Budelin Novolizer 200 Micrograms Per Actuation Inhalation Powder

1. PREPARATION:
The NOVOLIZER® Dry Powder Inhaler makes inhaling simple and reliable. Its straightforward use, fast cartridge replacement and simple cleaning are easy and quickly done.
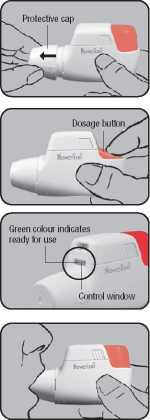
Place the NOVOLIZER® Dry Powder Inhaler in front of you. Lightly press together the ribbed surfaces on both sides of the cap, move the cap forwards (^) and lift off (41).
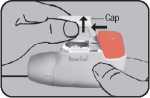
#
#
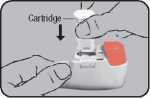
Remove the protective aluminium foil from the cartridge cylinder and take out the new cartridge. However, this should only be done just before using the cartridge. The colour coding on the cartridge must correspond to the colour of the dosage button.
First filling:
Insert the cartridge into the NOVOLIZER® Dry Powder Inhaler with the dosage counter facing the mouthpiece (^). Do not press the dosage button while inserting the cartridge.
Refilling:
Note: The NOVOLIZER® Dry Powder Inhaler should be cleaned every time the cartridge is exchanged after removal of empty cartridge.
If you have already used the nOvOLIZER® Dry Powder Inhaler, first remove the empty cartridge and then insert the new one (^). Do not press the dosage button while inserting the cartridge.

Check that the colour of the control window has changed back to red, also indicating a correct inhalation. Replace the protective cap on the mouthpiece - the inhalation procedure is now complete.
The number in the top window indicates the number of inhalations left. The numeric scale 200-60 is shown in steps of 20 and 60-0 in steps of 10.If the click sound and change of the colour did not appear, please repeat the procedure as described above.
NOTE: The coloured dosage button should only be pressed immediately before inhalation.
Inadvertent overdosing is not possible with the NOVOLIZER®. The click sound and the change of the colour in the control window indicate that inhalation has been performed correctly. If the colour of the control window does not change back to red, the inhalation should be repeated. If inhalation is not completed correctly after several attempts, then you should consult your doctor.
3. CLEANING:
The NOVOLIZER® Dry Powder Inhaler should be cleaned at regular intervals, but at least every time the cartridge is exchanged.
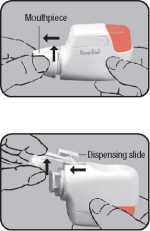
Remove protective cap and mouthpiece First remove the protective cap. Then grasp the mouthpiece and turn it briefly counter-clockwise (4") until it becomes loose. Then remove (^).
Cleaning
Now turn the NOVO-LIZER® upside down.
Grasp the loose dispensing slide and move it forwards (^) and upwards (41). Any remaining powder can be removed by tapping lightly.
Clean the mouthpiece, the dispensing slide and the powder inhaler with a soft and dry lint-free cloth.
Do NOT use water or detergent.
Assembly - Insert dosage slide After cleaning insert the dosing slide by sliding down at an angle (^l) and press into position
W.
Turn the inhaler back over.

o
CO
r-
o
o
lO
200 micrograms per actuation inhalation powder
Active Substance: Budesonide
Read all of this leaflet carefully before you start using this medicine because it contains important information for you.
- Keep this leaflet. You may need to read it again.
- If you have any further questions, ask your doctor or pharmacist.
- This medicine has been prescribed for you only. Do not pass it on to others. It may harm them, even if their symptoms are the same as yours.
- If you get any side effects, talk to your doctor or pharmacist. This includes any possible side effects not listed in this leaflet. See section 4.
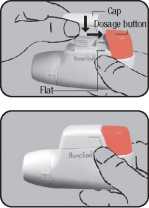
Replace the cap into the side guides from above (^) and push down flat towards the coloured dosage button until it snaps into place (^).
The NOVOLIZER® is now filled and ready for use.
You can leave the cartridge in the NOVO-LIZER® Dry Powder Inhaler until it has been used up or for up to 6 months after insertion. The cartridge is used up if you see a hatched "0" in the middle of the dosage counter. Then a new cartridge has to be inserted. The cartridges may only be used in the original dry powder inhaler.
2. USAGE:
Whenever possible, sit or stand while inhaling.
When using the NOVO-LIZER® always keep it horizontal. First remove the protective cap (^).
Completely depress the coloured dosage button.
A loud double click will be heard and the colour of the control window will change from red to green. Then release the coloured button. The colour green in the control window indicates that the NOVOLIZER® device is ready for use.
Exhale (but not into the NOVOLIZER® Dry Powder Inhaler). Put your lips tightly around the mouthpiece, inhale the powder with a deep breath and then hold the breath for a few seconds. During this breath a loud click should be heard, indicating correct. Then continue with normal breathing.
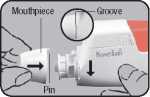
Assembly - Fit mouthpiece and protective cap Insert the mouthpiece with the pin into the groove on the left and turn to the right until it snaps into place. Finish by replacing the protective cap.
Notes
• The Patient Information Leaflet describes how the drug works. Please read it through carefully before using the inhaler for the first time.
• The NOVOLIZER® which comes with various active substances does not use any propellants and is designed for refilling. This makes the NOVOLIZER® a very environmentally friendly product.
• It is not possible to overdose with the NOVOLIZER®. Even if the button is pressed several times, no more powder is available for inhalation. Only press the button when you really want to inhale. If you cannot manage to inhale correctly after several attempts, consult your doctor.
• The NOVOLIZER® can be refilled using new cartridges* containing the active substance and is thus ideally suited to long-term usage (up to one year).
• Do not shake the filled NOVOLIZER®.
• Please support your children in proper handling of the device.
• Make sure your NOVOLIZER® is protected from moisture and heat and kept clean at all times.
• Regarding corresponding medicines, please ask your doctor.
MEDA Pharma GmbH & Co. KG Benzstr. 1
61352 Bad Homburg (Germany)
(€
Last Review April 2014
1 Instructions for Use (Gebrauchsanleitung) in compliance with the Medizinproduktegesetz (German Law on Medical Devices)
o
CO
Is
o
o
LO
What is in this leaflet:
1. What Budelin Novolizer 200 micrograms is and what it is used for
2. What you need to know before you use Budelin Novolizer 200 micrograms
3. How to use Budelin Novolizer 200 micrograms
4. Possible side effects
5. How to store Budelin Novolizer 200 micrograms
6. Contents of the pack and other information
1. WHAT BUDELIN NOVOLIZER 200 MICROGRAMS IS AND WHAT IT IS USED FOR
Budesonide, the active substance in Budelin Novolizer 200 micrograms, is a glucocorticoid (corticosteroid) for inhalation.
Budelin Novolizer 200 micrograms is used for regular treatment of persistent asthma.
NOTE:
Budelin Novolizer 200 micrograms should not be used for treatment of a sudden attack of respiratory distress (acute asthma attack or Status asthmaticus (asthma attacks occurring very frequently and/or persisting for several days)).
2. WHAT YOU NEED TO KNOW BEFORE YOU USE BUDELIN NOVOLIZER 200 MICROGRAMS
Do not use Budelin Novolizer 200 micrograms if you are allergic (hypersensitive ) to the active substance budesonide or to the excipient lactose monohydrate (which contains small amounts of milk proteins).
Warnings and precautions
Talk to your doctor or pharmacist before using Budelin Novolizer 200 micrograms.
Take special care in using Budelin Novolizer
200 micrograms, if you are suffering from lung tuberculosis or fungal infection or from other infection of the airways. This also applies if you have been affected by these conditions in the past. Ask your doctor for advice.
Budesonide is not suitable for treatment of acute respiratory distress or severe continuous spasm of the bronchial tubes (Status as-thmaticus). Your doctor will advise you to use a short-acting inhaled bronchodilating agent (bronchodilator) as rescue medication to relieve the acute symptoms of your complaints.
If you have a severe liver disease, the elimination of Budesonide may be impaired. This may lead to increased levels of Budesonide in your blood.
Any inhaled glucocorticoids may cause side effects, particularly when using high doses for prolonged periods. These effects are much less likely to occur with inhalation treatment than with the intake of glucocorticoid tablets. Possible effects include disorders in the function of the adrenal cortex, Cushing’s syndrome, Cushingoid features (hormone disorder caused by high levels of cortisol in the blood with central obesitiy, ‘moon face’, thinning of the skin, hypertension, etc.), decrease in bone density, growth retardation in children and adolescents as well as eye disease (cataract and glaucoma), and more rarely, a range of psychological or behavioural effects including psychomotor hyperactivity, sleep disorders, anxiety, depression or aggression (particularly in chil-
dren). Therefore, it is important that the lowest dose is administered at which effective control of asthma is maintained.
Should periods of stress or emergencies (e.g. severe infections, injuries and surgery) occur within the first few months of switching from intake of tablets to inhalation treatment, it may be necessary to resume systemic administration of glucocorticoids in form of tablets or infusions. This applies also to patients who have received prolonged treatment with high doses of inhaled glucocorticoids. They may also have impaired adrenocortical function and may need systemic glucocorticoid cover during periods of stress and/or for elective surgery.
After switching to inhalation treatment, symptoms may occur that had been suppressed by the previous systemic treatment with glucocorticoids, e.g. symptoms of allergic rhinitis, allergic rash or rheumatic complaints. These symptoms should be treated in addition by suitable medication.
Some patients might feel generally unwell in a non-specific way during the switching period despite maintenance or even improvement in respiratory function. In such a case, please, consult your doctor. He/she will then decide whether treatment can be continued as planned or if you have - for instance - symptoms of an insufficient function of the adrenal cortex conflicting with such continuation.
Other medicines and Budelin Novolizer 200 micrograms
Tell your doctor or pharmacist if you are taking / using or have recently taken / used any other medicines, including medicines obtained without prescription.
Concomitant use of Budelin Novolizer 200 micrograms and ketoconazole or itraconazole (medicine for treatment of fungal disease) will significantly increase the blood levels and thus the possible side effects of Budesonide. Therefore the time interval between administration of ketoconazole or itraconazole and Budesonide should be as long as possible. Concomitant administration of Budelin Novolizer 200 micrograms the HIV protease inhibitors nelfinavir and ritonavir (medicines used to treat patients with HIV infection) may also lead to increased blood levels of Budesonide.
Raised plasma concentrations of corticosteroids and enhanced effects of corticosteroids have been observed in women also treated with oestrogens and contraceptive steroids, but no effect has been observed with budesonide and concomitant intake of low dose combination oral contraceptives. Because adrenal function may be suppressed, an ACTH stimulation test for diagnosing pituitary insufficiency might show false results (low values).
Pregnancy and breast-feeding
If you are pregnant or breast-feeding, think you may be pregnant or are planning to have a baby, ask your doctor of pharmacist for advice before taking this medicine.
Pregnancy
Results from a large prospective epidemiological study and from world-wide post marketing experience indicate that inhaled budesonide during pregnancy has no adverse effects on the health of the foetus / new born child.
As with other drugs the administration of budesonide during pregnancy requires that the benefits for the mother are weighed against the risks for the foetus.
Breast-feeding
Budesonide is excreted in breast milk. However, at therapeutic doses no effects on the suckling child are anticipated. Maintenance treatment with inhaled budesonide (200 or 400 micrograms twice daily) in asthmatic nursing women results in negligible systemic exposure to budesonide in breast-fed infants. Therefore, Budelin Novolizer 200 micrograms can be used during breast feeding.
Driving and using machines
Budesonide has no influence on the ability to drive and use machines.
0/
D
fT3
C
o>
‘l/l
_c
u
l/l
a}
c
aj
fT3
~o
£
qj
up
<D
qj
_Q
fO
Q)
‘<D
, c
3 •.£ tj a/ qj
re
£
qj
U)
aJ
£
qj
CO
N
E
r—
O
<N
00
O
c
<u
3
<U
Z
Q- ro
iv .y
co %
^ +■» — IV Q.
N VO I
QJ
LZ
-§ <L>
<y "2
S O
£
o o o o o
2 2 2 2 2
Cl Cl Cl Cl Cl
r— cn ro 'vf lo

a
u
J2
-Q
cc
Q.
z
o
ft!
cn
3
O

2
zs
+->
u
E
45
D
C
ft!
U
IV
=8= CM A3
QJ O CD Q_
Important information about some of the ingredients of Budelin Novolizer 200 micrograms:
Budelin Novolizer 200 micrograms contains milk sugar (lactose), 10.7 mg of lactose monohydrate/delivered dose.
Normally, the content of lactose in a single dose does not cause any problems in people with lactose intolerance. If you are concerned that you may have an intolerance, you must talk to your doctor’.
Milk sugar (lactose) contains small amounts of milk protein.
3. HOW TO USE BUDELIN NOVOLIZER 200 MICROGRAMS
Always use Budelin Novolizer 200 micrograms exactly as your doctor has told you. You should check with your doctor or pharmacist if you are not sure.
Patients without previous treatment with glucocorticoids and patients previously treated with inhaled glucocorticoids Unless otherwise prescribed by your doctor, the following dosage is recommended for:
Adults (including older people) and children/adolescents over 12 years of age:
Initial recommended
dose: 1 -2 single doses
(200 - 400 micrograms) once or twice daily
Maximum recommended dose: 4 single doses
(800 micrograms) twice daily (daily dose: 1600 micrograms)
Children aged 6 to 12 years:
Initial recommended
dose: 1 single dose
(200 micrograms) twice or
200 - 400 micrograms once a day
Maximum recommended dose: 2 single doses
(400 micrograms) twice daily (daily dose:
800 micrograms)
In case of once daily dosing, it is recommended to take this dose in the evening.
Children below 6 years of age:
Budelin Novolizer 200 micrograms is not recommended for use in children below age 6 due to insufficient data on safety and efficacy.
Please support your children in proper handling of the Novolizer device.
Children
It is recommended that the growth of children receiving prolonged treatment with inhaled glucocorticoids at large doses is regularly monitored.
Older people
Usually, no special dose adjustment is required. In general, the lowest effective dose required for a sufficient control should be used.
In case of deterioration of symptoms (recognized by e.g. persistent respiratory distress and increased use of other inhaled broncho-dilating agents) you should seek advice from a medical doctor as soon as possible. If you have inhaled only once a day so far, it may become required in such a case to use the same dose twice daily now (in the morning and in the evening). In any case, you doctor should decide if your usual dose of Budelin Novolizer 200 micrograms needs to be increased.
Should you have the impression that the effect of Budelin Novolizer 200 micrograms is either too strong or too weak, discuss this with your doctor or pharmacist.
For the relief of acute symptoms of asthma, you should carry along a short acting inhaled bronchodilating agent (beta-2-agonist, such as Salbutamol) at all times.
When you switch from another Budesonide inhaler to Budelin Novolizer 200 micrograms, the treatment plan may have to be adjusted by your doctor.
Method of Administration
Inhalation use.
Inhale as shown in the Instructions for Use.
Important information for use
To reduce the risk of fungal infection in mouth and throat (oral candidiasis) and hoarseness it is recommended that inhalation be performed before meals and/or that the mouth is rinsed with water or the teeth are brushed after each inhalation.
Duration of treatment
Budelin Novolizer 200 micrograms is intended for long-term therapy. It should be used regularly according to the recommended treatment schedule even at times when no symptoms are experienced.
If vou have not used glucocorticoids previously or have been treated occasionally only over a short period with glucocorticoids, regular use of Budelin Novolizer 200 micrograms as directed should lead to an improvement in breathing after approximately 10 days. However, extreme mucous congestion and inflammatory processes may obstruct the bronchial passages to such an extent that Budesonide cannot fully exert its effects in the lung. In such cases, initiation of therapy should be supplemented with administration of cortisone products (systemic glucocorticoids) in form of tablets. Later on, the tablet dose should be reduced gradually but inhalative therapy will be continued.
If you have used cortisone products for a prolonged period already, you should be switched to Budelin Novolizer 200 micrograms at a time when your symptoms are completely under control. Normally, function of the adrenal cortex is suppressed in this situation and therefore, intake of cortisone tablets (systemic corticoid administration) should be reduced gradually and must not be stopped abruptly. At the beginning of the switchover period, Budelin Novolizer 200 micrograms should be given in addition for approximately 10 days. Then, depending on your response, the daily dose of the cortisone tablets can be reduced gradually at intervals of one to two weeks.
If you inhale more Budelin Novolizer 200 micrograms than you should
It is important that you take your dose as stated on the pharmacist’s label or as advised by your doctor. You should not increase or decrease your dose without seeking medical advice.
If you forget to use Budelin Novolizer 200 micrograms
Do not take a double dose to make up for a forgotten dose.
If you stop using Budelin Novolizer 200 micrograms
Do not stop treatment with the Budelin Novolizer 200 micrograms without talking to your doctor first because this could lead to an aggravation of the disease.
If you have any further questions on the use of this product, ask your doctor or pharmacist.
4. POSSIBLE SIDE EFFECTS
Like all medicines, Budelin Novolizer 200 micrograms can cause side effects, although not everybody gets them.
Most important side effects
Irritation of the oral mucosa (throat irritation) accompanied by difficulty in swallowing, hoarseness and cough may commonly occur.
Treatment with inhaled Budesonide may result in fungal disease in the mouth and the throat (oropharyngeale candidiasis). Experience has shown that this fungal infection occurs less often when inhalation is performed before meals and/or when the mouth is rinsed or the teeth are brushed after inhalation. In most cases this condition responds to topical anti-fungal therapy without discontinuing treatment with the Budelin Novolizer 200 micrograms.
As with other inhalation therapies, in rare cases bronchial spasm (paradoxical bron-chospasm) may occur, manifested by a temporary exacerbation of the respiratory distress and a immediate increase in wheezing after dosing. Only in such a case, you should discontinue use of the Budelin No-volizer 200 micrograms without previous consultation and you must contact your doctor immediately.
Inhalation of larger doses over a prolonged period may lead to increased susceptibility to infection. The ability to adapt to stress can be impaired.
List of all other side effects
Rare (may affect up to 1 in 1,000 treated patients):
Allergic reactions (hypersensitivity) and swelling of the face, eyes, lips, mouth and throat (angioneurotic oedema), anaphylactic reaction; suppression of the function of the adrenal cortex (adrenal suppression), growth retardation in children and adolescents; restlessnes, nervousness, depression, abnormal behaviour, over-excited or irritable (these effects are more likely to occur in children); skin reactions such as nettle rash (urticaria), eczema, topical inflammation of the skin (dermatitis), itching (pruritus), redness of the skin by excessively filled blood vessels (erythema), bruising, disturbance of voice and hoarse voice (in children).
Very rare (may affect up to 1 in 10,000 treated patients):
Bone density is decreased
Frequency not known (frequency cannot be estimated from the available data):
Sleeping problems, anxiety or feeling worried, aggression, excessive urge to be active accompanied by mental restlessness (psychomotor hyperactivity); cataract, glaucoma; Lactose-monohydrate contains small amounts of milk proteins and can therefore cause allergic reactions.
Reporting of side effects
If you get any side effects, talk to your doctor or pharmacist. This includes any possible side effects not listed in this leaflet. You can also report side effects directly via the Yellow Card Scheme at: www.mhra.gov.uk/ yellowcard. By reporting side effects, you can help provide more information on the safety of this medicine.
5. HOW TO STORE BUDELIN NOVOLIZER 200 MICROGRAMS
Keep out of the reach and sight of children.
Do not use this medicine after the expiry date which is stated on the label, the carton and the cartridge container. The expiry date refers to the last day of that month.
Storage conditions
Store in the original package. This medicinal product does not require any special temperature storage conditions.
In-Use storage conditions: Keep the Novolizer device tightly closed, in order to protect from moisture.
Information concerning in-use shelf-life
Exchange the cartridge six months after first opening.
Do not use the powder inhaler for more than a year.
Note: The Novolizer device has been shown to function for at least 2000 single doses. Therefore a maximum of 10 cartridges containing 200 single doses each and/or 20 cartridges containing 100 single doses each can be used with this device (within a single year) prior to replacement.
Medicines should not be disposed of via wastewater or household waste. Ask your pharmacist how to dispose of medicines no longer required. These measures will help to protect the environment.
6. CONTENTS OF THE PACK AND OTHER INFORMATION
What Budelin Novolizer 200 micrograms contains
The active substance is budesonide.
Each actuation (puff) contains 200 micrograms of budesonide.
The other ingredient is Lactose monohydrate.
What Budelin Novolizer 200 micrograms looks like and contents of the pack
Budelin Novolizer 200 micrograms, inhalation powder, contains a white powder (1.09 g or 2.18 g) in a cartridge containing 100 or 200 metered doses packed in a container sealed by aluminium foil plus a Novolizer powder inhaler device.
All components are made of plastic materials.
Pack sizes:
Original sales packs:
1 cartridge containing 100/200 metered doses and 1 Novolizer powder inhaler device
2 cartridges containing 200 metered doses each and 1 Novolizer powder inhaler device
Refill packs:
1 cartridge containing 100 metered doses (UK and Ireland only)
1 cartridge containing 200 metered doses
2 cartridges containing 200 metered doses each
Not all pack sizes may be marketed
Marketing Authorization Holder
Meda Pharmaceuticals Ltd Skyway House Parsonage Road Takeley
Bishops Stortford CM22 6PU UK
Manufacturer:
MEDA Manufacturing GmbH Neurather Ring 1 D-51063 Cologne Tel: (+49)-221-6472-0 Fax: (+49)-221-6472-696) or:
MEDA Pharma GmbH & Co. KG
Benzstrasse 1
D-61352 Bad Homburg
Germany
or:
VEMEDIA Manufacturing B.V.
Verrijn Stuartweg 60 NL-1112 AX Diemen Netherlands
This medicinal product is authorized in the Member States of the EEC under the following names:
[Name of Member States]
[Name of the medicinal product]
Austria
Novolizer® Budesonid Meda 200 Mikro-gramm Pulver zur Inhalation Belgium and Luxembourg:
Novolizer® Budesonide 200 microgrammes,
poudre pour inhalation
Denmark:
Novopulmon® Novolizer® 200 mikrogram/
dosis
Finland:
Novopulmon® Novolizer® 200 mikrog/annos
inhalaatiojauhe
France:
Novopulmon® Novolizer® 200 micro-
grammes/dose, poudre pour inhalation Germany:
Novopulmon® 200 Novolizer®, Pulver zur Inhalation
Ireland:
Novolizer® Budesonide 200 micrograms inhalation powder Italy:
Budesonide Viatris® Novolizer® 200 micro-grammi polvere per inalazione The Netherlands:
Budesonide Novolizer® 200 microgram, in-
halatiepoeder
Norway:
Novopulmon® Novolizer® 200 mikrog/dose
inhalasjonspulver
Portugal:
Budesonido Novolizer® 200 microgramas po
para inalagao
Spain:
Novopulm® Novolizer® 200 microgramos,
polvo para inhalacion
Sweden:
Novopulmon® Novolizer® 200 mikrogram/ dos inhalationspulver United Kingdom:
Budelin® Novolizer® 200 micrograms per actuation inhalation powder
This leaflet was last approved in 04/2014.
T5007310
of
(X3
C
'i/i
u
<D
c
D
<U
rtS
~o
E
QJ
i/i
<V
<v
_Q
fO
'<y
□
I -=5
ib cr
o
E <=
3 O
□
rtS
E
0J
U)
(V
E
<v
CD
N
E

03

— 4->
3
U
IV
r—
LO
=#: <N $2
QJ
lZ
3 ~o
s 6
D
C
03
E
u
CD O Ui Q_
E
E
o
# T5007310_p01.indd 2 27.08.14 10:24