Bunov 10 Microgram/H Transdermal Patch

Package leaflet:
Information for the patient
Bunov 5 microgram/h transdermal patch
Bunov 10 microgram/h transdermal patch
Bunov 20 microgram/h transdermal patch
buprenorphine


Package leaflet: Information for the patient
Bunov 5 microgram/h transdermal patch Bunov 10 microgram/h transdermal patch Bunov 20 microgram/h transdermal patch
buprenorphine
Read all of this leaflet carefully before you start using this medicine because it contains important information for you.
- Keep this leaflet. You may need to read it again.
- If you have any further questions, ask your doctor or pharmacist.
- This medicine has been prescribed for you only. Do not pass it on to others. It may harm them, even if their signs of illness are the same as yours.
- If you get any side effects, talk to your doctor or pharmacist.
This includes any possible side effects not listed in this leaflet.
See section 4.
What is in this leaflet
1. What Bunov is and what it is used for
2. What you need to know before you use Bunov
3. How to use Bunov
4. Possible side effects
5. How to store Bunov
6. Contents of the pack and other information
1. What Bunov is and what it is used for
Bunov contain the active ingredient buprenorphine which belongs to a group of medicines called strong analgesics or ‘painkillers’. They have been prescribed for you by your doctor to relieve moderate, long-lasting pain that requires the use of a strong painkiller.
Bunov should not be used to relieve acute pain.
2. What you need to know before you use Bunov
Do not use Bunov:
- if you are allergic to buprenorphine or any of the other ingredients of this medicine (listed in section 6);
- if you have breathing problems;
- if you are addicted to drugs;
- if you are taking a type of medicine known as a monoamine oxidase inhibitor (examples include tranylcypromide, phenelzine, isocarboxazid, moclobemide and linezolid), or you have taken this type of medicine in the last two weeks;
- if you suffer from myasthenia gravis (a condition in which the muscles become weak);
- if you have previously suffered from withdrawal symptoms such as agitation, anxiety, shaking or sweating upon stopping taking alcohol.
Bunov must not be used to treat symptoms associated with drug withdrawal.
Warnings and precautions
Talk to your doctor or pharmacist before using Bunov:
- if you suffer from seizures, fits or convulsions;
- if you have a severe headache or feel sick due to a head injury or increased pressure in your
- skull (for instance due to brain disease). This is because the patches may make symptoms worse or hide the extent of a head injury;
- if you are feeling light-headed or faint;
- if you have severe liver problems;
- if you have ever been addicted to drugs or alcohol;
- if you have a high temperature, as this may lead to larger quantities of the active ingredient being absorbed into the blood than normal.
If you have recently had an operation, please speak to your doctor before
using these patches.
Athletes should be aware that this medicine may cause a positive reaction to
sports doping control tests.
Children and adolescents
Do not give this medicine to children and adolescents below 18 years.
Other medicines and Bunov
Tell your doctor or pharmacist if you are taking, have recently taken or might take any other medicines.
- Bunov must not be used together with a type of medicine known as a monoamine oxidase inhibitor (examples include tranylcypromide, phenelzine, isocarboxazid, moclobemide and linezolid), or if you have taken this type of medicine in the last two weeks.
- If you take some medicines such as phenobarbital or phenytoin (medicines commonly used to treat seizures, fits or convulsions), carbamazepine (a medicine to treat seizures, fits or convulsions and certain pain conditions), or rifampicin (a medicine to treat tuberculosis) the effects of Bunov may be reduced.
- Bunov may make some people feel drowsy, sick or faint or make them breathe more slowly or weakly. These side effects may be made worse if other medicines that produce the same effects are taken at the same time. These include certain medicines to treat pain, depression, anxiety, psychiatric or mental disorders, medicines to help you sleep, medicines to treat high blood pressure such as clonidine, other opioids (which may be found in painkillers or certain cough mixtures e.g. morphine, dextropropoxyphene, codeine, dextromethorphan, noscapine), antihistamines which make you drowsy, or anaesthetics such as halothane.
- Bunov must be used with caution if you are also taking benzodiazepines (medicines used to treat anxiety or to help you sleep). This combination may cause serious breathing problems.
Bunov with alcohol
Alcohol may make some of the side effects worse and you may feel unwell if you drink alcohol whilst wearing Bunov. Drinking alcohol whilst using Bunov may also affect your reaction time.
Pregnancy, breast-feeding and fertility
If you are pregnant or breast-feeding, think you may be pregnant or are planning to have a baby, ask your doctor or pharmacist for advice before using this medicine.
Pregnancy
There is not sufficient experience regarding the use of buprenorphine in pregnant women. Therefore you should not use Bunov if you are pregnant or if you could become pregnant during treatment.
Breast-feeding
Buprenorphine, the active substance contained in the transdermal patch, may inhibit milk formation and passes into the breast milk. Therefore, you should not use Bunov if you are breast-feeding.
Driving and using machines
Bunov may affect your reactions to such an extent that you may not react adequately or quickly enough in the event of unexpected or sudden occurrences. This applies particularly:
- at the beginning of treatment;
- if you are taking medicines to treat anxiety or help you sleep;
- if your dose is increased.
If you are affected (e.g. feel dizzy, drowsy or have blurred vision), you should not drive or operate machinery whilst using Bunov, or for 24 hours after removing the patch.
3. How to use Bunov
Always use this medicine exactly as your doctor or pharmacist has told you. Check with your doctor or pharmacist if you are not sure.
Different strengths of Bunov are available. Your doctor will decide which strength of Bunov will suit you best.
During treatment, your doctor may change the patch you use to a smaller or larger one if necessary. Do not cut or divide the patch or use a higher dose than recommended. You should not apply more than two patches at the same time.
If you feel that the effect of the Bunov is too weak or too strong, talk to your doctor or pharmacist.
Adults and elderly patients
Unless your doctor has told you differently, attach one Bunov patch (as described in detail below) and change it every seventh day, preferably at the same time of day. Your doctor may wish to adjust the dose after 3-7 days until the correct level of pain control has been found. If your doctor has advised you to take other painkillers in addition to the patch, strictly follow the doctor’s instructions, otherwise you will not fully benefit from treatment with Bunov. The patch should be worn for 3 full days before increasing the dose, this is when the maximum effect of a given dose is established. Patients with kidney disease/dialysis patients In patients with kidney disease, no change in dose is necessary.
Patients with liver disease
In patients with liver disease, the effects and period of action of the Bunov may be affected and your doctor will therefore check on you more closely. Patients under 18 years of age
Bunov should not be used in patients below the age of 18 years.
Method of administration
Bunov transdermal patch is for transdermal use.
Bunov act through the skin. After application, buprenorphine passes through the skin into the blood.
Before applying the transdermal patch
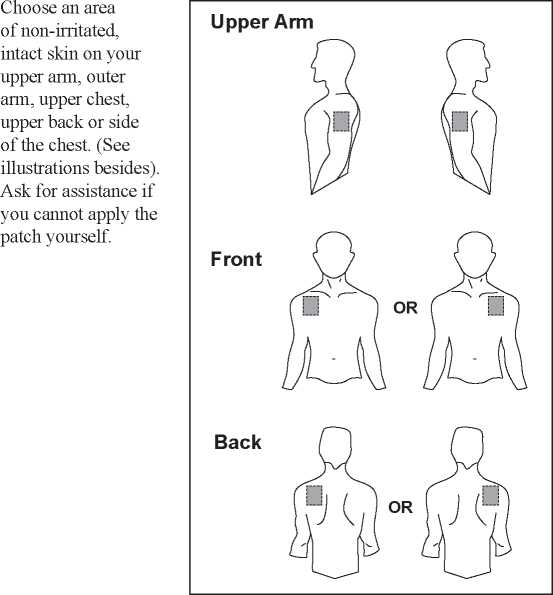
- The Bunov patch should be applied to a relatively hairless or nearly hairless skin site. If no suitable hair free sites are available the hairs should be cut off with a pair of scissors. Do not shave them off.
- Avoid skin which is red, irritated or has any other blemishes, for instance large scars.
- The area of skin you choose must be dry and clean. If necessary, wash it with cold or lukewarm water. Do not use soap, alcohol, oil, lotions or other detergents. After a hot bath or shower, wait until your skin is completely dry and cool. Do not apply lotion, cream or ointment to the chosen area. This might prevent your patch from sticking properly.
Applying the transdermal patch
- Step 1: Each transdermal patch is sealed in a sachet. Just before use, cut the sachet along the sealed edge with scissors. Take out the transdermal patch. Do not use the patch if the sachet seal is broken.
- Step 2: The sticky side of the transdermal patch is covered with a transparent protective foil. Carefully peel off one part of the foil. Try not to touch the sticky part of the transdermal patch.
Step 3: Stick the transdermal patch on to the area of skin you have chosen and remove the remaining foil.
Step 4: Press the transdermal patch against your skin with the palm of your hand and count slowly to 30. Make sure that the whole transdermal patch is in contact with your skin, especially at the edges.
Wearing the transdermal patch
You should wear the patch for seven days. Provided that you have applied the patch correctly, there is little risk of it coming off. If the edges of the patch begin to peel off, they may be taped down with a suitable skin tape. You may shower, bathe or swim whilst wearing it.
Do not expose the patch to extreme heat (e.g. heating pads, electric blanket, heat lamps, sauna, hot tubs, heated water beds, hot water bottle, etc) as this may lead to larger quantities of the active ingredient being absorbed into the blood than normal. External heat may also prevent the patch from sticking properly. If you have a high temperature this may alter the effects of Bunov (see “Warnings and precautions” section above).
In the unlikely event that your patch falls off before it needs changing, do not use the same patch again. Stick a new one on straight away (see “Changing the transdermal patch” below).
Changing the transdermal patch
- Take the old transdermal patch off.
- Fold it in half with the sticky side inwards.
- Open and take out a new patch. Use the empty sachet to dispose of the old patch. Now discard the sachet safely.
- Stick a new transdermal patch on a different appropriate skin site (as described above). You should not apply a new patch to the same site for 3-4 weeks.
- Remember to change your patch at the same time of day. It is important that you make a note of the time of day.
Duration of treatment
Your doctor will tell you how long you should be treated with the Bunov. Do not stop treatment without consulting a doctor, because your pain may return and you may feel unwell (see also “If you stop using Bunov” below).
If you use more Bunov than you should
As soon as you discover that you have used more patches than you should, remove all patches and call your doctor or hospital straight away. People who have taken an overdose may feel very sleepy and sick. They may also have breathing difficulties or lose consciousness and may need emergency treatment in hospital. When seeking medical attention make sure that you take this leaflet and any remaining patches with you to show to the doctor.
If you forget to use Bunov
Stick a new patch on as soon as you remember. Also make a note of the date, as your usual day of changing may now be different. If you are very late changing your patch, your pain may return. In this case, please contact your doctor.
Do not apply additional patches to make up for the forgotten application.
If you stop using Bunov
If you stop using Bunov too soon or you interrupt your treatment your pain may return. If you wish to stop treatment please consult your doctor. They will tell you what can be done and whether you can be treated with other medicines.
Some people may have side effects when they have used strong painkillers for a long time and stop using them. The risk of having effects after stopping Bunov is very low. However, if you feel agitated, anxious, nervous or shaky, if you are overactive, have difficulty sleeping or digestive problems, tell your doctor.
The pain relieving effect of Bunov is maintained for some time after removal of the patch. You should not start another opioid analgesic (strong painkiller) within 24 hours after removal of the patch.
If you have any further questions on the use of this medicine, ask your doctor or pharmacist.
4. Possible side effects
Like all medicines, this medicine can cause side effects, although not everybody gets them.
Serious side effects that may be associated with Bunov are similar to those seen with other strong painkillers and include difficulty in breathing and low blood pressure.
This medicine can cause allergic reactions, although serious allergic reactions are rare. Remove the patch and tell your doctor immediately if you get any sudden wheeziness, difficulties in breathing, swelling of the eyelids, face or lips, rash or itching especially those covering your whole body.
There is a risk that you may become addicted or reliant on Bunov.
In patients treated with buprenorphines, the following other side effects have been reported:
Very common (may affect more than 1 in 10 people):
• Headache, dizziness, drowsiness.
• Constipation, feeling or actually being sick.
• Itchy skin
• Rash, redness, itching, inflammation or swelling of the skin at the application site.
Common (may affect up to 1 in 10 people):
• Loss of appetite.
• Confusion, depression, anxietydifficulty in sleeping, nervousness, shaking (tremors).
• Shortness of breath.
• Abdominal pain or discomfort, diarrhoea, indigestion, dry mouth.
• Sweating, rash, skin eruptions.
• Tiredness, a feeling of unusual weakness, muscle weakness, swelling of hands, ankles or feet.
Uncommon (may affect up to 1 in 100 people):
• Mood swings, restlessness, agitation, a feeling of extreme happiness, hallucinations, nightmares , decreased sexual drive.
• Changes in taste, difficulty in speaking, reduced sensitivity to pain or touch, tingling or numbness.
• Loss of memory, migraine, fainting, problems with concentration or coordination
• Dry eyes, blurred vision.
• A ringing or buzzing sound in the ears, a feeling of dizziness or spinning.
• High or low blood pressure, chest pain, fast or irregular heartbeat.
• Cough, hiccups, wheezing.
• Wind.
• Weight loss.
• Dry skin.
• Spasms, aches and pains.
• Difficulty in beginning the flow of urine.
• Inability to fully empty the bladder.
• Fever
• An increase in accidental injuries (e.g. falls).
• Withdrawal symptoms such as agitation, anxiousness, sweating or shaking upon stopping using Bunov.
If you need to have blood tests remind your doctor that you are using Bunov. This is important because Bunov may change the way your liver works and this could affect the results of some blood tests.
Rare (may affect up to 1 in 1,000 people):
• Angina (chest pain associated with heart disease).
• Mental disorder.
• Difficulties with balance.
• Swelling of the eyelids or face, a reduction in size of the pupils in the eye.
• Difficulty in breathing, worsening of asthma, over breathing.
• A feeling of faintness, especially on standing up.
• Difficulty in swallowing.
• Local allergic reaction with marked signs of swelling (in such cases treatment should be stopped).
• Swelling and irritation inside the nose.
• Decreased erection, sexual dysfunction.
• A flu like illness.
• Flushing of the skin.
• Dehydration.
Very rare (may affect up to 1 in 10,000 people):
• Muscle twitching.
• Ear pain.
• Blisters.
Not known (frequency cannot be estimated from the available data):
• Seizures, fits or convulsions.
• Inflammation of the bowel wall. Symptoms may include fever, vomiting and stomach pain or discomfort.
• Colicky abdominal pain or discomfort.
• Feeling detached from oneself.
Reporting of side effects
If you get any side effects, talk to your doctor or pharmacist. This includes any possible side effects not listed in this leaflet. You can also report side effects directly via Yellow Card Scheme Website: www.mhra.gov.uk/yellowcard. By reporting side effects, you can help provide more information on the safety of this medicine.
5. How to store Bunov
• Keep this medicine out of the sight and reach of children.
• Do not use this medicine after the expiry date which is stated on the carton and sachet after EXP. The expiry date refers to the last day of that month.
[5 microgram/h] & [10 microgram/h]:
• Do not store above 25°C.
[20 microgram/h]:
• This medicine does not require any special storage conditions.
• Do not use the patch if the sachet seal is broken.
• Used patches must be folded over on themselves with the adhesive layer inwards, and discarded safely.
• Do not throw away any medicines via wastewater or household waste. Ask your pharmacist how to throw away medicines you no longer use. These measures will help protect the environment.
6. Contents of the pack and other information
What Bunov patches contain
- The active substance is buprenorphine.
[5 microgram/h:]
Each transdermal patch contains 5 mg of buprenorphine in a patch size of 6.25 cm2 and releases 5 micrograms of buprenorphine per hour (over a period of 7 days).
[10 microgram/h:]
Each transdermal patch contains 10 mg of buprenorphine in a patch size of 12.5 cm2 and releases 10 micrograms of buprenorphine per hour (over a period of 7 days).
[20 microgram/h:]
Each transdermal patch contains 20 mg of buprenorphine in a patch size of 25 cm2 and releases 20 micrograms of buprenorphine per hour (over a period of 7 days).
- The other ingredients are:
Adhesive matrix (containing buprenorphine): povidone K90, levulinic acid, oleyl oleate, Poly[acrylic acid-co-butylacrylate-co-(2-ethylhexyl) acrylate-co-vinylacetate] (5:15:75:5)
Adhesive matrix (without buprenorphine): Poly[(2-ethylhexyl)acrylate-
co-glycidylmethacrylate-co-(2-hydroxyethyl)acrylate-co-vinylacetate]
(68:0,15:5:27),
Separating foil between adhesive matrices with and without buprenorphine: Polyethylene terephthalate film,
Backing foil: polyester,
Release liner: Polyethylene terephthalate film, siliconised Blue printing ink
What Bunov look like and contents of the pack
Transdermal patch Three sizes are available.
[5 microgram/h:]
Each transdermal patch is beige coloured with rounded corners and is imprinted with “Buprenorphin” and “5 pg/h”
[10 microgram/h:]
Each transdermal patch is beige coloured with rounded corners and is imprinted with “Buprenorphin” and “10 pg/h”
[20 microgram/h:]
Each transdermal patch is beige coloured with rounded corners and is imprinted with “Buprenorphin” and “20 pg/h”
One transdermal patch is sealed in one child-resistant sachet. The patches are available in cartons containing 2, 4, 5, 8 or 12 transdermal patches.
Not all pack sizes may be marketed.
Marketing Authorisation Holder
Glenmark Pharmaceuticals Europe Limited
Laxmi House
2B Draycott Avenue
Kenton, Middlesex
HA30BU
United Kingdom
Manufacturer
Tesa Labtec GmbH Heykenaukamp 10 21147 Hamburg Germany
This leaflet was last revised in June 2016
