Carteolol Hydrochloride Eye Drops 1%
531 LEAFLET Teoptic 20150413
PACKAGE LEAFLET: INFORMATION FOR THE USER
TEOPTIC EYE DROPS 1% CARTEOLOL HYDROCHLORIDE EYE DROPS 1% TEOPTIC EYE DROPS 2% CARTEOLOL HYDROCHLORIDE EYE DROPS 2% (carteolol hydrochloride)
Your medicine is known by either of the above names but will be referred to as Teoptic throughout the following leaflet.
Read all of this leaflet carefully before you start to use this
medicine because it contains important information for you.
• Keep this leaflet. You may need to read it again.
• If you have further questions, ask your doctor, pharmacist or nurse.
• This medicine has been prescribed for you only. Do not pass it on to others. It may harm them, even if their signs of illness are the same as yours.
• If you get any side effects, talk to your doctor, pharmacist or nurse. This includes any possible side effects not listed in this leaflet.
What is in this leaflet:
1. What Teoptic is and what it is used for
2. What you need to know before you use Teoptic
3. How to use Teoptic
4. Possible side effects
5. How to store Teoptic
6. Contents of the pack and other information
1. WHAT TEOPTIC IS AND WHAT IT IS USED FOR
Teoptic is available in two different strengths containing either 1% or 2% of the active ingredient, carteolol hydrochloride.
Carteolol hydrochloride is one of a group of drugs called beta-blockers which can help to reduce pressure in the eye. Teoptic is used to treat conditions such as glaucoma (increased pressure in the eye).
2. WHAT YOU NEED TO KNOW BEFORE YOU USE TEOPTIC
Do not use Teoptic eye drops solution:
• if you are allergic to carteolol hydrochloride, beta-blockers or to any of the other ingredients of this medicine (listed in section 6.)
• if you have now or have had in past respiratory problems such as asthma, severe chronic obstructive bronchitis (severe lung disease which may cause wheeziness, difficulty in breathing and/or long-standing cough)
• if you have a slow heart beat, heart failure or disorders of heart rhythm (irregular heart beats).
Warnings and precautions
Before you use this medicine, tell your doctor if you have now or have
had in the past
• coronary heart disease (symptoms can include chest pain or tightness, breathlessness or choking), heart failure
• disturbances of heart rate such as slow heart beat
• breathing problems, asthma or chronic obstructive pulmonary disease
• poor blood circulation disease (such as Raynaud's disease or Raynaud's syndrome)
• diabetes as carteolol hydrochloride may mask signs and symptoms of low blood sugar
• overactivity of the thyroid gland as carteolol hydrochloride may mask signs and symptoms
Tell your doctor before you have an operation that you are using
Teoptic as carteolol hydrochloride may change effects of some
medicines used during anaesthesia.
Other medicines and Teoptic
Teoptic can affect or be affected by other medicines you are using, including other eye drops for the treatment of glaucoma. Tell your doctor if you are using or intend to use medicines to lower blood pressure, heart medicine or medicines to treat diabetes.
Please tell your doctor or pharmacist if you are taking or have recently taken any other medicines, including medicines obtained without a prescription.
Pregnancy, breast-feeding and fertility
Do not use Teoptic if you are pregnant or trying to get pregnant unless your doctor considers it necessary.
Do not use Teoptic if you are breast-feeding. Carteolol hydrochloride may get into your milk.
Ask your doctor for advice before taking any medicine during breastfeeding.
Driving and using machines
Some people may have problems with their eyes such as blurred vision, while they are using Teoptic. If you are affected, you should not drive or use machinery.
Teoptic contains benzalkonium chloride
Benzalkonium chloride may cause eye irritation. Avoid contact with soft lenses.
Take out lenses before you use Teoptic and do not put them back for at least 15 minutes. Benzalkonium chloride is known to discolour soft contact lenses.
3. HOW TO USE TEOPTIC
Always use Teoptic eye drops solution exactly as your doctor has told you. You should check with your doctor or pharmacist if you are not sure.
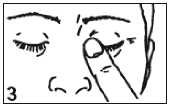
The recommended dose is one drop into the affected eye twice daily. After using Teoptic, press a finger into the corner of your eye, by the nose (picture 3) for 2 minutes. This helps to stop carteolol hydrochloride getting into the rest of the body.
Use in children and adolescents
Teoptic Eye Drops are not recommended for children.
How to use Teoptic
Wash your hands before using the drops
Remove the cap from the dropper bottle
Make sure that the tip of the bottle does not touch anything
Hold the bottle in one hand between the thumb and forefinger
Tilt your head back, and use your other forefinger to pull down the lower eyelid
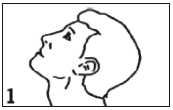
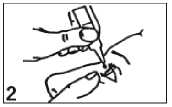
Place the dropper tip close to your eye, but not touching the eye or lid, and gently squeeze the bottle to release one drop into your eye
• Close your eyelid and gently press the corner of your eye with yo ur forefinger for 2 minutes
• Replace the cap and wash your hands.
If you forget to use Teoptic
If you forget to use your eye drops, put them in as soon as possible. Do not take a double dose to make up for a forgotten dose.
If you use more Teoptic than you should
If you use too much or if you accidentally swallow the eye drops, see your doctor at once or go to your nearest hospital casualty department. Take your medicine with you.
If you have any further questions on the use of this medicine, ask your doctor, pharmacist or nurse.
4. POSSIBLE SIDE EFFECTS
Like all medicines, Teoptic eye drops, solution can cause side effects, although not everybody gets them
You can usually carry on taking the drops, unless the effects are serious. If you are worried, talk to a doctor or pharmacist. Do not stop using Teoptic without speaking to your doctor.
Like other medicines applied into eyes, carteolol hydrochloride is absorbed into the blood. This may cause similar side effects as seen with ‘intraveneous' and/or ‘oral' as applicable beta-blocking agents. Incidence of side effects after topical ophthalmic administration is lower than when medicines are, for example, taken by mouth or injected. Listed side effects include reactions seen within the class of beta-blockers when used for treating eye conditions:
• Generalized allergic reactions including swelling beneath the skin that can occur in areas such as the face and limbs, and can obstruct the airway which may cause difficulty swallowing or breathing, hives or itchy rash, localized and generalized rash, itchiness, severe sudden life-threatening allergic reaction.
• Low blood glucose levels.
• Difficulty sleeping (insomnia), depression, nightmares, memory loss.
• Fainting, stroke, reduced blood supply to the brain, increases in signs and symptoms of myasthenia gravis (muscle disorder), dizziness, unusual sensations like pins and needles, and headache.
• Signs and symptoms of eye irritation (e.g. burning, stinging, itching, tearing, redness), inflammation of the eyelid, inflammation in the cornea, blurred vision and detachment of the layer below the retina that contains blood vessels following filtration surgery which may cause visual disturbances, decreased corneal sensitivity, dry eyes, corneal erosion (damage to the front layer of the eyeball), drooping of the upper eyelid (making the eye stay half closed), double vision, unusual sensitivity to light.
• Slow heart rate, chest pain, palpitations, oedema (fluid build up), changes in the rhythm or speed of the heartbeat, congestive heart failure (heart disease with shortness of breath and swelling of the feet and legs due to fluid build up), a type of heart rhythm disorder, heart attack, heart failure.
• Low blood pressure, Raynaud's phenomenon, cold hands and feet.
• Constriction of the airways in the lungs (predominantly in patients with pre-existing disease), difficulty breathing, cough.
• Taste disturbances, nausea, indigestion, diarrhoea, dry mouth, abdominal pain, vomiting.
• Hair loss, skin rash with white silvery coloured appearance (psoriasiform rash) or worsening of psoriasis, skin rash.
• Muscle pain not caused by exercise.
• Sexual dysfunction, decreased libido.
• Muscle weakness/tiredness, feeling of discomfort, feeling of tension or fullness in the nose, cheeks and behind your eyes, sometimes with a throbbing ache, fever, stuffy nose and loss of the sense of smell (sinusitis).
5. HOW TO STORE TEOPTIC
• KEEP OUT OF THE SIGHT AND REACH OF CHILDREN.
• Do not store the eye drops above 25°C.
• Do not break the bottle seal until you are going to use the drops. They are sterile until the seal is broken. They must be used within 28 days of first breaking the seal.
• Do not use the drops after the expiry date shown on the pack.
• If your doctor tells you to stop using the drops, please take them back to the pharmacist for safe disposal. Only keep them if your doctor tells you to. If the drops become discoloured or show any other signs of deterioration take them back to the pharmacist.
• REMEMBER this medicine was prescribed for you. You should not give it to anyone else even if you think their symptoms are similar. It may harm them.
6. FURTHER INFORMATION
What Teoptic contains
• Teoptic Eye Drops 1% contain 1% w/v of the active ingredient carteolol hydrochloride.
• Teoptic Eye Drops 2% contain 2% w/v of the active ingredient carteolol hydrochloride.
• The other ingredients are: benzalkonium chloride (a preservative), sodium chloride, sodium acid phosphate, sodium phosphate and purified water.
What Teoptic looks like and contents of the pack
Teoptic is a clear sterile solution. The bottle is fitted with a dropper
device and a white plastic cover with tamper-evident seal.
Teoptic is available as bottle of 5 ml solution.
Product Licence holder
Procured from within the EU and repackaged by the Product Licence holder: Chemilines Ltd, Chemilines House, Alperton Lane, Wembley, Middlesex, HA0 1DX, UK.
Manufacturer
This product is manufactured by Otsuka Pharmaceutical s.a Avda. Diagonal, 609-615 08028 Barcelona.
| POM | PLPI 08747/0531 PLPI 08747/0532
Leaflet revision date: 13 April 2015
Teoptic is a registered trademark used by agreement with Otsuka, Japan.
531 LEAFLET Teoptic 20150413
Reporting of side effects
If you get any side effects, talk to your doctor, pharmacist or nurse. This includes any possible side effects not listed in this leaflet. You can also report side effects directly via the Yellow Card Scheme at: www.mhra.gov.uk/yellowcard
By reporting side effects, you can help provide more information on the safety of this medicine.