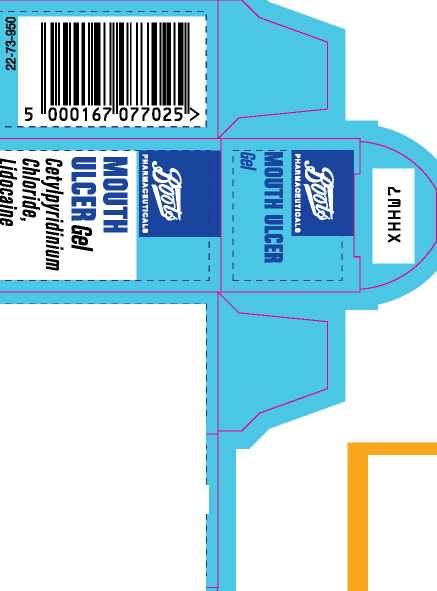
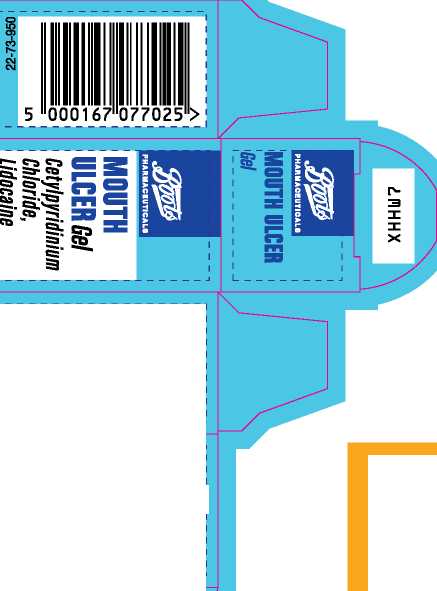
Boots Mouth Ulcer Gel
180 mm
'Active ingredients
' This oral gel contains Cetylpyridinium Chloride 0.02% w/w,
Lidocaine 0.6% w/w.
Also contains: purified water, ethanol (33.9 vol %), sucrose, hypromellose, cetomacrogol 1000, star anise oil, eucalyptol, levomenthol. PL 00014/0150 Text prepared 12/14
Manufactured tor the Marketing Authorisation Holder The Boots Company PLC, Nottingham, NG2 3AA by_BCM Ltd^ Nottingham NG2_3AA________
15g_e

If you need more advice ask your pharmacist.


mm
Read all of this carton for fuH instmctions.
What this medicine is for
An antiseptic gel for the rapid relief of common mouth ulcer pain.
Before you use this medicine
XDonotuse:
• If you are allergic to any of the ingredients You can use this medicine if you are pregnant or breastfeeding.
/How to use this medicine
Check the tube seal is not broken before first use.
If it is, do not use the gel.
Pierce tube seal with end of cap.
Adults and children: Apply a small amount of gel to the affected area with a clean finger, every 3 hours during the day, if you need to.
Mouth Ulcer Gel is not suitable for teething troubles _ _ in babies.
If symptoms do not go away within 7 days talk to your doctor or dentist.
If you use too much: Talk to a pharmacist or doctor.
Possible side effects
Most people will not have problems, but some may get some. If you get any of these serious side effects, stop using the gel. See a doctor at once:
i* Difficulty in breathing, swelling of the face, neck, tongue or throat (severe allergic reactions)
______«, ofside effects
If you get any side effects, talk to your doctor, pharmacist or nurse. This includes any possible side effects not listed in this leaflet. You can also report side effects directly via the Yellow Card Scheme at: www.mhragov.uk/yellowcard. By reporting side effects you can help provide more information on the safety of this medicine.
How to store this medicine
Do not store above 25°C. Keep all medicines out of the sight and reach of children.
Use by the date on the end flap of the carton.
TECHNICAL INFO: PRINTER PLEASE NOTE
Trident have created this artwork to a generic print process specification and repro has not been applied. Please call your Boots contact to discuss any amends or if anything falls beneath your print tolerances.
|
Trident Reference No: |
BTC171242 |
|
Zen Ref: |
TR898287 |
|
Category: |
Healthcare |
|
Sub-Category: |
Mouth Care |
|
Brand: |
Core |
|
Pack Type: |
Carton |
|
Variant: |
Boots Mouth Ulcer Gel 15g - Carton |
|
Action: |
A |
|
Date: |
08/12/14 |
|
Country: |
UK |
|
Component Code: |
XHHW7 |
|
Item Code: |
22-73-950 |
|
CAD Ref No: |
26x24x106mm |
|
Printer: |
N/A |
|
Substrate: |
White Carton Board |
|
Barcode Type: |
EAN 13 |
|
Barcode Number: |
5000167077025 |
|
Magnification: |
80% |
|
Barcode Truncated By: 4.3 mm | |
|
(smallest bar) | |
|
Edgemark Position: |
N/A |
|
Pharmacode No/NE: |
N/A |
|
Technical Et Non Printing Items | |
|
Cutter | Guides | |
|
Colours | |
|
Main Headings: |
apt |
|
Sub Headings: |
7pt |
|
Body Copy: |
6.25pt |
|
PACK 1VIOCK OP | |||
|
Product Name: |
Mouth Ulcer Gel | ||
|
Product Licence No.: |
00014/0150 | ||
|
Wording Ref: |
MHRA approved VI dated 04/02/2014 (BTC 12G506 action C] | ||
|
Status: |
Internally approved | ||
|
Pack Details: |
Internally lacquered aluminium tube (polyamide/polyimide lacquer system) with a membrane seal fitted with a wadless | ||
|
polyethylene cap or a polypropylene cap. | |||
|
Pack size: |
15 g | ||
|
Version No. |
Date Issued |
Reason For Change | |
|
1 |
03/12/14 |
Correct BCM postcode to NG2 3AA | |
'Active ingredients
' This oral gel contains Cetylpyridinium Chloride 0.02% w/w,
Lidocaine 0.6% w/w.
Also contains: purified water, ethanol (33.9 vol %), sucrose, hypromellose, cetomacrogol 1000, star anise oil, eucalyptol, levomenthol. PL 00014/0150 Text prepared 12/14
Manufactured tor the Marketing Authorisation Holder The Boots Company PLC, Nottingham, NG2 3AA by_BCM Ltd^ Nottingham NG2_3AA________
15g_e

If you need more advice ask your pharmacist.

breastfeeding.____________________in babies.

mm
Read all of this carton for fuH instmctions.
What this medicine is for
An ar+iseptr gel fo- the rapirt relief oS common mruth 1 ulcer Gain.
'Before you use this ruedicme
XDo not us«=.
• If you are allergic to any of the ingredients You can use this medicine if you are pregnant or
/How to use this medicine
Check the tube seal is not broken before first use. iritis, do ”otMse the gel.
Pierce +|jhe se^l with end of cap.
Adults and chilaren: Apply a small amount of gel to me infected area with a clean finger, every 3 hours during the day, if you need to.
Mouth Ulcer Gel is not suitable for teething troubles
If symptoms do not go away within 7 days talk to your doctor or dentist.
If you use too much: Talk to a pharmacist or doctor.
Pos&jle s.Je e.e-ts. •
Most people ./ill not have pnoLarr.L jt some rrUyfetsor! i. ? tfyot g la /of these serious ;de effects, stop using the gel. See a doctor at once:
i* Difficulty in breathing, swelling of the face, neck, tongue or throat (severe allergic reactions)
....______„ ofside effects
If you get any side effects, talk to your doctor, pharmacist or nurse. This includes any possible side effects not listed , Jn this leaflet. You can also report side effects directly via he Yellow Card Scheme at: www.mhragov.uk/yellowcard. 3y reporting side effects you can help provide more "formation on the safety of this medicine.
How to store this medicine
Do not store above 25°C. Keep all medicines out of the sight and reach of children.
Use by the date on the end flap of the carton.
TECHNICAL INFO: PRINTER PLEASE NOTE
Trident have created this artwork to a generic print process specification and repro has not been applied. Please call your Boots contact to discuss any amends or if anything falls beneath your print tolerances.
|
Trident Reference No: |
BTC171242 |
|
Zen Ref: |
TR898287 |
|
Category: |
Healthcare |
|
Sub-Category: |
Mouth Care |
|
Brand: |
Core |
|
Pack Type: |
Carton |
|
Variant: |
Boots Mouth Ulcer Gel 15g - Carton |
|
Action: |
A |
|
Date: |
08/12/14 |
|
Country: |
UK |
|
Component Code: |
XHHW7 |
|
Item Code: |
22-73-950 |
|
CAD Ref No: |
26x24x106mm |
|
Printer: |
N/A |
|
Substrate: |
White Carton Board |
|
Barcode Type: |
EAN 13 |
|
Barcode Number: |
5000167077025 |
|
Magnification: |
80% |
|
Barcode Truncated By: 4.3 mm | |
|
(smallest bar) | |
|
Edgemark Position: |
N/A |
|
Pharmacode No/NE: |
N/A |
|
Technical Et Non Printing Items | |
|
Cutter | Guides | |
|
Colours | |
|
BRAILLE |
CONVERSION | |
|
• • • •• • |
• •• •• | |
|
• • • |
• |
• • •• • |
|
• • •• • |
• |
• • • • |
|
• |
• •• |
• • • • |
|
• • • |
• |
• |
|
Main Pleadings: |
8pt |
|
Sub Headings: |
7pt |
|
Body Copy: |
6.25pt |
|
PACK 1VIOCK OP | |
|
Product Name: |
Mouth Ulcer Gel |
|
ProductLicenceNo.: 00014/0150 | |
|
Wording Ref: |
MHRA approved VI dated 04/02/2014 (BTC 12G506 action C] |
|
Status: |
Internally approved |
|
Pack Details: |
Internally lacquered aluminium tube (polyamide/polyimide lacquer system) with a membrane seal fitted with a wadless |
|
polyethylene cap or a polypropylene cap. | |
|
Pack size: |
15 g |
03/12/14
Reason For Change
Correct BCM postcode to NG2 3AA
113 mm





• • • •• • • • • •


• • • •
• ••
• • • • •
• •

TECHNICAL INFO: PRINTER PLEASE NOTE
Trident have created this artwork to a generic print process specification and repro has not been applied. Please call your Boots contact to discuss any amends or if anything falls beneath your print tolerances.
|
Trident Reference No: |
BTC171242 |
|
Zen Ref: |
TR898287 |
|
Category: |
Healthcare |
|
Sub-Category: |
Mouth Care |
|
Brand: |
Core |
|
Pack Type: |
Carton |
|
Variant: |
Boots Mouth Ulcer Gel 15g - Carton |
|
Action: |
A |
|
Date: |
08/12/14 |
|
Country: |
UK |
|
Component Code: |
XHHW7 |
|
Item Code: |
22-73-950 |
|
CAD Ref No: |
26x24x106mm |
|
Printer: |
N/A |
|
Substrate: |
White Carton Board |
|
Barcode Type: |
EAN 13 |
|
Barcode Number: |
5000167077025 |
|
Magnification: |
80% |
|
Barcode Truncated By: 4.3 mm | |
|
(smallest bar) | |
|
Edgemark Position: |
N/A |
|
Pharmacode No/NE: |
N/A |
|
Technical Et Non Printing Items | |
|
Cutter | Guides | |
|
Colours | |
|
Main Headings: |
apt |
|
Sub Headings: |
7pt |
|
Body Copy: |
6.25pt |
|
BRAILLE |
CONVERSION | |
|
• • • •• • |
• •• •• | |
|
• • • |
• |
• • •• • |
|
• • •• • |
• |
• • • • |
|
• |
• •• |
• • • • |
|
• • • |
• |
• |
|
PACK 1VIOCK OP | |||
|
Product Name: |
Mouth Ulcer Gel | ||
|
Product Licence No.: |
00014/0150 | ||
|
Wording Ref: |
MHRA approved VI dated 04/02/2014 (BTC 12G506 action C] | ||
|
Status: |
Internally approved | ||
|
Pack Details: |
Internally lacquered aluminium tube (polyamide/polyimide lacquer system) with a membrane seal fitted with a wadless | ||
|
polyethylene cap or a polypropylene cap. | |||
|
Pack size: |
15 g | ||
|
Version No. |
Date Issued |
Reason For Change | |
|
1 |
03/12/14 |
Correct BCM postcode to NG2 3AA | |
'Active ingredients
' This oral gel contains Cetylpyridinium Chloride 0.02% w/w, Lidocaine 0.6% w/w.
Also contains: purified water, ethanol (33.9 vol %), sucrose,
Typromellose, cetomacrogol 1000, star anise oil, eucalyptol, levomenthol. PL00014/0150 ~ ■
Text prepared 12/14 Manufactured tor the Marketing Authorisation Holder The Boots Company PLC, Nottingham, NG2 3AA by_BCM Ltd^ Nottingham NG2_3AA________
J5g.e_
000167 077025 >

mm J1
Read all of this carton for fuH instmctions.
What this medicine is for
An antiseptic gel for the rapid relief of common mouth ulcer pain.
Before you use this medicine
XDonotuse:
• If you are allergic to any of the ingredients You can use this medicine if you are pregnant or breastfeeding.
/How to use this medicine
Check the tube seal is not broken before first use.
If it is, do not use the gel.
Pierce tube seal with end of cap.
Adults and children: Apply a small amount of gel to the affected area with a clean finger, every 3 hours during the day, if you need to.
Mouth Ulcer Gel is not suitable for teething troubles _ _ in babies.___________________

If you need more advice ask your pharmacist.

If symptoms do not go away within 7 days talk to your doctor or dentist.
If you use too much: Talk to a pharmacist or doctor.
Possible side effects
Most people will not have problems, but some may get some. If you get any of these serious side effects, stop using the gel. See a doctor at once:
i* Difficulty in breathing, swelling of the face, neck, tongue or throat (severe allergic reactions)
of side effects
If you get any side effects, talk to your doctor, pharmacist ------e. This includes any possible side effects
or nurse. This includes any possible side effects not listed in this leaflet. You can also report side effects directly via the Yellow Card Scheme at: www.mhra.gov.uk/yellowcard, By reporting side effects you can help provide more irmation on the safety of this medicine.
How to store this medicine
Do not store above 25°C. Keep all medicines out of the sight and reach of children.
Use by the date on the end flap of the carton.

ARTWORK ONLY


TECHNICAL INFO: PRINTER PLEASE NOTE
Trident have created this artwork to a generic print process specification and repro has not been applied. Please call your Boots contact to discuss any amends or if anything falls beneath your print tolerances.
|
Category: |
Healthcare |
|
Sub-Category: |
Mouth Care |
|
Brand: |
Core |
|
Pack Type: |
Carton |
|
Variant: |
Boots Mouth Ulcer Gel 15g - Carton |
|
Action: |
A |
|
Date: |
08/12/14 |
|
Country: |
UK |
|
Component Code: |
XHHW7 |
|
Item Code: |
22-73-950 |
|
CAD Ref No: |
26x24x106mm |
|
Printer: | |
|
Substrate: |
White Carton Board |
|
Barcode Type: |
EAN 13 |
|
Barcode Number: |
5000167077025 |
|
Magnification: |
80% |
|
Barcode Truncated By: 4.3 mm | |
|
(smallest bar) | |
|
Edgemark Position: |
N/A |
|
Pharmacode No/NE: |
N/A |
|
Technical Et Non Printing Items | |
|
Cutter | Guides | |
|
Colours | |
|
Main Headings: |
apt |
|
Sub Headings: |
7pt |
|
Body Copy: |
6.25pt |
|
PACK MOCK OP | |||
|
Product Name: |
Mouth Ulcer Gel | ||
|
Product Licence No.: |
00014/0150 | ||
|
Wording Ref: |
MHRA approved VI dated 04/02/2014 (BTC 12G506 action C] | ||
|
Status: |
Internally approved | ||
|
Pack Details: |
Internally lacquered aluminium tube (polyamide/polyimide lacquer system) with a membrane seal fitted with a wadless | ||
|
polyethylene cap or a polypropylene cap. | |||
|
Pack size: |
15 g | ||
|
Version No. |
Date Issued |
Reason For Change | |
|
1 |
03/12/14 |
Correct BCM postcode to NG2 3AA | |

113 mm






7
TECHNICAL INFO: PRINTER PLEASE NOTE
Trident have created this artwork to a generic print process specification and repro has not been applied. Please call your Boots contact to discuss any amends or if anything falls beneath your print tolerances.
|
Trident Reference No: BTCl 71242 | |
|
Zen Ref: |
TR898287 |
|
Category: |
Healthcare |
|
Sub-Category: |
Mouth Care |
|
Brand: |
Core |
|
Pack Type: |
Carton |
|
Variant: |
Boots Mouth Ulcer Gel 15g - Carton |
|
Action: |
A |
|
Date: |
08/12/14 |
|
Country: |
UK |
|
Component Code: |
XHHW7 |
|
Item Code: |
22-73-950 |
|
CAD Ref No: |
26x24x106mm |
|
Printer: |
N/A |
|
Substrate: |
White Carton Board |
|
Barcode Type: |
EAN 13 |
|
Barcode Number: |
5000167077025 |
|
Magnification: |
80% |
|
Barcode Truncated By: |
4.3 mm |
|
(smallest bar) | |
|
Edgemark Position: |
N/A |
|
Pharmacode No/NE: |
N/A |
|
Technical Et Non Printing Items | |
|
Cutter | Guides | |
|
Colours | |
|
Main Headings: |
apt |
|
Sub Headings: |
7pt |
|
Body Copy: |
6.25pt |
|
PACK MOCK OP | |||
|
Product Name: |
Mouth Ulcer Gel | ||
|
Product Licence No.: |
00014/0150 | ||
|
Wording Ref: |
MHRA approved VI dated 04/02/2014 (BTC 12G506 action C] | ||
|
Status: |
Internally approved | ||
|
Pack Details: |
Internally lacquered aluminium tube (polyamide/polyimide lacquer system) with a membrane seal fitted with a wadless | ||
|
polyethylene cap or a polypropylene cap. | |||
|
Pack size: |
15 g | ||
|
Version No. |
Date Issued |
Reason For Change | |
|
1 |
03/12/14 |
Correct BCM postcode to NG2 3AA | |
M

MOUTH ULCER fie/
Cetylpyridinium Chloride, Lidocaine
V Rapidly relieves mouth ulcer pain y Soothing antiseptic y Adults and children
60 mm

Read all of the carton for full instructions.
/How to use this medicine
Pierce tube seal with end of cap. Adults and children: Apply a small amount of gel to the affected area with a clean finger, every 3 hours during the day, if you need to. Mouth Ulcer Gel is not suitable for teething trouble in babies.
How to store this medicine
Do not store above 25°C.
Keep all medicines out of the sight and reach of children.
Active ingredients: This oral gel contains Cetylpyridinium Chloride 0.02% w/w, Lidocaine 0.6% w/w.
Also contains: purified water, ethanol (33.9 vol %), sucrose, hypromellose, o cetomacrogol 1000, star anise oil, J eucalyptol, levomenthol. ^
pl 00014/oi so 15gfc?
The Boots Company PLC PWVQ1 Nottingham NG2 3AA 22-73-950
ARTWORK ONLY
Main Headings: 7pt
Sub Headings: 7pt
Body Copy: 6pt
|
PACK MOCK OP | ||
|
Product Name: Mouth Ulcer Gel Product Licence No.: 00014/0150 Wording Ref: BTC126493 vC 27/06/13 Status: MHRA approved Pack Size: 15 g Pack Details: Internally lacquered aluminium tube (polyamide/polyimide lacquer system) | ||
|
Version No. |
Date Issued |
Reason For Change |
|
1 |
04/02/2014 |
Re-design, BCM address and Yellow card update |
|
Trident Reference No: BTCl 26493 | |
|
Zen Ref: |
TR694315 |
|
Category: |
Healthcare |
|
Sub-Category: |
Mouth Care |
|
Brand: |
Core |
|
Pack Type: |
Tube |
|
Variant: |
Boots Mouth Ulcer Gel 15g - Tube |
|
Action: |
D |
|
Date: |
10/02/14 |
|
Country: |
UK |
|
Component Code: |
PWVQ1 |
|
Item Code: |
22-73-950 |
|
CAD Ref No: |
19mm diameter x 104mm |
|
Printer: |
N/A |
|
Substrate: |
White Aluminum |
|
Barcode Type: |
N/A |
|
Barcode Number: |
N/A |
|
Magnification: |
N/A |
|
Barcode Truncated By: (smallest bar) |
N/A |
|
Edgemark Position: |
N/A |
|
Pharmacode No/NE: |
1011(26) |
|
Technical Et Non Printing Items Cutter | Guides | |
|
Colours | |
I
|
Pattern Gloss Varnish | |
|
_5_ | |
TECHNICAL INFO: PRINTER PLEASE NOTE
Trident have created this artwork to a generic print process specification and repro has not been applied. Please call your Boots contact to discuss any amends or if anything falls beneath your print tolerances.