Braltus 10 Microgram Per Delivered Dose Inhalation Powder Hard Capsule

I
tu3ZZD
Package leaflet:
Information for the patient
£Braltus
10 microgram per delivered dose inhalation powder, hard capsule
tiotropium
Read all of this leaflet carefully before
you start taking this medicine because it
contains important information for you.
- Keep this leaflet. You may need to read it again.
- If you have any further questions, ask your doctor, pharmacist or nurse.
- This medicine has been prescribed for you only. Do not pass it on to others. It may harm them, even if their signs of illness are the same as yours.
- If you get any side effects, talk to your doctor, pharmacist or nurse. This includes any possible side effects not listed in this leaflet. See section 4.
What is in this leaflet
1. What Braltus is and what it is used for
2. What you need to know before you take Braltus
3. How to take Braltus
4. Possible side effects
5. How to store Braltus
6. Contents of the pack and other information
1. What Braltus is and what it is used for
Braltus contains the active substance tiotropium. Tiotropium helps people who have chronic obstructive pulmonary disease (COPD) to breathe more easily. COPD is a chronic lung disease that causes shortness of breath and coughing. The term COPD is associated with the conditions chronic bronchitis and emphysema. As COPD is a chronic disease you should take this medicine every day and not only when you have breathing problems or other symptoms of COPD.
Braltus is a long-acting bronchodilator that helps to open your airways and makes it easier to get air in and out of the lungs. Regular use of this medicine can also help you when you have on-going shortness of breath related to your disease and will help you to minimise the effects of the disease on your everyday life. It also helps you to be active longer. Daily use of this medicine will also help to prevent sudden, short-term worsening of your COPD symptoms which may last for several days. The effect of this medicine lasts for 24 hours, so you only need to take it once a day.
This medicine should not be used as a rescue therapy for the treatment of unexpected tightness of the chest, coughing or sudden attacks of breathlessness or wheezing. Please use a fast-acting 'reliever' (rescue) inhaler, such as salbutamol instead. Please keep this 'reliever' inhaler with you at all times.
2. What you need to know before you take Braltus
Do not take Braltus:
- if you are allergic (hypersensitive) to tiotropium or any of the other ingredients of this medicine (listed in section 6)
- if you are allergic (hypersensitive) to atropine or substances related to it, e.g. ipratropium or oxitropium
- if you are allergic (hypersensitive) to lactose or any other sugars
Warnings and precautions
Talk to your doctor, pharmacist or nurse before taking Braltus:
- if you are taking any other medicinal products containing ipratropium or oxitropium
- if you are suffering from narrow angle glaucoma, prostate problems or have difficulty passing urine
- if you have any kidney problems
- if you have suffered from a heart attack (myocardial infarction) during the last 6 months, or from any unstable or life-threatening irregular heart beat or severe heart failure in the past year.
Braltus is indicated for maintenance treatment of your chronic obstructive pulmonary disease; it should not be used to treat a sudden attack of breathlessness or wheezing. Immediate allergic reactions such as rash, swelling, itching, wheezing or breathlessness may occur after administration of Braltus. If this occurs, please consult your doctor immediately (see section 4).
Inhaled medicines such as Braltus may cause tightness of the chest, coughing, wheezing or breathlessness (bronchospasm) immediately after inhalation. In such instances you should immediately use a fast-acting 'reliever' inhaler, such as salbutamol. If these symptoms occur stop taking Braltus inhaler and please consult your doctor immediately.
Take care not to let the inhalation powder enter your eyes as this may result in watering eyes or it may cause narrow-angle glaucoma or may worsen this condition. Narrow-angle glaucoma is a disease of the eyes. Eye pain or discomfort, blurred vision, seeing halos around lights or coloured images in association with red eyes may be signs of an acute attack of narrow-angle glaucoma. Eye symptoms may be accompanied by headache, nausea or vomiting. You should stop using this medicine and immediately consult your doctor, preferably an eye specialist, when signs and symptoms of narrow-angle glaucoma appear.
The medicine in your inhaler may decrease the normal secretions (saliva) in your mouth and make your mouth feel dry. Long-term this may be associated with dental caries (tooth decay). Therefore, please remember to pay attention to oral hygiene, rinse your mouth out and brush your teeth regularly.
If you have suffered from a heart attack (myocardial infarction) during the last 6 months, or from any unstable or life-threatening irregular heart beat or severe heart failure in the past year, please inform your doctor. This is important so that the doctor can decide whether or not Braltus is the right treatment for you to take.
Do not take this medicine more than once daily (see section 3).
Children and adolescents
Braltus should not be used in children and adolescents under the age of 18 years.
Other medicines and Braltus
Tell your doctor or pharmacist if you are taking, have recently taken or might take any other medicines, including any other inhalers and medicines bought over the counter from the pharmacy.
Please tell your doctor or pharmacist if you are taking/have taken similar medicines for your lung disease, such as ipratropium or oxitropium.
No specific side effects have been reported when this medicine has been taken together with other products used to treat COPD such as reliever inhalers, e.g. salbutamol, methylxanthines, e.g. theophylline and/or oral and inhaled steroids e.g. prednisolone. Pregnancy and breast-feeding If you are pregnant or breast-feeding, think you may be pregnant or are planning to have a baby, ask your doctor or pharmacist for advice before taking this medicine. You should not take this medicine unless specifically recommended by your doctor.
Driving and using machines The occurrence of dizziness, blurred vision, or headache may influence the ability to drive and use machinery.
Braltus contains lactose Lactose is a type of sugar found in milk. If you have been told by your doctor that you have an allergy to some sugars, contact your doctor before using this inhaler. Lactose may contain very small amounts of milk protein, which may cause reactions in those who have severe hypersensitivity or allergy to milk proteins. When taken according to the dose recommendation of one capsule once a day, each dose supplies up to 18 mg of lactose monohydrate.
3. How to take Braltus
Always take this medicine exactly as your doctor has told you. Check with your doctor or pharmacist if you are not sure.
The recommended dose is to inhale the contents of one capsule once daily with the Zonda inhaler. One capsule provides the required daily dose of tiotropium (a delivered dose of 10 micrograms of tiotropium); do not take more than the recommended dose.
You should try to take the capsule at the same time every day. This is important because this medicine is effective over 24 hours.
The capsules are only for inhalation and not for oral intake.
Do not swallow the capsules.
The Zonda inhaler, which you should put the Braltus capsule into, makes holes in the capsule and allows you to breathe in the powder. The capsules must only be inhaled using the Zonda inhaler. Do not use any other inhalers to take Braltus capsules.
Make sure you know how to use the Zonda inhaler properly. The instructions for use of the Zonda inhaler are provided on the other side of this leaflet. If you have any problems using the Zonda inhaler, ask your doctor, pharmacist or nurse to show you how it works.
If necessary, you may wipe the mouthpiece of your Zonda inhaler after use with a dry cloth or tissue.
Make sure you do not blow into the Zonda inhaler. When taking Braltus, take care not to let any of the powder enter your eyes. If any powder does get into your eyes, you may get blurred vision, eye pain and/or red eyes; you should wash your eyes in warm water immediately. Then talk to your doctor immediately for further advice.
If you feel that your breathing is worsening, you should tell your doctor as soon as possible. Use in children and adolescents Braltus should not be used in children and adolescents under the age of 18 years.
If you take more Braltus than you should
If you inhale the contents of more than 1 capsule of Braltus in a day, you should talk to your doctor immediately. You may be at a higher risk of experiencing side effects such as dry mouth, constipation, difficulties passing urine, increased heart beat, or blurred vision.
If you forget to take Braltus
If you forget to take a dose, take one as soon as you remember, but do not take two doses at the same time or on the same day. Then take your next dose as usual.
Do not take a double dose to make up for a forgotten dose.
If you stop taking Braltus
Before you stop taking Braltus, you should talk to your doctor or pharmacist. If you stop taking this medicine, the signs and symptoms of COPD may get worse.
If you have any further questions on the use of this medicine, ask your doctor, pharmacist or nurse.
4. Possible side effects
Like all medicines, this medicine can cause side effects, although not everybody gets them. Serious side effects including allergic reactions which cause swelling of the face or throat (angioedema) or other hypersensitivity reactions (such as sudden reduction of your blood pressure or dizziness) or increased wheezing and shortness of breath may occur individually or as part of severe allergic reaction (anaphylactic reaction) after taking this medicine. Such serious side effects are rare. In addition, in common with all inhaled medicines, some patients may experience unexpected tightness of the chest, coughing, wheezing or breathlessness immediately after inhalation (known as bronchospasm).
If any of these side effects occur, please consult your doctor immediately.
Do not use your Braltus, inhaler again until you have seen or at least spoken with, your doctor. If you are wheezy and short of breath you should use your fast-acting 'reliever' (rescue) inhaler straightaway.
Other side effects have been experienced by people taking this medicine, and they are listed below according to their frequency. Common: may affect up to 1 in 10 people -Dry mouth: this is usually mild Uncommon: may affect up to 1 in 100 people
- Dizziness
- Headache
- Taste disorders
- Blurred vision
- Irregular heart beat (atrial fibrillation)
|
TEVA UK Ref: 231-30-21403-B LEA TIOTROPIUM (BRALTUS ZONDA) 10mcg INHAL CAPS TUK <CHEMO |
Version: 5 27 May 2016 | ||||
|
IT7Tr77l |
UK |
Template | |||
- Sore throat (pharyngitis)
- Hoarseness (dysphonia)
-Cough
- Heart burn (gastrooesophageal reflux disease)
- Constipation
- Fungal infections of the mouth and throat (oropharyngeal candidiasis)
- Rash
- Difficulty passing urine (urinary retention)
- Painful urination (dysuria)
Rare: may affect up to 1 in 1,000 people
- Difficulty in sleeping (insomnia)
- Seeing halos around lights or coloured images in association with red eyes (glaucoma)
- Increase of the measured eye pressure
- Irregular heart beat (supraventricular tachycardia)
- Faster heart beat (tachycardia)
- Feeling your heart beat (palpitations)
- Tightness of the chest, associated with coughing, wheezing or breathlessness immediately after inhalation (bronchospasm)
- Nosebleed (epistaxis)
- Inflammation of the larynx (laryngitis)
- Inflammation of the sinuses (sinusitis)
- Blockage of intestines or absence of bowel movements (intestinal obstruction including ileus paralytic)
- Inflammation of the gums (gingivitis)
- Inflammation of the tongue (glossitis)
- Difficulties swallowing (dysphagia)
- Inflammation of the mouth (stomatitis)
- Feeling sick (nausea)
- Nettle rash (urticaria)
- Itching (pruritus)
- Infections of the urinary tract Frequency cannot be estimated from the available data:
- Depletion of body water (dehydration)
- Tooth decay (dental caries)
- Infections or ulcerations of the skin
- Dryness of the skin
- Swelling of joints Reporting of side effects
If you get any side effects, talk to your doctor, pharmacist or nurse. This includes any possible side effects not listed in this leaflet. You can also report side effects directly via the Yellow Card Scheme at: www.mhra.gov.uk/yellowcard By reporting side effects you can help provide more information on the safety of this medicine.
5. How to store Braltus
Keep this medicine out of the sight and reach of children.
Do not use this medicine after the expiry date which is stated on the carton and the bottle label after EXP. The expiry date refers to the last day of that month.
Do not refrigerate or freeze.
Keep the bottle tightly closed. Store in the original packaging to protect from moisture. Use the product within 30 days (15 capsule bottle) or 60 days (30 capsule bottle) of opening the bottle.
The Zonda inhaler should only be used with the bottle of capsules provided. Do not reuse the inhaler for another bottle of capsules.
Do not throw away any medicines via wastewater or household waste. Ask your pharmacist how to throw away medicines you no longer use. These measures will help protect the environment.
6. Contents of the pack and other information
What Braltus contains
- The active substance is tiotropium. Each capsule contains 13 micrograms of the active ingredient tiotropium (as bromide). During inhalation, 10 micrograms of tiotropium are delivered to the patient from each capsule from the mouthpiece of the Zonda inhaler and are inhaled into the lungs.
- The other ingredients are lactose monohydrate (capsule content) and Hypromellose (capsule shell).
What Braltus looks like and contents of the pack
Braltus 10 microgram per delivered dose
inhalation powder, hard capsule is a colourless and transparent hard capsule, containing white powder.
This medicine is provided in bottles, with a screw-cap closure. The bottle is supplied in a carton with a Zonda inhaler. The Zonda Inhaler has a green body and cap with a white push button.
Braltus is available in packs containing either 15 or 30 capsules and a Zonda inhaler, and in multipacks containing either 60 capsules (2 packs of 30) and 2 Zonda inhalers or 90 capsules (3 packs of 30) and 3 Zonda inhalers. Not all pack sizes may be marketed. Marketing Authorisation Holder:
TEVA UK Limited, Eastbourne, BN22 9AG, UK Manufacturer:
Laboratorios LICONSA S.A.
Avda. Miralcampo, No 7, Poligono Industrial Miralcampo
19200 Azuqueca de Henares, Guadalajara Spain
This leaflet was last revised in 05/2016
PL 00289/1870
21403-B
Zonda Instructions for Use
Dear Patient,
The Zonda inhaler enables you to inhale the medicine contained in the Braltus capsule that your doctor has prescribed for your breathing problems.
Remember to carefully follow your doctor's instructions for using Braltus. The Zonda inhaler is especially designed for Braltus capsules: you must not use it to take any other medication. The capsules must only be inhaled using the Zonda inhaler. Do not use any other inhalers to take Braltus capsules. Each capsule contains only a small amount of powder. Do not open the capsule or it may not work.
The Zonda inhaler should only be used with the bottle of capsules provided. Do not reuse the inhaler for another bottle of capsules. Discard the Zonda device after 15 uses (if used with a 15 capsule bottle) or 30 uses (if used with a 30 capsule bottle).
1. Dust cap
2. Mouthpiece
3. Base
4. Piercing button
5. Centre chamber
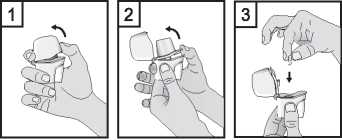
1. Pull the cap upwards.
2. Hold the base of the inhaler firmly and open the mouthpiece by pulling it upwards, in the direction of the arrow.
3. Remove a Braltus capsule from the bottle immediately before use and close the bottle tightly. Place one capsule in the capsule-shaped compartment in the base of the inhaler. Do not store the capsule in the Zonda inhaler.
4. Never place a capsule directly into the mouthpiece.
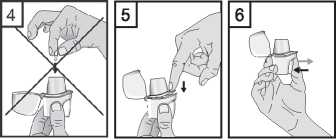
5. Close the mouthpiece until you hear a click, leaving the cap open.
6. Hold the inhaler with the mouthpiece upwards, and firmly press the piercing button completely in only once. Release the button. This will pierce the capsule and allows the medication to be released when you breathe in.
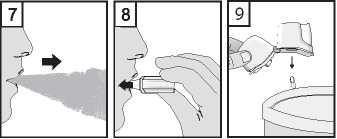
7. Breathe out fully. It is important to do this away from the mouthpiece. Avoid breathing into the mouthpiece at any time.
8. Place the mouthpiece in your mouth and keep your head in an upright position. Close your lips around the mouthpiece and breathe in slowly and deeply enough to hear or feel the capsule vibrating inside the compartment. Hold your breathe for as long as you comfortably can whilst taking the inhaler out of your mouth. Then breathe normally. Repeat steps 7 and 8 to empty the capsule completely.
9. After use, open the mouthpiece again, and tip out the empty capsule. Close the mouthpiece and cap, and store your Zonda inhaler.
The Zonda inhaler is a Medical Device (CE)
Manufacturer:
Plastiape S.p.A.
Via 1 Maggio, 8, 23875 Osnago LC, Italy
21403-B