Clinimix N17G35E Solution For Infusion
Package leaflet: Information for the patient CLINIMIX N17G35E, Solution for Infusion
Read all of this leaflet carefully before this medicine is administered, because it contains important information for you.
- Keep this leaflet. You may need to read it again.
- If you have any further questions, ask your doctor or nurse.
- If you get any side effects, talk to your doctor or nurse. This includes any possible side effects not listed in this leaflet. See section 4.
What is in this leaflet:
1. What CLINIMIX is and what it is administered for
2. What you need to know before CLINIMIX is administered
3. How CLINIMIX is administered
4. Possible side effects
5. How to store CLINIMIX
6. Contents of the pack and other information
1. What CLINIMIX is and what it is administered for
CLINIMIX is a solution for infusion. It is supplied in a bag with 2 chambers. One chamber contains an amino acid solution with electrolytes and the second contains a glucose solution with calcium chloride. The chambers are separated by a non-permanent seal. Just before administration, the contents of the chambers are mixed together by rolling the top of the bag to open the seals.
CLINIMIX is administered to provide nutrition in adults and children by a tube into a vein when normal feeding by mouth is not suitable.
CLINIMIX must only be administered under medical supervision.
What you need to know before CLINIMIX is administered
2.
CLINIMIX shall not be administered if
- you are allergic to any of the ingredients of this medicine (listed in section 6).
- your body has problems using certain amino acids.
- you have too much sugar in your blood (severe hyperglycaemia),
- your blood is excessively acid (metabolic acidosis due to an excess of lactate),
- your blood level of sodium, potassium, magnesium, calcium and/or phosphorus is too high (hypernatraemia, hyperkalemia, hypermagnesemia, hypercalcemia and/or hyperphosphatemia)
- In children less than 28 days of age, ceftriaxone must not be co-administered with calcium-containing IV solutions, because particles may form.
In all cases, your doctor will base his/her decision on whether you should receive this medicine on factors such as age, weight and clinical condition, together with the results of any tests performed.
Warnings and precautions
Talk to your doctor or nurse before CLINIMIX is administered.
If any abnormal signs or symptoms of an allergic reaction develop, such as fever, chills, skin rashes or difficulty in breathing, excessive sweating, nausea or headache, tell the doctor or the nurse: the infusion will be stopped immediately. Your doctor will monitor your condition while you receive this medicine and may change the dosage or give you additional nutrients such as lipids, vitamins, electrolytes and trace elements if he/she feels it is appropriate.
Certain medications and illnesses can increase the risk of developing infection or sepsis (bacteria in the blood). There is a particular risk of infection or sepsis when a tube (intravenous catheter) is placed in your vein. Your doctor will carefully watch you for any signs of infection. Using aseptic (germ free) techniques when placing and maintaining the catheter and when making the nutritional formula can reduce the risk of infection.
CLINIMIX with electrolytes contains calcium. It should not be given-together with the antibiotic ceftriaxone because particles may form.
If you are severely malnourished such that you need to receive feeding through a vein, it is recommended that parenteral nutrition is started slowly and carefully.
Your doctor will monitor your condition at the onset of the infusion, particularly if you currently have liver, kidney, adrenal, heart or circulation problems. Your doctor should also be aware of severe conditions affecting how your body handles sugars, fats, proteins or salt (metabolic disorders).
Should any abnormal sign occur, including venous irritation, the infusion must be stopped.
To check the effectiveness and ongoing safety of the administration, your doctor will perform clinical and laboratory tests while you are receiving this medicine. If you are given this medicine for several weeks, your blood will be monitored on a regular basis. In particular, in case of glucose intolerance, blood and urine glucose should be monitored on a routine basis, and, if you are a diabetic patient, insulin dosage may have to be adapted.
Other medicines and CLINIMIX
Tell your doctor if you are taking or using, have recently taken or used or might take or use any other medicines.
CLINIMIX with electrolytes contains calcium. It should not be given-together with the antibiotic ceftriaxone because particles may form.
Due to the potassium content of CLINIMIX, special care should be taken in patients treated with potassium-saving diuretics (e.g, amiloride, spironolactone, triamterene) angiotensin converting enzyme (ACE) inhibitors, angiotensin II receptor antagonists or the immunosuppressants tacrolimus or cyclosporine in view of the risk of hyperkalemia.
Pregnancy, breast-feeding and fertility
If you are pregnant or breast-feeding, think you may be pregnant or are planning to have a baby, ask your doctor for advice before this medicine is administered.
3. How CLINIMIX is administered
Before the product is administered, the non-permanent seal between the two compartments should be broken and the contents of the two compartments should be mixed.
CLINIMIX can be given in adults and children.
It is a solution for infusion, to be administered via a plastic tube into a vein in your arm or into a large vein in your chest.
Dosage - Adults and Children
Your doctor will decide the dose you will need and for how long it will be given. This will depend on age, weight and height, clinical condition, daily fluid volume, energy and nitrogen requirements.
Always take CLINIMIX exactly as your doctor has told you. You should check with your doctor if you are not sure.
The prescription may be continued for as long as it is needed, depending upon your clinical condition. The infusion of one bag usually lasts between 8 and 24 hours.
If more CLINIMIX was administered than should have been
If the dose given is too high or the infusion too fast, you may have an increased volume of circulating blood or your blood may become too acid. The glucose content may increase the glucose in your blood and urine. Giving too high a volume may cause nausea, vomiting, shivering, and electrolyte disturbances, in such situations the infusion should be stopped immediately.
In some severe cases, your doctor may have to give you temporary renal dialysis to help your kidneys eliminate the excess product.
To prevent these events occurring, your doctor will regularly monitor your condition and test your blood parameters.
If you have any further questions on the use of this product, ask your doctor.
4. Possible side effects
Like all medicines, this medicine can cause side effects, although not everybody gets them. If you notice any changes in the way you feel during or after the treatment, tell your doctor or nurse straight away.
The tests your doctor will perform while the medicine is administered to you should minimise the risk of side effects.
If any abnormal signs or symptoms of an allergic reaction develop, such as abnormally low or high blood pressure, appearance of a blue or purple coloration of the skin, abnormally high heart rate, breathing difficulties, vomiting, nausea, skin rashes, raised body temperature, excessive sweating chills, and shivering, the infusion will be stopped immediately.
Other side effects have been noticed, occurring more or less frequently:
• Anaphylaxis (a serious allergic reaction that is rapid in onset and may cause death)
• High blood level of glucose, ammonia and nitrogen-containing compounds
• Impairment of hepatic functions, abnormal blood test for the liver function
• Inflammation of the gall bladder, the presence of gallstones in the gallbladder
• Vein inflammation at infusion site, venous irritation, pain, irritation, warmth, swelling
• Presence of glucose in urine
• Diabetic coma
• Formation of small particles blocking lung blood vessels Reporting of side effects
If you get any side effects, talk to your doctor or nurse. This includes any possible side effects not listed in this leaflet. You can also report side effects directly (see details below). By reporting side effects you can help provide more information on the safety of this medicine.
Via the Yellow Card Scheme at: www.mhra.gov.uk/yellowcard
5. How to store CLINIMIX
Keep this medicine out of the sight and reach of children.
This medicine should not be administered after the expiry date, which is stated on the container and the outer packaging (MM/YYYY). The expiry date refers to the last day of that month.
Do not freeze.
Keep the container in the outer carton.
Do not throw away any medicines via wastewater or household waste. Ask your pharmacist how to throw away medicines you no longer use. These measures will help protect the environment.
6. Contents of the pack and other information What CLINIMIX contains
The active substances for each bag of the reconstituted solution are:
|
Active substances |
1 l |
1.5 l |
2 l |
|
L-alanine |
10.35 g |
15.53 g |
20.70 g |
|
L-arginine |
5.75 g |
8.63 g |
11.50 g |
|
Glycine |
5.15 g |
7.73 g |
10.30 g |
|
L-histidine |
2.40 g |
3.60 g |
4.80 g |
|
L-isoleucine |
3.00 g |
4.50 g |
6.00 g |
|
L-leucine |
3.65 g |
5.48 g |
7.30 g |
|
L-lysine |
2.90 g |
4.35 g |
5.80 g |
|
(as lysine hydrochloride) |
(3.63 g) |
(5.44 g) |
(7.25 g) |
|
L-methionine |
2.00 g |
3.00 g |
4.00 g |
|
L-phenylalanine |
2.80 g |
4.20 g |
5.60 g |
|
L-proline |
3.40g |
5.10 g |
6.80 g |
|
L-serine |
2.50 g |
3.75 g |
5.00 g |
|
L-threonine |
2.10 g |
3.15 g |
4.20 g |
|
L-tryptophan |
0.90 g |
1.35 g |
1.80 g |
|
L-tyrosine |
0.20 g |
0.30 g |
0.40 g |
|
L-valine |
2.90 g |
4.35 g |
5.80 g |
|
Sodium acetate, 3H2O |
3.40 g |
5.10 g |
6.80 g |
|
Dibasic potassium phosphate |
2.61 g |
3.92 |
5.22 g |
|
Sodium chloride |
0.59 g |
0.88 g |
1.17 g |
|
Magnesium chloride, 6H2O |
0.51 g |
0.77 g |
1.02 g |
|
Glucose anhydrous |
175 g |
263 g |
350 g |
|
(as glucose monohydrate) |
(193 g) |
(289 g) |
(385 g) |
|
Calcium chloride, 2H2O |
0.33 g |
0.50 g |
0.66 g |
The other ingredients are:
- acetic acid, hydrochloric acid (to adjust pH of the solution),
- water for injections.
What CLINIMIX looks like and contents of the pack
CLINIMIX is a solution for infusion packaged in a two-chamber bag, which is a multi-layer plastic bag. The inner (contact) layer of the bag material is designed to be compatible with the constituents and authorised additives.
Prior to the reconstitution, the amino acid and the glucose solutions are clear and colourless or slightly yellow. After the reconstitution, the solution is clear and colourless or slightly yellow too.
To prevent contact with oxygen from the air the bag is packaged in an oxygen barrier overpouch, which contains an oxygen absorber sachet.
Pack sizes 1000 ml bag: carton with 8 bags 1000 ml: 1 bag
1500 ml bag: carton with 6 bags 1500 ml: 1 bag
2000 ml bag: carton with 4 bags 2000 ml: 1 bag
Not all pack sizes may be marketed.
Marketing Authorisation Holder
For any information about CLINIMIX, please contact the Marketing Authorisation Holder:
Baxter Healthcare Ltd
Caxton Way
Thetford
Norfolk
IP24 3SE
The United Kingdom
Manufacturers
Baxter Healthcare, Caxton way, Thetford, Norfolk IP24 3SE, The United Kingdom Baxter, Boulevard Rene Branquart, 80, 7860 Lessines, Belgium
This medicinal product is authorised in the Member States of the EEA under the following names:
CLINIMIX N17G35E, Solution for Infusion
In some countries it is registered under a different trade name, as described below:
Austria: CLINIMIX 5% G-E
Germany: CLINIMIX 5 % G-E
This leaflet was last revised in 05/2015
The following information is intended for healthcare professionals only:
1. QUANTITATIVE COMPOSITION
After the contents of the two compartments have been mixed, the composition of the binary mixture, for all available bag sizes, provides the following:
|
1 l |
1.5 l |
2 l | |
|
Nitrogen (g) |
8.3 |
12.4 |
16.5 |
|
Amino acids (g) |
50 |
75 |
100 |
|
Glucose (g) |
175 |
263 |
350 |
|
Total calories (kcal) |
900 |
1350 |
1800 |
|
Glucose calories (kcal) |
700 |
1050 |
1400 |
|
Sodium (mmol) |
35 |
53 |
70 |
|
Potassium (mmol) |
30 |
45 |
60 |
|
Magnesium (mmol) |
2.5 |
3.8 |
5.0 |
|
Calcium (mmol) |
2.3 |
3.4 |
4.5 |
|
Acetate (mmol) |
75 |
113 |
150 |
|
Chloride (mmol) |
40 |
60 |
80 |
|
Phosphate as HPO42- (mmol) |
15 |
23 |
30 |
|
pH Osmolarity (mOsm/l) |
6 1625 | ||
2. POSOLOGY AND METHOD OF ADMINISTRATION
Only administer the product after the non-permanent seal between the two compartments has been broken and the contents of the two compartments have been mixed.
Dosage and infusion rate
The dosage is chosen according to the metabolic needs, the energy expenditure, and the clinical status of the patient.
In adults, the requirements range from 0.16 g of nitrogen/kg/d (approximately 1 g of amino acid/kg/d) to 0.35 g of nitrogen/kg/d (approximately 2 g of amino acid/kg/d).
In infants, the requirements range from 0.35 g of nitrogen/kg/d (approximately 2 g of amino acid/kg/d) to 0.45 g of nitrogen/kg/d (approximately 3 g of amino acid/kg/d).
The calorie requirements range from 25 kcal/kg/d to 40 kcal/kg/d, depending on the nutritional status of the patient and the degree of catabolism.
The rate of administration should be adjusted according to the dosage, the characteristics of the infused solution, the total volume intake per 24 hours and the duration of the infusion.
The infusion time should be higher than 8 hours. Normally, the flow rate is increased gradually during the first hour without exceeding 1.4 ml per kilogram of bodyweight per hour, and the maximal dose is 30 ml per kilogram of bodyweight per day.
Route of administration
The choice of the peripheral or central vein depends on the final osmolarity of the mixture. The general accepted limit for peripheral infusion is about 800 mOsm/l but it varies considerably with the age and the general condition of the patient and the characteristics of the peripheral veins.
3. SPECIAL WARNINGS AND PRECAUTIONS FOR USE
WARNINGS
Hypersensitivity/infusion reactions including hypotension, hypertension, peripheral cyanosis, tachycardia, dyspnoea, vomiting, nausea, urticaria, rash, pruritus, erythema, hyperhidrosis, pyrexia, and chills have been reported with CLINIMIX formulations.
Anaphylaxis has been reported with other parenteral nutrition products.
Special clinical monitoring is required at the beginning of any intravenous infusion. Should any abnormal sign or symptom occur, e.g. for hypersensitivity or infusion reaction, the infusion must be stopped immediately.
Solutions containing glucose should be used with caution, if at all, in patients with known allergy to corn or corn products.
Pulmonary vascular precipitates have been reported in patients receiving parenteral nutrition. In some cases, fatal outcomes have occurred. Excessive addition of calcium and phosphate increases the risk of the formation of calcium phosphate precipitates. Precipitates have been reported even in the absence of phosphate salt in the solution. Precipitation distal to the in-line filter and suspected in vivo precipitate formation have also been reported.
If signs of pulmonary distress occur, the infusion should be stopped and medical evaluation initiated.
In addition to inspection of the solution, the infusion set and catheter should also periodically be checked for precipitates.
In patients older than 28 days (including adults), ceftriaxone must not be administered simultaneously with intravenous calcium-containing solutions, including CLINIMIX N17G35E, through the same infusion line (e.g., via a Y - connector).
If the same infusion line is used for sequential administration, the line must be thoroughly flushed with a compatible fluid between infusions.
Infection and sepsis may occur as a result of the use of intravenous catheters to administer parenteral formulations, poor maintenance of catheters or contaminated solutions. Immunosuppression and other factors such as hyperglycaemia, malnutrition and/or their underlying disease state may predispose patients to infectious complications.
Careful symptomatic and laboratory monitoring for fever/chills, leukocytosis, technical complications with the access device, and hyperglycaemia can help recognize early infections.
The occurrence of septic complications can be decreased with heightened emphasis on aseptic technique in catheter placement, maintenance, as well as aseptic technique in nutritional formula preparation.
Refeeding severely undernourished patients may result in the refeeding syndrome that is characterized by the shift of potassium, phosphorus, and magnesium intracellularly as the patient becomes anabolic. Thiamine deficiency and fluid retention may also develop. Careful monitoring and slowly increasing nutrient intakes while avoiding overfeeding can prevent these complications.
Hypertonic solutions may cause venous irritation if infused into a peripheral vein. The choice of a peripheral or central vein depends on the final osmolarity of the mixture.
The general accepted limit for peripheral infusion is about 800 mOsm/l but it varies considerably with the age and the general condition of the patient and the characteristics of the peripheral veins.
Do not connect bags in series in order to avoid air embolism due to possible residual air contained in the primary bag.
PRECAUTIONS
Severe water and electrolyte equilibration disorders, severe fluid overload states, and severe metabolic disorders should be corrected before starting the infusion.
Metabolic complications may occur if the nutrient intake is not adapted to the patient's requirements, or the metabolic capacity of any given dietary component is not accurately assessed. Adverse metabolic effects may arise from administration of inadequate or excessive nutrients or from inappropriate composition of an admixture for a particular patient's needs.
Frequent clinical evaluation and laboratory determinations are necessary for correct monitoring during administration. These should include ionogram and kidney and liver function tests.
The electrolyte requirements of patients receiving the solutions should be carefully determined and monitored especially for the electrolyte-free solutions.
Glucose intolerance is a common metabolic complication in severely stressed patients. With the infusion of the products, hyperglycaemia, glycosuria, and hyperosmolar syndrome may occur. Blood and urine glucose should be monitored on a routine basis and for diabetics insulin dosage should be adapted, if necessary.
Use with caution in patients with renal insufficiency, particularly if hyperkalaemia is present, because of the risk of developing or worsening metabolic acidosis and hyperazotemia if extra-renal waste removal is not being performed. Fluid and electrolyte status should be closely monitored in these patients. In case of severe kidney failure, specially formulated amino acid solutions should be preferred.
Caution should be exercised in administering CLINIMIX to patients with adrenal insufficiency.
Care should be taken to avoid circulatory overload particularly in patients with pulmonary oedema, cardiac insufficiency and/or failure. Fluid status should be closely monitored.
In patients with pre-existing liver disease or hepatic insufficiency, apart from routine liver function tests, possible symptoms of hyperammonaemia should be controlled.
Hepatobiliary disorders including cholestasis, hepatic steatosis, fibrosis and cirrhosis, possibly leading to hepatic failure, as well as cholecystitis and cholelithiasis are known to develop in some patients on parenteral nutrition. The aetiology of these disorders is thought to be multifactorial and may differ between patients. Patients developing abnormal laboratory parameters or other signs of hepatobiliary disorders should be assessed early by a clinician knowledgeable in liver diseases in order to identify possible causative and contributory factors, and possible therapeutic and prophylactic interventions.
Increase in blood ammonia levels and hyperammonemia may occur in patients receiving amino acid solutions. In some patients this may indicate the presence of a congenital disorder of amino acid metabolism (see section 4.3 of the SmPC) or hepatic insufficiency.
Blood ammonia should be measured frequently in newborns and infants to detect hyperammonemia, which may indicate the presence of a congenital abnormality of amino acid metabolism.
Depending on extent and aetiology, hyperammonemia may require immediate intervention.
A too rapid infusion of amino acid may result in nausea, vomiting and chills. In such cases, discontinue the infusion immediately.
In general, dose selection for an elderly patient should be cautious, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or drug therapy.
Paediatric population
• There have been no studies performed in the paediatric population.
• See above regarding monitoring for hyperammonemia in paediatric patients.
4. PRACTICAL INFORMATION ON PREPARATION AND HANDLING
Warning: Administer the product only after breaking the seal and mixing the contents of both compartments.

Place the bag flat on a horizontal and clean surface with the handle in front of you.
Tear from the top to open the overpouch.
Peel the front of the overpouch to reveal the CLINIMIX bag. Discard the overpouch and oxygen absorber sachet.
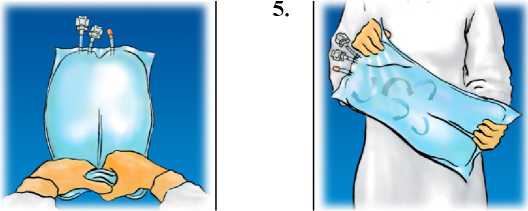
Mix by turning the bag upside-down at least 3 times.
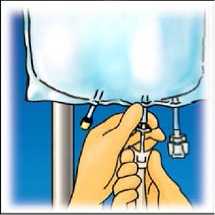
Hang the bag. Twist off the protector from the administration outlet. Firmly plug the spike connector.
Lift the hanger area to remove the solution from the top of the bag. Firmly roll the bag until the peal seal is fully open (approximately half way).
Use only if the solution is clear, colourless or slightly yellow and if the container is undamaged. CLINIMIX should be at room temperature before use.
CLINIMIX activation can be performed in the overpouch or after its removal.
For single use only.
Do not store partly used containers and discard all equipment after use.
Do not reconnect partially used bag.
Do not connect in series.
Supplementation
Lipids, vitamins and trace elements should be provided to patients receiving parenteral nutrition for a long period.
If additives are necessary, compatibilities should be checked and the stability of mixtures should be controlled.
The supplementation can be made after opening the peel seal (once the two solutions have been mixed) for all additives. CLINIMIX may be supplemented with:
- Lipid emulsions (for example ClinOleic) at a rate of 50 to 250 ml per litre of CLINIMIX
|
CLINIMIX N17G35E |
CLINIMIX N17G35E |
CLINIMIX N17G35E | |
|
1 L + 250 mL lipids |
1.5 L + 500 mL lipids |
2 L + 500 mL lipids | |
|
20% |
20% |
20% | |
|
Nitrogen (g) |
8.3 |
12.4 |
16.5 |
|
Amino acids (g) |
50 |
75 |
100 |
|
Glucose (g) |
175 |
263 |
350 |
|
Lipid (g) |
50 |
100 |
100 |
|
Total calories (kcal) |
1400 |
2350 |
2800 |
|
Glucose calories (kcal) |
700 |
1050 |
1400 |
|
Lipid calories (kcal) |
500 |
1000 |
1000 |
|
Glucose/lipids Ratio |
58 / 42 |
51 / 49 |
58 / 42 |
|
Sodium (mmol) |
35 |
53 |
70 |
|
Potassium (mmol) |
30 |
45 |
60 |
|
Magnesium (mmol) |
2.5 |
3.8 |
5.0 |
|
Calcium (mmol) |
2.3 |
3.4 |
4.5 |
|
Acetate (mmol) |
75 |
113 |
150 |
|
Chloride (mmol) |
40 |
60 |
80 |
|
Phosphate as HPO42- |
15 |
23 |
30 |
|
(mmol) | |||
|
pH |
6 |
6 |
6 |
|
Osmolarity (mOsm/l) |
1360 |
1290 |
1360 |
Electrolytes: per litre of CLINIMIX
|
Sodium |
Potassium |
Magnesium |
Calcium | |
|
Up to a final concentration of |
80 mmol |
60 mmol |
5.6 mmol |
3.0 mmol |
Trace elements: per litre of CLINIMIX
|
Up to a final concentration of |
Copper |
10 pmol |
Zinc |
77 pmol |
|
Chromium |
0.14 pmol |
Manganese |
2.5 pmol | |
|
Fluorine |
38 pmol |
Cobalt |
0.0125 pmol | |
|
Selenium |
0.44 pmol |
Molybdenum |
0.13 pmol | |
|
Iodine |
0.5 pmol |
Iron |
10 pmol |
Vitamins: per litre of CLINIMIX
|
Up to a final concentration of |
vitamin A |
1750IU |
Biotin |
35 Pg |
|
vitamin B6 |
2.27 mg |
vitamin B1 |
1.76 mg | |
|
vitamin D |
110 IU |
Folic acid |
207 pg | |
|
vitamin B12 |
30 Pg |
vitamin B2 |
2.07 mg | |
|
vitamin E |
5.1 mg |
vitamin C |
63 mg | |
|
vitamin PP |
23 mg |
vitamin B5 |
8.63 mg | |
|
vitamin K |
75 pg |
Stability data for supplementation of CLINIMIX with other marketed lipid emulsions and other additives or nutrients are available upon request.
If some light creaming is observed, mix thoroughly the admixture by gentle agitation to get a uniform emulsion before the infusion.
Additions should be performed under aseptic conditions.
Additions can be made with a syringe or a transfer set.
• Addition with a syringe or a transfer set fitted with a needle
- Prepare the injection site (the single port, see Figure 2 or 4 in the SmPC).
- Puncture the port and inject.
- Mix the solutions and the additives.
• Addition with a transfer set fitted with a spike
- Please refer to the “Directions for use” of the lipid transfer set used.
- Connect the spike to the transfusion site (the longest port).
Incompatibilities
Additives may be incompatible, refer to the manufacturer for further details.
If additives are necessary, compatibilities should be checked and the stability of mixtures should be controlled.
The solution should not be administered with, before, or after an administration of blood through the same equipment because of the possibility of pseudoagglutination.
CLINIMIX N17G35E contains calcium ions which pose additional risk of coagulation precipitated in citrate anticoagulated/preserved blood or components.
As with any parenteral nutrition admixture, calcium and phosphate ratios must be considered. Excess addition of calcium and phosphate, especially in the form of mineral salts, may result in the formation of calcium phosphate precipitates.
As for other calcium-containing infusion solutions, concomitant treatment with ceftriaxone and CLINIMIX N17G35E is contraindicated in newborns (<28 days of age), even if separate infusion lines are used (risk of fatal ceftriaxone calcium salt precipitation in the neonate’s bloodstream).
In patients older than 28 days (including adults), ceftriaxone must not be administered simultaneously with intravenous calcium-containing solutions, including CLINIMIX N17G35E, through the same infusion line (see section Warnings).
If the same infusion line is used for sequential administration, the line must be thoroughly flushed with a compatible fluid between infusions.
5. SHELF LIFE 2 years when stored in the overpouch.
It is recommended that the product be used immediately after the non-permanent seal between the 2 chambers have been opened. However, once reconstituted (i.e. internal non-permanent seal opened), stability of the reconstituted emulsion has been demonstrated for a maximum of 7 days between 2°C and 8°C followed by a maximum of 48 h at temperature not exceeding 25°C.
When additions have been made, from a microbiological point of view, the admixture should be used immediately. If not used immediately, in-use storage times and conditions prior to use are the responsibility of the user and would normally not be longer than 24 hours at 2 to 8°C, unless additions have been made under controlled and validated aseptic conditions. If longer storage periods are required in exceptional circumstances, the company can be contacted as chemical and physical in-use stability data for 7 days at 2-8°C followed by 48 hours below 25°C are available for the products listed in the previous section.
Baxter Confidential