Bocouture 50 Units Powder For Solution For Injection
Package leaflet: Information for the user BOCOUTURE 4 units/0.1 ml powder for solution for injection
Botulinum toxin type A (150 kD), free from complexing proteins
Read all of this leaflet carefully before you start using this medicine because it contains important information for you.
• Keep this leaflet. You may need to read it again.
• If you have any further questions, ask your doctor, pharmacist or nurse.
• This medicine has been prescribed for you only. Do not pass it on to others. It may harm them, even if their signs of illness are the same as yours.
• If you get any side effects, talk to your doctor, pharmacist or nurse. This includes any possible side effects not listed in this leaflet. See section 4.
What is in this leaflet:
1. What BOCOUTURE is and what it is used for
2. What you need to know before you use BOCOUTURE
3. How to use BOCOUTURE
4. Possible side effects
5. How to store BOCOUTURE
6. Contents of the pack and other information
1. What BOCOUTURE is and what it is used for
BOCOUTURE is a medicine that relaxes the muscles.
It works by blocking the nerve impulses in all injected muscles. This prevents muscles from contracting leading to temporary and reversible relaxation.
BOCOUTURE is used for temporary improvement of vertical frown lines between the eyebrows and of lateral periorbital lines seen at maximum smile in adults under 65 years of age when the severity of these lines has an important psychological impact for the patient.
2. What you need to know before you use BOCOUTURE Do not use BOCOUTURE
• if you are allergic to Botulinum neurotoxin type A or any of the other ingredients of this medicine (listed in section 6)
• if you suffer from a generalised disorder of muscle activity (e.g. myasthenia gravis, Lambert-Eaton syndrome)
• if you have an infection or inflammation at the proposed injection site.
Warnings and precautions
Side effects may occur from misplaced injections of Botulinum neurotoxin type A temporarily paralysing nearby muscle groups._There have been very rare reports of side effects that may be related to the spread of Botulinum toxin far from the injection site (e.g. excessive muscle weakness, swallowing difficulties or accidental swallowing of food or drink into the airways). Patients who receive the recommended doses may experience excessive muscle weakness.
If the dose is too high or the injections too frequent, the risk of antibody formation may increase. Antibody formation can cause treatment with Botulinum toxin type A to fail, whatever the reason for its use.
Talk to your doctor before BOCOUTURE is used:
• if you suffer from any type of bleeding disorder
• if you receive substances that prevent the blood from clotting (e.g. coumarin, heparin, acetylsalicylic acid, clopidogrel)
• if you suffer from pronounced weakness or decreased muscle volume in the muscle where you will receive the injection
• if you suffer from amyotrophic lateral sclerosis (ALS), which can lead to generalised muscle decrease
• if you suffer from any disease that disturbs the interaction between nerves and skeletal muscles (peripheral neuromuscular dysfunction)
• if you have or have had swallowing difficulties
• if you have had problems with injections of Botulinum toxin type A in the past
• if you are due to have surgery
Repeated injections with BOCOUTURE
If you have repeated injections with BOCOUTURE, the effect may increase or decrease. Possible reasons for this are:
• your doctor may follow a different procedure when preparing the solution for injection
• injections into another muscle
• non-response/treatment failure in the course of the treatment Your doctor will clarify the reasons for this and discuss them with you.
If you develop swallowing, speech or breathing difficulties, please seek immediate medical care or ask your relatives to do so (see section 4).
Special Populations
There is limited information on the treatment of patients over 65 years of age. Therefore, BOCOUTURE should not be used in patients over the age of 65 years.
Children and adolescents
As there is no information regarding the use in children and adolescents, BOCOUTURE should not be given to patients under the age of 18 years.
Other medicines and BOCOUTURE
Tell your doctor or pharmacist if you are taking, have recently taken or might take any other medicines. The effect of BOCOUTURE may be increased by:
• medicines used to treat certain infectious diseases (spectinomycin or aminoglycoside antibiotics [e.g. neomycin, kanamycin, tobramycin])
• other medicines that relax the muscles (e.g. muscle relaxants of the tubocurarine type). Such medicines are used, for example, in general anaesthesia. Before you have surgery, tell your anaesthetist if you have received BOCOUTURE.
In these cases, BOCOUTURE must be used carefully.
The effect of BOCOUTURE may be reduced by certain medicines for malaria and rheumatism (known as aminoquinolines).
Pregnancy, breast-feeding and fertility
If you are pregnant or breast-feeding, think you may be pregnant or are planning to have a baby, ask your doctor or pharmacist for advice before this medicine is administered.
BOCOUTURE should not be used during pregnancy unless your doctor decides that the necessity and potential benefit of the treatment justifies the possible risk on the foetus. BOCOUTURE is not recommended if you are breast-feeding.
Driving and using machines
You should not drive or engage in other potentially hazardous activities if drooping eyelids, weakness (asthenia), muscle weakness, dizziness or vision disorders occur.
If in doubt, ask your doctor for advice.
3. How to use BOCOUTURE
BOCOUTURE may only be administered by doctors with appropriate specialist knowledge of treatment with Botulinum neurotoxin.
Dissolved BOCOUTURE is intended for injections into the muscle (intramuscular use; see information for medical and healthcare professionals at the end of this leaflet).
The interval between two treatments should not be less than 3 months.
Vertical Lines between the Eyebrows (Glabellar Frown Lines)
When treating your vertical lines between the eyebrows (glabellar frown lines) the usual total dose is 20 units. Your doctor will inject the recommended volume of 0.1 ml (4 units) BOCOUTURE into each of the 5 injection sites. The total dose may be increased by the physician to up to 30 units if required by the individual needs of the patient, with at least ‘3-months’ interval between treatments.
A reduction in the vertical lines between the eyebrows generally occurs within 2 to 3 days with the maximum effect observed on day 30. The effect lasts for up to 4 months after the injection.
Lateral Periorbital Lines seen at maximum smile (Crow’s feet lines)
For the treatment of lateral periorbital lines seen at maximum smile (crow’s feet lines) your doctor will inject a standard dose of 24 units (12 units per eye). The recommended volume of 0.1 ml with 4 units will be applied bilaterally in 3 injection sites and will result in a total injection volume of 0.3 ml per eye.
A reduction in lateral periorbital lines seen at maximum smile mostly occurs within 6 days with the maximum effect observed on day 14. The effect lasts for up to 3 months after the injection.
If you are given more BOCOUTURE than you require
Symptoms of overdose
Symptoms of overdose are not apparent immediately after the injection and may include general weakness, drooping eyelid, double vision, breathing difficulties, speech difficulties, and paralysis of the respiratory muscles or swallowing difficulties which may result in pneumonia.
Measures in cases of overdose
In case you feel symptoms of overdose please seek medical emergency services immediately or ask your relatives to do so, and have yourself admitted to hospital. Medical supervision for up to several days and assisted ventilation may be necessary.
If you have any further questions on the use of this medicine, ask your doctor, pharmacist or nurse.
4. Possible side effects
Like all medicines, this medicine can cause side effects, although not everybody gets them.
An allergic reaction may occur with BOCOUTURE. Serious immediate allergic reactions (anaphylaxis) or allergic reactions to the serum in the product (serum sickness) causing for example difficulty in breathing (dyspnoea) (, hives (urticaria) or swelling of the soft tissue (oedema) have been rarely reported. Some of these reactions have been observed following the use of conventional Botulinum toxin type A complex. They occurred when the toxin was given alone or in combination with other products known to cause similar reactions. These effects cannot be ruled out completely when using BOCOUTURE.
An allergic reaction can cause any of the following symptoms:
• difficulty with breathing, swallowing or speaking due to the swelling of the face, lips, mouth or throat
• swelling of the hands, feet or ankles.
If you notice any of these side effects, please inform your doctor immediately or ask your relatives to do so and go to the accident and emergency department of your nearest hospital.
Side effects may be related to the medicine, injection technique or both. At the injection sites, the action of Botulinum toxin causes localised muscle weakness. Drooping eyelids can be caused by the injection technique and the effect of the medicine. Usually, side effects occur in the first week after the injection and are temporary in nature.
The following reactions may occur around the injection site:
• Pain
• Inflammation
• Pins and needles
• Decreased skin sensation
• Tenderness
• Itching
• Swelling (general)
• Swelling of the soft tissue (oedema)
• Skin redness (erythema)
• Localised infection
• Haematoma
• Bleeding
• Bruising
Patients with a fear of injections may experience temporary generalised reactions, e.g.
• Fainting
• Circulatory problems
• Nausea
• Noises in the ears
Other possible side effects
Vertical Lines between the Eyebrows (Glabellar Frown Lines)
The following side effects have been observed when using BOCOUTURE:
Common (may affect up to 1 in 10 people):
• Headache, muscle activity disorders (raised eyebrows)
Uncommon (may affect up to 1 in 100 people):
• Nasopharyngitis
• Bronchitis
• Influenza like illness
• Localised muscle weakness in the face (drooping eyebrow, brow ptosis)
• Drooping eyelids (eyelid ptosis)
• Accumulation of fluid in the eyelids (eyelid oedema)
• Blurred vision
• Muscle twitching (muscle spasm)
• Feeling of heaviness at the upper part of the face
• Feeling of tightness at the injection site
• Tiredness
• Pain or bruising at the injection site
• Itching
• Skin nodule
• Depression
• Insomnia
Lateral Periorbital Lines seen at maximum smile (Crow’s feet lines)
The following side effects have been observed when using BOCOUTURE:
Common (may affect up to 1 in 10 people):
• Eyelid swelling
• Dry eye
• Bruising at the injection site
Post-marketing experience
There have been reports of flu-like symptoms and hypersensitivity reactions, such as swelling, swelling of the soft tissue (oedema), redness, itching, rash (local and generalised) and breathlessness.
The following very rare side effects have been observed during Botulinum toxin treatment of conditions other than vertical lines between the eyebrows or lateral periorbital lines seen at maximum smile (see section 2):
• Excessive muscle weakness
• Swallowing difficulties
• Swallowing difficulties causing inhalation of foreign bodies resulting in lung inflammation and, in some cases, death
Such side effects are caused by the relaxation of muscles far from the injection site.
Reporting of side effects
If you get any side effects, talk to your doctor, pharmacist or nurse. This includes any possible side effects not listed in this leaflet. You can also report side effects directly via the Yellow Card Scheme at www.mhra.gov.uk/yellowcard. By reporting side effects, you can help provide more information on the safety of this medicine.
5. How to store BOCOUTURE
Keep this medicine out of the the sight and reach of children.
Do not use this medicine after the expiry date which is stated on the carton and vial label after "EXP". The expiry date refers to the last day of that month.
Unopened vial: Do not store above 25°C.
Reconstituted solution: Chemical and physical in-use stability has been demonstrated for 24 hours at 2°C to 8°C.
From a microbiological point of view, the product should be used immediately. If not used immediately, in-use storage times and conditions prior to use are the responsibility of the user and would normally not be longer than 24 hours at 2°C to 8°C, unless reconstitution has taken place in controlled and validated aseptic conditions.
Your doctor should not use BOCOUTURE if the solution has a cloudy appearance or contains visible particles.
For instructions on disposal, please see information for medical and healthcare professionals at the end of this leaflet.
6. Contents of the pack and other information What BOCOUTURE contains
• The active substance is: Botulinum toxin type A (150 kD), free from complexing proteins. One vial contains 50 units Botulinum toxin type A (150 kD), free from complexing proteins. 0.1 ml solution contains 4 units of Botulinum toxin type A (150 kD), free from complexing proteins when reconstituted in 1.25 ml.
• The other ingredients are: human albumin, sucrose.
What BOCOUTURE looks like and contents of the pack
BOCOUTURE is presented as a powder for solution for injection. The powder is white.
Dissolving the powder produces a clear, colourless, particle-free solution.
Pack sizes of 1, 2, 3 or 6 vials.
Not all pack sizes may be marketed.
Marketing Authorisation Holder
Merz Pharmaceuticals GmbH Eckenheimer Landstrahe 100 60318 Frankfurt/Main Germany
Manufacturer
Merz Pharma GmbH & Co. KGaA Eckenheimer Landstrahe 100 60318 Frankfurt/Main P.O. Box 11 13 53 60048 Frankfurt/Main Germany
Telephone: +49-69/15 03-1 Fax: +49-69/15 03-200
This leaflet was last revised in 12/2015.
The following information is intended for medical or healthcare professionals only:
Instructions for reconstitution of the solution for injection:
50 units of BOCOUTURE are reconstituted prior to use in 1.25 ml unpreserved sodium chloride 9 mg/ml (0.9%) solution for injection. This corresponds to a concentration of 40 units/ml. Reconstitution and dilution should be performed in accordance with good clinical practice guidelines, particularly with respect to asepsis.
BOCOUTURE may only be applied for its intended use to treat one patient for one session.
It is good practice to reconstitute the vial contents and prepare the syringe over plastic-lined paper towels to catch any spillage. An appropriate amount of sodium chloride solution is drawn up into a syringe. After vertical insertion of the needle through the rubber stopper, the solvent is injected gently into the vial in order to avoid foam formation. A 20-27 G short bevel needle is recommended for reconstitution. The vial must be discarded if the vacuum does not pull the solvent into the vial. Remove
the syringe from the vial and mix BOCOUTURE with the solvent by carefully swirling and inverting the vial - do not shake vigorously. If needed, the needle used for reconstitution should remain in the vial and the required amount of solution should be drawn up with a new sterile syringe suitable for injection.
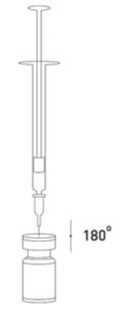
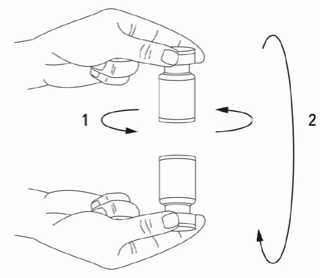
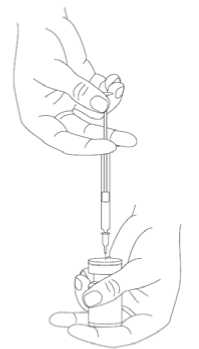
Reconstituted BOCOUTURE is a clear, colourless solution free of particulate matter.
BOCOUTURE must not be used if the reconstituted solution (prepared as above) has a cloudy appearance or contains floccular or particulate matter.
Instructions for disposal
Any solution for injection that has been stored for more than 24 hours as well as any unused solution for injection must be discarded.
Procedure to follow for a safe disposal of vials, syringes and materials used Any unused vials, residual reconstituted solution in the vial and/or syringes should be autoclaved. Alternatively, the remaining BOCOUTURE can be inactivated by adding one of the following solutions: 70% ethanol, 50% isopropanol, 0.1% SDS (anionic detergent), diluted sodium hydroxide solution (0.1 N NaOH), or diluted sodium hypochlorite solution (at least 0.1% NaOCl).
After inactivation used vials, syringes and materials should not be emptied and must be discarded into appropriate containers and disposed of in accordance with local requirements.
Recommendations should any incident occur during the handling of Botulinum toxin
• Any spills of the product must be wiped up: either using absorbent material impregnated with any of the above solutions in case of the powder, or with dry, absorbent material in case of reconstituted product. The contaminated surfaces should be cleaned using absorbent material impregnated with any of the above solutions, then dried.
• If a vial is broken, proceed as mentioned above by carefully collecting the pieces of broken glass and wiping up the product, avoiding any cuts to the skin.
• If the product comes into contact with skin, rinse the affected area abundantly with water.
• If product gets into the eyes, rinse thoroughly with plenty of water or with an ophthalmic eyewash solution.
• If product comes into contact with a wound, cut or broken skin, rinse thoroughly with plenty of water and take the appropriate medical steps according to the dose injected.
These instructions for use, handling and disposal should be strictly followed.
25