Betafact 50 Iu/Ml Powder And Solvent For Solution For Injection
|
Intensity of the bleeding/ Type of surgical procedure |
Target plasma factor IX levels (%)(IU/dl) |
Frequency of injections (hours)/ Treatment time (days) |
|
Haemorrhage Early haemarthrosis, muscle haemorrhage or oral bleeding. |
20 to 40 |
Repeat every 24 hours for at least 1 day, until the pain has resolved or-bieeding has stopped. |
|
Extensive haemarthrosis, muscle haemorrhage or haematoma. |
30 to 60 |
Repeat injection every 24 hours for 3 - 4 days or more until range of movement returns and pain has stopped. |
|
Life threatening haemorrhages |
60 to 100 |
Repeat the injection every 8 to 24 hours until bleeding is controlled. |
|
Surgery Minor surgery including tooth extraction |
30 to 60 |
Every 24 hours, for at least 1 day, until bleeding has stopped. |
|
Major surgery |
80 to 100 (pre- and post-surgery) |
Repeat injections every 8 to 24 hours until adequate wound healing; continue treatment for at an additional 7 days maintaining a factor IX activity between 30% and 60% (lU/dl). |
Reconstitution:
Reconstitute the preparation with water for injections as described below. Use current guidelines for aseptic procedure.
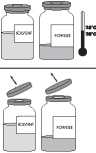
♦ if necessary, bring the two vials (powder and solvent) to ambient temperature.
♦ Remove the protective cap from the solvent vial (water for injections) and from the powder vial.
♦ Disinfect the surface of each stopper.

♦ Remove the translucent protective sheath from the transfer system and completely insert the exposed needle through the centre of the stopper of the solvent vial while simultaneously twisting the needle.
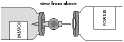
Remove the second protective sheath from the other end of the transfer system.
Keeping both vials horizontal (vented spike pointing upwards), quickly push the free end of the needle into the centre of the stopper of the powder vial.
Ensure that the needle always remains immersed in the solvent to avoid releasing the vacuum prematurely.

♦ immediately place the system upright in a vertical position, keeping the solvent vial directly above the powder vial, to allow the solvent to transfer into the powder.
♦ During the transfer, direct the jet of solvent over the whole surface of the powder. Ensure that ail of the solvent is transferred.
♦ The vacuum is automatically released at the end of the transfer procedure (sterile air).
♦ Remove the empty vial (solvent) with the transfer system.
♦ Gently swirl for a few minutes with a rotating movement to avoid the formation of foam until the powder has completely dissolved.

BIOMEDICAMENTS
LFB BiOMEDICAMENTS
3, avenue des Tropiques - BP 305 - LES ULIS - 91958 Courtabceuf Cedex - FRANCE Telephone : +33 1 69 82 70 10 0000000-000000/0.0
Under certain circumstances, larger amounts of BETAFACT than those calculated may be required, particularly for the starting dose.
During the course of treatment, appropriate determination of factor IX levels is advised to guide the dose to be administered and the frequency of repeated infusions. In the case of major surgical interventions in particular, precise monitoring of the substitution therapy by means of coagulation analysis (plasma factor IX activity) is indispensable. Individual patients may vary in their response to factor IX, demonstrating different half-lives and recoveries.
For long term prophylaxis for severe haemophilia B, the recommended dose is 20 to 40 IU of BETAFACT per kg of body weight to be administered every 3 to 4 days.
In certain cases, especially in young patients, it maybe necessary to increase the dose or shorten the time between two injections.
Patients should be monitored for the development of factor IX inhibitors. If the expected factor IX activity plasma levels are not attained, or if bleeding is not controlled with an appropriate dose, an assay should be performed to determine if a factor IX inhibitor is present. If the inhibitor is present at levels less than 10 Bethesda Units (BU) per ml, administration of additional human coagulation factor IX may neutralise the inhibitor. In patients with inhibitor titres above 10 BU or with high anamnestic response, the use of (activated) prothrombin complex concentrate (aPCC) or activated factor VII (FVIIa) has to be considered. These therapies should be directed by physicians with experience in the care of patients with haemophilia.
In a clinical trial, 13 children less than 6 years old have been treated with BETAFACT, for them the dosage is similar to that administered in adults.
The powder generally dissolves instantly and should be completely dissolved in less than 5 minutes.
The solution should be clear.
Do not use a solution which is cloudy or has deposits.
Do not mix with other medicinal products.
Do not dilute the reconstituted product.
Administration:
Draw the product into a sterile syringe using the filter needle provided. Remove the needle from the syringe.
Connect the syringe to an intravenous needle; expel the air from the syringe, disinfect the skin over the vein and insert the needle.
Inject intravenously as a single dose immediately after reconstitution, at a maximum rate of 4 ml/minute.
Storage after reconstitution:
For sterility reasons, the product should be used immediately. However chemical and physical in-use stability has been demonstrated for 3 hours at 25°C after reconstitution.
o
BETAFACT
50 lU/ml
Powder and solvent for solution for injection
Human coagulation factor IX PACKAGE LEAFLET: INFORMATION FOR THE USER
Read all of this leaflet carefully before you start using this medicine because It contains Important Information for you.
■ Keep this leaflet. You may need to read it again.
■ If you have any further questions, ask your doctor or pharmacist.
■ This medicine has been prescribed for you only. Do not pass it on to others. It may harm them, even if their signs of illness are the same as yours.
■ If you get any side effects talk to your doctor or pharmacist. This includes any possible side effects not listed in this leaflet, see section 4.
What is in this leaflet:
1. What BETAFACT 50 lU/ml, is and what it is used for
2. What you need to know before you use BETAFACT
3. How to use BETAFACT
4. Possible side effects
5. How to store BETAFACT
6. Contents of the pack and other information
1. What BETAFACT is and what it is used for
BETAFACT is a medicine which belongs to the class of antihaemorrhagics. The active substance is human coagulation factor IX, a protein which is naturally present in the body. The role of this protein is to ensure normal coagulation of the blood and to prevent that bleeding lasts too long.
BETAFACT is used to compensate for the lack of coagulation factor IX and, thus, prevent and treat bleeding (haemorrhages) in patients with haemophilia B. Haemophilia B is a hereditary disease characterised by a lack of a protein called coagulation factor IX. This lack causes coagulation disorders.
2. What you need to know before you use BETAFACT Do not use BETAFACT
■ If you are allergic to the active substance (factor IX) or any of the other ingredients of this medicine (listed in section 6. “Contents of the pack and other information”).
■ If you are allergic to heparin or its derivatives.
■ If you have in the past had a fall in your platelet count because of a medicine containing heparin.
■ If your doctor has told you that you are allergic to heparin, contact him/her before taking this medicine.
Your doctor should evaluate the possible benefits of treatment with BETAFACT because of the risk of abnormal formation of clots in the blood (thromboembolic complications)
■ in patients with signs of degradation of blood clots (fibrinolysis),
■ in patients presenting with the formation of multiple clots in the circulating blood (disseminated intravascular coagulation),
■ in new-born babies,
■ if you just underwent a surgery,
■ if your blood coagulation is abnormally high,
■ if you have a liver disease.
Your doctor will ask you to undergo blood tests to detect the signs of these complications as soon as possible.
Risk of allergic reactions
Due to the risk of allergies (see section 4. “Possible side effects”) during the administration of factor IX, the first injections of BETAFACT must be performed under medical supervision to provide immediate treatment for allergies, if necessary.
Your doctor will inform you of the warning signs of an allergic reaction (see section 4. “Possible side effects”). If one of these effects does occur, immediately stop the treatment and alert a doctor to start appropriate treatment depending on the type and severity of the reaction.
After repeated treatments with BETAFACT, your immune system may react to factor IX by producing inhibitors (anti-factor IX antibodies). The appearance of such inhibitors could reduce the treatment's efficacy. Your doctor must regularly perform blood tests to control for the appearance of these inhibitors and measure their quantity.
A link has been demonstrated between the presence of factor IX inhibitors and the occurrence of allergic reactions. Therefore:
■ If you present with allergic reactions following the use of factor IX, tests for inhibitors must be performed.
■ If factor IX inhibitors are detected, there is a greater risk of developing a severe allergic reaction during the injection of factor IX.
BETAFACT contains traces of human proteins other than factor IX. These proteins could also play a role in the appearance of allergic reactions.
Information on safety measures due to the origin of BETAFACT
BETAFACT is manufactured from human plasma (the liquid part of blood).
When medicines are made from human blood or plasma, certain measures are put in place to prevent infections being passed on to patients.
These include:
■ careful selection of blood and plasma donors to make sure those at risk of carrying infections are excluded,
■ the testing of each donation and pools of plasma for the signs of virus infections.
The inclusion of steps in the processing of the blood or plasma that can inactivate or remove viruses.
O
Despite these measures, when medicines prepared from human blood or plasma are administered, the possibility of passing on infection cannot be totally excluded. This also applies to any unknown or emerging viruses or other types of infections. The measures taken are considered effective for enveloped viruses such as human immunodeficiency virus (HIV or AIDS vims), hepatitis B virus and hepatitis C virus. The measures taken may be of limited value against non-enveloped viruses such as hepatitis A virus and parvovirus B19. Parvovirus B19 infection may be serious pregnant women (fetal infection) and for individuals whose immune system is depressed or who have some types of anaemia (e.g. sickle cell disease or haemolytic anaemia).
Your doctor may recommend that you consider vaccination against hepatitis A and B if you regularly/repeatedly receive human plasma-derived factor IX products.
It is strongly recommended that every time you receive a dose of BETAFACT, the name and batch number of the medicine are recorded in order to maintain a record of the batches used.
Children
The listed warnings and precautions apply both to adults and children.
Other medicines and BETAFACT
Please tell your doctor or pharmacist if you are using, have recently used or might use any other medicines, including medicines obtained without a prescription.
No interactions between BETAFACT and other medicines have been observed to date.
Pregnancy and breast-feeding
The use of BETAFACT has not been evaluated in women who are pregnant or breast-feeding.
If you are pregnant or breast-feeding, your doctor should evaluate the benefits of treatment with BETAFACT. The potential benefit of this treatment should be compared with the risks involved.
If you are pregnant or breast-feeding, think you may be pregnant or are planning to have a baby, ask your doctor or pharmacist for advice before taking any medicine. Driving and using machines
Nothing suggests that factor IX has an effect on the ability to drive or use machines. BETAFACT contains sodium
This medicine contains around 2.6 mg of sodium per ml of product (13 mg per 5 ml vial, 26 mg per 10 ml vial, 52 mg per 20 ml vial). You should take this into account if you are on a salt-free or low-salt diet.
BETAFACT contains heparin
This medicine can cause allergic reactions and a fall in the number of blood cells, which may affect coagulation.
3. How to use BETAFACT
Treatment should be initiated under the supervision of a physician experienced in the treatment of haemophilia.
Dose
Your doctor will indicate the appropriate dose of BETAFACT.
The appropriate dose and frequency depend on the following:
■ your weight,
■ the severity of your haemophilia,
■ the location and extent of bleeding,
■ your health condition,
■ and, in certain cases, the operation you are to undergo (e.g.: surgical procedure, tooth extraction, etc.).
Your doctor will recommend that you undergo blood tests during treatment to control:
■ factor IX levels,
■ the presence of factor IX inhibitors.
Based on the results of these tests, your doctor may decide to adapt the dose and frequency of your injections.
The appropriate dose is expressed in the number of units (IU).
Frequency of administration
Your doctor will indicate how often BETAFACT injections must be administered.
Your doctor will adapt the frequency of the injections for you based on the severity of your bleeding and the efficacy of the treatment.
A table describing the frequency and duration of treatment for various situations is shown at the end of this leaflet in the section reserved for health professionals.
Mode and route of administration
This medicine should be injected into the veins by infusion.
If you have further questions on use of this product, ask your doctor or pharmacist. If you use more BETAFACT than you should
Immediately contact your doctor or pharmacist.
However, no case of overdose has been reported with human coagulation factor IX.
If you forget to use BETAFACT
Do not use a double dose to make up for a forgotten dose.
4. Possible side effects
Like all medicines, this medicine can cause side effects, although not everybody gets them.
Risk of allergic reactions
■ Hypersensitivity or allergic reactions can occur rarely. In some cases, these reactions have progressed to a serious allergic reaction.
■ Allergic reactions may occur together with the development of factor IX inhibitor and may affect kidney function (see also section 2. “Risk of allergic reactions”).
The warning signs of allergic reactions are :
■ swelling of the face or throat,
■ feeling of burning and tingling at the injection site,
■ chills,
■ redness,
■ itching and rash,
■ low blood pressure,
■ extreme fatigue (lethargy),
■ feeling sick (nausea), vomiting,
■ restlessness,
■ fast heart rate,
■ tightness of the chest,
■ pins and needles,
■ wheezing (asthma-like).
If one of these effects occurs, immediately stop the treatment and alert a doctor to start appropriate treatment depending on the type and severity of the reaction.
The following adverse reactions have been directly observed with Betafact in clinical studies and can occur rarely (may affect up to 1 in 1,000 injections):
e
■ hypersensitivity and allergic reactions (see also section 2 and 4 “Risk of allergic reactions”),
■ headache,
■ pruritus
■ allergic oedema
■ feeling sick (nausea),
■ injection reactions (malaise, chest pain),
■ reaction at injection site.
Two cases of activity-neutralizing antibodies (inhibitors) in one previously untreated patient and in one previously treated patient have been reported with BETAFACT in the post-marketing period.
The following adverse reactions have not been observed in clinical studies with Betafact but have been observed in patients using the same family drug as Betafact:
Blood clots
Blood clots can occur with the use of low-purity factor IX preparations. It may:
■ block the supply of blood and oxygen to the heart and cause heart attack.
■ block the supply of blood and oxygen to the lungs and cause a serious condition called pulmonary embolism,
■ cause clot in a vein (venous thrombosis),
■ cause blood clots inside the blood vessels throughout the body (disseminated intravascular coagulation).
Betafact is a high-purity factor IX and it is rarely associated with this type of effect. Inhibitors development
■ Patients using factor IX preparations may develop anti-factor IX antibodies (so-called inhibitors - see section 2. “Risk of allergic reactions”).
■ These inhibitors were not observed in clinical trials performed with Betafact on 11 previously-untreated patients.
Reporting of side effects
If you get any side effects, talk to your doctor, pharmacist or nurse. This includes any possible side effects not listed in this leaflet. You can also report side effects directly via ‘national reporting system'. By reporting side effects you can help provide more information on the safety of this medicine.
5. How to store BETAFACT
Keep this medicine out of the sight and reach of children.
Do not use this medecine after the expiry date which is stated on the vial label and carton.
Store in a refrigerator (2°C to 8°C). Do not freeze.
Keep the vial in the outer carton in order to protect from light.
The product should be used immediately after reconstitution.
For the purpose of ambulatory use, before opening, the product may be taken out of the refrigerator, without being replaced, for a maximum period of 6 months at a temperature not above 25°C. The date when the medicine is taken out and the new expiry date should be written on the outer packaging. This new expiry date should never exceed the one initially mentioned on the outer carton. If the medicine has not been used before the new expiry date, it should be disposed of. Do not use this medecine if the solution is cloudy or if it has deposits.
Do not throw away any medicine via wastewater or household waste. Ask your pharmacist how to throw away medicines you no longer use. These measures will help protect the environment.
6. Contents of the pack and other information
What BETAFACT contains
The active substance is human coagulation factor IX at the concentration of 50 lU/ml after reconstitution. Once reconstituted with 5, 10 or 20 ml of water for injections, one vial contains 250 IU/5 ml, 500 IU/10 ml or 1000 IU/20 ml of human coagulation factor IX.
The specific activity of BETAFACT is an average of 110 lU/mg total protein.
The other ingredients are:
sodium chloride, heparin sodium, lysine hydrochloride, arginine, sodium citrate and water for injections : see section 2. “What you need to know before you use BETAFACT”)
What BETAFACT 50 lU/ml, looks like and contents of the pack
BETAFACT is presented as a powder and a solvent for solution for injection in glass vials, a transfer system and a filter needle. BETAFACT is available in pack sizes of 250 IU/5 ml, 500 IU/10 ml and 1000 IU/20 ml.
Marketing Authorisation Holder and Manufacturer:
LFB-BIOMEDICAMENTS 3 avenue des Tropiques - BP 305 - Les Ulis 91958 Courtaboeuf Cedex - FRANCE
This medicinal product is authorised in the Member States of the EEA under the following names:
Austria: BETAFACT 50 I.E./ml Czech republic: BETAFACT 50 lU/ml France: BETAFACT 50 Ul/ml Germany: BETAFACT 250 I.E./ 500 I.E./1000 I.E.
Greece: BETAFACT 50 lU/ml Hungary: BETAFACT 50 NE/ml Poland: BETAFACT 250 IU/500 IU/1000 IU Portugal: BETAFACT 50 Ul/ml
Romania: BETAFACT 50 Ul/ml, pulbere si solvent pentru solutie injectabila
Slovak Republic: BETAFACT 50 lU/ml
Spain: BETAFACT 50 lU/ml
The Netherlands: BETAFACT 50 I.E/ml
United Kingdom: BETAFACT 50 lU/ml
This leaflet was last revised in
The following information is intended for healthcare professionals only:
The appropriate dose is expressed in the number of units (IU) calculated using the following equation:
Number of units to be administered = body weight (kg) x
target increase in factor IX levels (%) (lU/dL) x 0.93
Posology
For each of the bleeding episodes listed below, factor IX activity should not fall below the given level in the corresponding period. This table can be used to determine effective dosage in the following bleeding episodes and surgeries:
0000000-000000/0.0