaged or infected
• if you get Amorolfine hydrochloride 5% w/v in your eyes or ears wash it out with water immediately and contact your doctor, pharmacist or nearest hospital straight away
• avoid the lacquer coming into contact with mucous membranes (e.g. mouth and nostrils). Do not breathe it in.
Using other medicines
You can use the nail lacquer whilst you are taking other medicines.
Using other nail products
Nail varnish or artificial nails should not be used while using Amorolfine hydrochloride 5% w/v.
Pregnancy and breast-feeding
Tell your doctor if you are pregnant, planning to become pregnant or are breast-feeding. Your doctor will then decide whether you should use Amorolfine hydrochloride 5% w/v. Ask your doctor or pharmacist for advice before taking any medicine.
3. How to use Amorolfine hydrochloride 5% w/v
Always use Amorolfine hydrochloride 5% w/v exactly as your doctor has told you. You should check with your doctor or pharmacist if you are not sure.
Amorolfine hydrochloride 5% w/v should be applied to the affected finger or toe nails once or twice a week exactly as directed by your doctor.
Instructions for use:
Step 1: File the nail
Before the first application, file down the infected areas of nail, including the nail surface, as much as possible using the nail file provided.
CAUTION: Do not use nail files used for infected nails on healthy nails, otherwise you may spread the infection. To prevent the spread of infection take care that no one else uses the files from your kit.
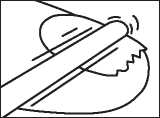
Step 2: Clean the nail
Use one of the swabs provided (or nail varnish remover) to clean the nail surface. Repeat steps 1 and 2 for each affected nail.
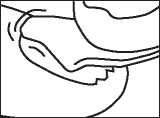
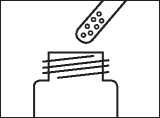
Step 3: Take some lacquer from the bottle
Dip one of the re-usable applicators into the bottle of nail lacquer. The lacquer must not be wiped off the edge of the bottle before it is applied.
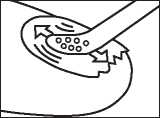
Step 4: Apply the lacquer
Apply the lacquer evenly over the entire surface of the nail. Repeat this step for each affected nail.
Let the treated nail(s) dry for approximately 3 minutes.
Step 5: Clean the applicator
The applicators provided are re-usable. However, it is important to clean them thoroughly after completing each treatment procedure, using the same swab you used for nail cleansing. Avoid touching newly treated nails with the swab. Close the nail lacquer bottle tightly. Dispose of the swab carefully as it is inflammable.
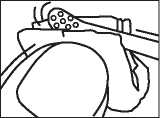
Please read the back of this leaflet.

GENUS PHARMACEUTICALS 25218002
’Trademark
GEN-AMR-PIL-307_02
• Before using the nail lacquer again, first remove the old lacquer from your nails using a swab, then file down the nails again if necessary.
• Re-apply the lacquer as previously described.
• When dry, the nail lacquer is unaffected by soap and water, so you may wash your hands and feet as normal. If you need to use chemicals such as paint thinners or white spirit, rubber or other impermeable (waterproof) gloves should be worn to protect the lacquer on your fingernails.
• It is important to carry on using Amorolfine hydrochloride 5% w/v until the infection has cleared and healthy nails have grown back. This usually takes 6 months for fingernails and 9 to 12 months for toenails.
Your doctor will probably check how your treatment is progressing every 3 months or so.
If you accidentally swallow Amorolfine hydrochloride 5% w/v
If you, or anyone else, accidentally swallows the lacquer contact your doctor, pharmacist or nearest hospital straight away.
If you forget to use Amorolfine hydrochloride 5% w/v
Do not worry if you forget to use the lacquer at the right time. When you remember, start using the product again, in the same way as before.
If you stop using Amorolfine hydrochloride 5% w/v
Do not stop using Amorolfine hydrochloride 5% w/v before your doctor tells you to or your infection could come back. If you have any further questions on the use of this product, ask your doctor or pharmacist.
4. Possible side-effects
Like all medicines, Amorolfine hydrochloride 5% w/v can cause side-effects, although not everybody gets them.
Rare side-effects (occurring in less than 1 in 1000 people)
Your nail may become discoloured or it may become loose or start to separate from the nail bed.
Very rare side-effects (occurring in less than 1 in 10,000 people)
A burning sensation or allergic skin reaction (contact dermatitis) may occur in the area around the nail.
Reporting of side effects
If you get any side effects, talk to your doctor, pharmacist or nurse. This includes any possible side effects not listed in this leaflet. You can also report side effects directly via the Yellow Card Scheme at: www.mhra.gov.uk/yellowcard
By reporting side effects you can help provide more information on the safety of this medicine.
5. How to store Amorolfine hydrochloride 5% w/v
• Keep out of the sight and reach of children.
• Do not use Amorolfine hydrochloride 5% w/v after the expiry date which is stated on the pack. The expiry date refers to the last day of that month.
• Store below 30°C. Protect from heat. Keep the bottle tightly closed and upright.
This product is flammable! Keep the solution away from fire and flames!
Medicines should not be disposed of via wastewater or household waste. Ask your pharmacist how to dispose of medicines no longer required. These measures will help to protect the environment.
6. Further information
What Amorolfine hydrochloride 5% w/v contains:
Amorolfine hydrochloride 5% w/v contains 50mg/ml of the active substance amorolfine (equivalent to 55.74mg/ml amorolfine hydrochloride). The other ingredients are Eudragit RL 100 (ammonio methacrylate copolymer A), triacetin, butyl acetate, ethyl acetate and ethanol (anhydrous).
What Amorolfine hydrochloride 5% w/v looks like and contents of the pack
Amorolfine hydrochloride 5% w/v is a clear, colourless to pale yellow solution. It is available in a 5ml pack; 1 bottle packed with or without cleansing swabs, spatulas and / or nail files.
PL 06831/0273 Amorolfine Hydrochloride 5% w/v Nail Lacquer
Marketing Authorisation Holder:
Genus Pharmaceuticals,
Linthwaite,
Huddersfield,
HD7 5QH,
UK.
Manufacturer:
Chanelle Medical,
Loughrea,
Co. Galway,
Ireland.
This leaflet was last revised in December 2014
"Trademark
25218002
LA7774

GENUS PHARMACEUTICALS
GEN-AMR-PIL-307_02
05/02/2015