Betoptic Suspension Single Dose 0.25% W/V Eye Drops
BETOPTIC®
Suspension Single Dose 0.25% w/v eye drops
Betaxoloi (as hydrochloride)
Read all of this leaflet carefully before you start using this medicine
• Keep this leaflet. You may need to read it again.
• If you have any further questions, ask your doctor or your pharmacist.
• This medicine has been prescribed for you. Do not pass it on to others. It may harm them, even if their symptoms are the same as yours.
• If any of the side effects get serious, or if you notice any side effects not listed in this leaflet, please tell your doctor or pharmacist.
IN THIS LEAFLET
1. What BETOPTIC Suspension Single Dose is and what it is used for
2. Before you use BETOPTIC Suspension Single Dose
3. How to use BETOPTIC Suspension Single Dose
4. Possible side effects
6. How to store BETOPTIC Suspension Single Dose 6. Further information
1. WHAT BETOPTIC SUSPENSION SINGLE DOSE IS AND WHAT IT IS USED FOR
BETOPTIC Suspension Single Dose belongs to a group of medicines known as beta blockers.
It is used to treat glaucoma or ocular hypertension (high pressure in the eye) by reducing the fluid pressure in your eye(s).
2. BEFORE YOU USE BETOPTIC SUSPENSION SINGLE DOSE
Do not use BETOPTIC Suspension Single Dose eye drops, suspension...
• If you are allergic to betaxoloi, beta-blockers or any of the other ingredients listed in section 6.
• Ifyouhavenoworhavehadinthepast, respiratoryproblemssuchas severe asthma, severe chronic obstructive bronchitis (severe lung condition which may cause wheeziness, difficulty in breathing and/or long-standing cough).
• If you have a slow heart beat, heart failure or disorders of heart rhythm (irregular heartbeats).
Ask your doctor for advice.
Take special care...
Before you use this medicine, tell your doctor if you have now or have had in the past
• coronary heart disease (symptoms can include chest pain or tightness, breathlessness or choking), heart failure, low blood pressure (hypotension)
• disturbances of heart rate such as slow heart beat (bradycardia)
• breathing problems, asthma or chronic obstructive pulmonary disease (lung disease which may cause wheeziness, difficulty in breathing and/or long-standing cough)
• poor blood circulation disease (such as Raynaud's disease or Raynaud's syndrome)
• diabetes, as betaxoloi may mask the signs and symptoms of low blood
• overactivity of the thyroid gland as betaxoloi may mask the signs and symptoms
• muscle weakening disease such as myasthenia gravis. Your symptoms may get worse
• angle-closure glaucoma
• dry eyes (Sicca Syndrome)
Tell your doctor before you have an operation that you are using Betoptic as betaxoloi may change the effects of some medicines used during anaesthesia.
If any of these apply you may still be able to use BETOPTIC Suspension Single Dose, but discuss it with your doctor first.
Using other medicines
BETOPTIC Suspension Single Dose can affect or be affected by other medicines you are using including other eye drops for the treatment of glaucoma. Tell your doctor if you are using or intend to use medicines to lower blood pressure, heart medicine, medicines to treat diabetes or medicines to treat emotional, behavioural or mental disorders such as anxiety or depression.
BETOPTIC Suspension Single Dose may reduce the effectiveness of adrenaline, which can be used to treat serious allergic reactions
429736

(anaphylaxis). Tell your doctor if you have a history of anaphylaxis or allergic reactions.
Please tell your doctor or pharmacist if you are taking or have recently taken any other medicines, including medicines obtained without a prescription.
If you are using more than one type of eye drop, wait S minutes between using each one. Eye ointments should be administered last. Pregnancy and breast-feeding
Do not use BETOPTIC Suspension Single Dose if you are pregnant unless your doctor considers it necessary.
Do not use BETOPTIC Suspension Single Dose if you are breastfeeding. Betaxoloi may get into your breast rnilk.
Ask your doctor for advice before taking any medicine during breast- feeding. Driving and using machines
If your sight is affected in any way following the use of BETOPTIC Suspension Single Dose, you should not drive or operate machinery. Important information if you wear Contact Lenses
Do not use the drops while wearing contact lenses. Wait at least 15 minutes after use before putting your lenses back in.
3. HOWTO USE BETOPTIC SUSPENSION SINGLE DOSE
BETOPTIC Suspension Single Dose should only be used in the eye(s). The usual dose is...
The usual dose is 1 drop in the affected eye(s) twice daily.
Not recommended for use in CHILDREN.
Always use BETOPTIC Suspension Single Dose exactly as your doctor has told you. You should check withyour doctor or pharmacist if youare not sure. How to use
• Wash your hands before you start.
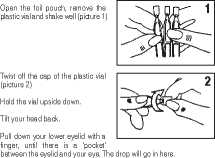
• Bring the tip of the vial close to the eye. Do this in front of a mirror if it
• Do not touch your eye or eyelid,
surrounding areas or other surfaces with the tip of the vial. It could infect the drops.
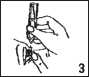
• Gently squeeze the vial so that one drop falls into the gap between the eye and the lower lid (picture 3).

'RAAPftKlVAL The undersigned:

Signature and date:
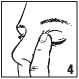
• Release the lower eyelid. Alter using BETOPTIC Suspension Single Dose, press a finger into the corner of your eye by the nose (picture 4) (or 2 minutes. This helps to stop betaxoloi getting into the rest o( the body.
• l( you use drops in both eyes, repeat the steps (or your other eye.
• Dispose ot the vial alter use. Do not keep to use again.
• If a drop misses your eye, tryagain.
• If you miss a dosejusttakeitassoonas possible. However, it it is almost time tor your next dose, skip the missed dose and go back to your regular dosing schedule.
Do not use a double dose to make up for a missed dose.
• If you use more BETOPTIC Suspension Single Dose than you should it can be washed out of your eye with warm water.
If you have any further questions on the use of BETOPTIC Suspension Single Dose, ask your doctor or pharmacist.
4. POSSIBLE SIDE EFFECTS
Like all medicines, BETOPTIC Suspension Single Dose ran cause side effects, although not everybody gets them.
You can usually carry on taking the drops, unless the effects are serious. If you are worried, talk to a doctor or pharmacist. Do not stop using BETOPTIC Suspension Single Dose without speaking to your doctor. Like other medicines applied into eyes, betaxoloi is absorbed into the blood. This may cause similar side effects as seen with beta-blockers given by injection or taken by mouth. Incidence of sideeffects after beta-blockers are used in the treatment of eye conditions is lower than when medicines are, for e ample, taken by mouth or injected. Listed side effects include reactions seen following treatment with Betaxoloi eye drops and within the class of other beta-blockers used for treating eye conditions.
Side effects experienced by patients during clinical trials with BETOPTIC Suspension Single Doseare:
Very common: may ailed more than 1 in 10 users
• Eye discomfort (includes a feeling of something in the eye)
Common (affects 1 to 10 users in 100):
• Blurred vision, watery eyes
• Headache
Uncommon (affects 1 to 10 users in 1000):
• Inflammation of the eye surface, conjunctivitis or symptoms of conjunctivitis, visual impairment, sensitivity to light, painful, dry or tired eyes, excessive blinking, irritated, red or swollen eyes, a feeling of something in the eye, eye itchiness eye discharge, weeping eyelids, bloodshot eyes
• Slow heart beat or unusually rapid heart beat
• Asthma, difficulty breathing, blocked nose
• Nausea
Rare (affects 1 to 10 users in 10,000):
• Cataract formation, decreased sensitivity of the eye, inflammation of the eyelid
• Anxiety, difficulty sleeping (insomnia}, depression
• Fainting
• Low blood pressure
• Cough, runny nose
• Taste disturbances
• Inflamed, itchy skin or rash, hair loss
• Libido decreased
The following side effects have also been reported by people using BETOPTIC Suspension Single Dose. The frequency cannot be estimated from the available data:
• Hypersensitivity reaction
• Changes in the rhythm or speed of the heartbeat
• Loss or lack of strength
Additional side effects have been seen with other ophthalmic beta-blockers and could potentially occur with BETOPTIC Suspension Single Dose. The frequency is unknown:
• Generalised allergic reactions including swelling beneath the skin (that can occur in areas such as the face and limbs, and can obstruct the airway which may cause difficulty swallowing or breathing}, hives (or itchy rash}, localised and generalised rash, itchiness, severe sudden life-threatening allergic reaction.
• Low blood glucose levels.
• Nightmares, memory loss, hallucinations, delusions and confusion.
• Stroke, reduced blood supply to the brain, increases in signs and symptoms of myasthenia gravis (muscle disorder}, unusual sensations (like pins and needles}.
• Detachment of the layer below the retina that contains blood vessels following filtration surgery which may cause visual disturbances, corneal erosion (damage to the front layer of the eyeball}, drooping of the upper eyelid (making the eye stay half closed}, double vision.
• Chest pain, palpitations, oedema (fluid build-up}, congestive heart failure (heart disease with shortness of breath and swelling of the feet and legs due to fluid build-up}, a type of heart rhythm disorder, heart attack, heart failure.
• Raynaud's phenomenon, cold hands and feet with a blue colour, leg pains (especially if you have a history of poor circulation}.
• Constriction of the airways in the lungs (predominantly in patients with pre-existing disease).
• Indigestion, diarrhoea, dry mouth, abdominal pain, vomiting.
• Skin rash with white silvery coloured appearance (psoriasiform rash) or worsening of psoriasis.
• Muscle pain not caused by exercise.
• Sexual dysfunction, impotence.
• Tiredness.
An increased in Anti Nuclear Antibodies has also been seen in patients taking ophthalmic beta blockers.
Reporting of side effects
If you get any side effects, talk to your doctor, pharmacist or nurse. This includes any possible side effects not listed in this leaflet. You can also report side effects directly via the rational reporting system listed below. By reporting side effects, you can help provide more information on the safety of this medicine.
United Kingdom - Yellow Card Scheme
Website: www.rnhra.gov.uk/yellowcard
Ireland - HPRA Pharma covigilance
Earlsfort Terrace, IRL - Dublin 2; Tel: +353 1 6764971
Fax: +353 1 6762517. Website: www.hpra.ie; E-mail: rnedsafety@hpra.ie.
5. HOWTO STORE BETOPTIC SUSPENSION SINGLE DOSE
• Keep out of the reach and sight of children.
• Do not store above 25°C.
• Do not freeze.
• Store in the original package in order to protect from light.
• Do not use the drops after the expiry date (marked 'Exp'} on the sachet and the carton. The expiry date refers to the last day of that month.
• Once opened, use immediately.
• Dispose of the vial after use. Do not keep an open vial to use again.
• Medicines should not be disposed of via waste water or household waste. Ask your pharmacist how to dispose of medicines no longer required. These measures will help to protect the environment.
• Do not pass this medicine on to others. It may harm them even if their symptoms are the same as yours.
6. FURTHER INFORMATION
What BETOPTIC Suspension Single Dose contains
• The active substance is betaxoloi 0.25% w/v (2.5 rng/rnl) (as the hydrochloride). Each single dose dispenser contains 62Srnicrograrns of betaxoloi (as the hydrochloride).
• The other ingredients are arnberlite (Poly (styrene divinyl benzene} sulphonic acid), carborner, mannitol, hydrochloric acid and/or sodium hydroxide (to adjust pH} and purified water.
What BETOPTIC Suspension Single Dose looks like and contents of the pack
BETOPTIC Suspension Single Dose is a white to off-white liquid supplied in a plastic single-dose dispenser containing 0.25 rnl. Five dispensers are enclosed in a foil overwrap. Each pack contains 50 single dose dispensers.
Suitable for people who are sensitive to preservatives.
Marketing authorisation holder: Manufacturer:
Alcon Laboratories (UK} Ltd SA Alcon-Couvreur NV
Frirnley Business Park Rijksweg 14, B-2870 Puurs, Belgium.
Frirnley Carnberley
Surrey, GU16 7SR
United Kingdom.
This leaflet was last revised in November 2015.
Alcori
© 2013,2015 Novartis a Nova rtis com pa ny
429736