Donepezil 5 Mg Orodipersible Film
|
Sandoz Limited | |
|
Donepezil 5 mg and 10 mg Orodispersible Film |
PL 04416/1213-1214 |
Package leaflet: Information for the user
Donepezil 5 mg Orodispersible Films Donepezil 10 mg Orodispersible Films
donepezil hydrochloride
Read all of this leaflet carefully before you start taking this medicine because it contains important information for you.
• Keep this leaflet. You may need to read it again.
• If you have any further questions, ask your doctor or pharmacist
• This medicine has been prescribed for you only. Do not pass it on to others. It may harm them, even if their signs of illness are the same as yours.
• If you get any of the side effects talk to your doctor or pharmacist. This includes any possible side effects not listed in this leaflet. See section 4.
What In this leaflet:
1. What Donepezil is and what it is used for
2. What do you need to know before you take Donepezil
3. How to take Donepezil
4. Possible side effects
5. How to store Donepezil
6. Contents of the pack and other information
1. What DONEPEZIL is and what it is used for
Donepezil belongs to a group of medicines called acetylcholinesterase inhibitors. Donepezil increases the levels of a substance (acetylcholine) in the brain involved in memory function by slowing down the breakdown of acetylcholine.
Donepezil is used to treat the symptoms of dementia in people diagnosed as having mild to moderately severe Alzheimer’s Disease. The symptoms include increasing memory loss, confusion and behavioural changes. As a result, sufferers of Alzheimer’s disease find it more and more difficult to carry out their normal daily activities
Donepezil is for use in adult patients only.
2. What you need to know before you take DONEPEZIL Do not take Donepezil
if you are allergic to the active substance
• donepezil hydrochloride,
• “piperidine derivatives”, which are similar substances to donepezil or
• any of the other ingredients of this medicine (listed in section 6).
|
PIL.1213-14.007A |
28/10/201 |
V010:SPC & PIL update in line with the PRAC |
KP |
|
5 |
recommendations |
|
Sandoz Limited | |
|
Donepezil 5 mg and 10 mg Orodispersible Film |
PL 04416/1213-1214 |
Warnings and precautionsTreatment with Donepezil should only be started and supervised by a doctor with experience in diagnosing and treating Alzheimer’s dementia.
Talk to your doctor, pharmacist or nurse before taking Donepezil if you:.
• have ever had stomach or duodenal ulcers, or if you are taking non steroidal antiinflammatory drugs (NSAIDS)
• have ever had seizures (fits) or convulsions (donepezil may have the potential to cause seizures. You doctor will monitor your symptoms).
• have a heart condition (especially if you have an irregular heart beat, sick sinus syndrome, or other conditions that affect the rhythm of the heart), as donepezil may slow down your heart rate.
• have ever had extrapyramidal symptoms (uncontrolled movements of the body or face). Donepezil may induce or exacerbate extrapyramidal symptoms.
• have asthma or other long term lung disease
• have ever had liver problems or hepatitis
• have difficulty passing urine
• are going to have an operation that requires you to have a general anaestetic, as the dose of the medication used in anaesthesia may need to be adjusted.
Donepezil can be used in patients with kidney disease or mild to moderate liver disease. Tell your doctor first if you have kidney or liver disease. Patients with severe liver disease should not take Donepezil.
Children and adolescents
Donepezil is not recommended for children and adolescents below the age of 18 years.
Other medicines and DONEPEZIL
Tell your doctor or pharmacist if you are taking, have recently taken or might take any other medicines.This includes medicines that your doctor has not prescribed for you but which you have bought yourself from a chemist/pharmacist. It also applies to medicines you may take sometime in the future if you continue to take Donepezil. This is because these medicines may weaken or strengthen the effects of Donepezil
Especially tell your doctor if you are taking any of the following types of medicines:
• other Alzheimer’s disease medicines that act the same way as donepezil, e.g. galantamine or rivastigmine, and some medicines for diarrhoea, Parkinson’s disease or asthma
• pain killers or treatment for arthritis e.g. aspirin, non-steroidal anti-inflammatory (NSAID) drugs such as ibuprofen, or diclofenac sodium
• anticholinergics medicines, e.g. tolterodine
• antibiotics e.g. erythromycin, rifampicin
• anti-fungal medicine e.g. ketoconazole or itraconazole
• anti-depressants e.g. fluoxetine
• anticonvulsants e.g. phenytoin, carbamazepine
• medication for a heart condition e.g. quinidine, or high blood pressure, such as beta-blockers (propanolol and atenolol)
• muscle relaxants e.g. diazepam, succinylcholine
• general anaesthetic
• medicines obtained without a prescription e.g. herbal remedies
|
PIL.1213-14.007A |
28/10/201 |
V010:SPC & PIL update in line with the PRAC |
KP |
|
5 |
recommendations |
|
Sandoz Limited | |
|
Donepezil 5 mg and 10 mg Orodispersible Film |
PL 04416/1213-1214 |
Tell your doctor or pharmacist the name of your caregiver. Your caregiver will help you to take your medicine as it is prescribed.
Donepezil with food, drink and alcohol
Food will not influence the effect of Donepezil.
Donepezil must not be taken with alcohol because alcohol may change its effect.
Pregnancy and breast-feeding and fertility
Donepezil should not be used while breastfeeding.
If you are pregnant, or think you might be pregnant, ask your doctor for advice before taking any medicine.
Driving and using machines
Donepezil and the Alzheimer’s disease may impair your ability to drive or operate machines. Do not perform these activities unless your doctor has confirmed that this is safe.
This medicine can cause fatigue, dizziness and muscle cramps particularly during the beginning of therapy and dose increase. If affected you must not drive or operate machines.
Donepezil Orodispersible Films contain sucralose.
If you have been told by your doctor that you have an intolerance to some sugars, contact your doctor before taking this medicine.
Donepezil contains contains small amounts of ethanol (alcohol), less than 100mg per dose.
3. How to take Donepezil
Always take Donepezil exactly as your doctor has told you. Check with your doctor or pharmacist if you are not sure. The usual dose is:
The tablet strength you will take may change depending on the length of time you have been taking the medicine and on what your doctor will recommend.
Adults and elderly patients Donepezil 5 mg Orodispersible Films:
Usually, you will start by taking one orodispersible film (5 mg donepezil hydrochloride) every night. After one month, your doctor may tell you to take two orodispersible films (10 mg donepezil hydrochloride) every night. The maximum recommended dose is 10 mg each night
Donepezil 10 mg Orodispersible Films:
Usually, you will start by taking 5 mg donepezil hydrochloride every night. which can not be dosed using this medicinal product. Donepezil hydrochloride 5 mg orodispersible films are available to start treatment. After one month, your doctor may tell you to take one orodispersible film (10 mg donepezil hydrochloride) every night.
The maximum recommended dose is 10 mg each night.
|
PIL.1213-14.007A |
28/10/201 |
V010:SPC & PIL update in line with the PRAC |
KP |
|
5 |
recommendations |
|
Sandoz Limited | |
|
Donepezil 5 mg and 10 mg Orodispersible Film |
PL 04416/1213-1214 |
Do not alter the dose yourself without your doctor’s advice.
Patients with kidney dysfunction
You can take the usual dose as described above. No adjustment is required.
Patients with mild to moderate liver dysfunction Your doctor mey need to adjust your dose.
Patients with severe liver dysfunction
If you have severe liver problems, you should not take Donepezil (see section 2. “Warnings and precaution”). If you have an unexplained liver disease, your doctor may decide to stop your treatment with Donepezil completely.
Method of administration
Take your orodispersible films at night before you go to bed, independently from meals. Place them on your tongue where they dissolve rapidly so that they can be easily swallowed with or without water, according to your preference.
Instructions for use
Important: Do not handle the orodispersible films with wet hands!
a) Take the sachet, locate the arrow mark at one of the shorter sides and hold the sachet with this side facing up. The sachet is not sealed here.
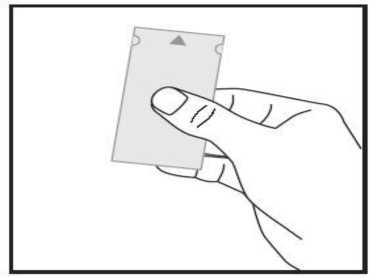
b) Gently peel both parts of the sachet apart at the arrow mark. Now you can hold each between your thumb and your index finger using one hand for each part.
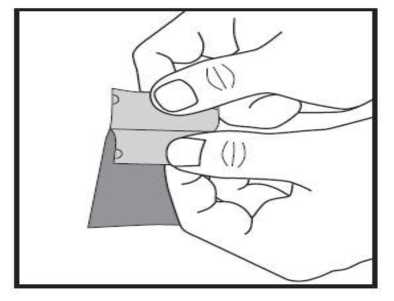
|
PIL.1213-14.007A |
28/10/201 |
V010:SPC & PIL update in line with the PRAC |
KP |
|
5 |
recommendations |
|
Sandoz Limited | |
|
Donepezil 5 mg and 10 mg Orodispersible Film |
PL 04416/1213-1214 |
c) Carefully tear both parts of the sachet in opposite directions until they are separated. The orodispersible film is now visible and placed on one of the separated sachet parts.
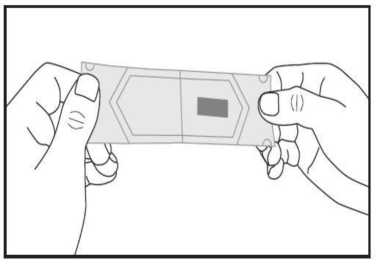
d) Take the orodispersible film with dry fingers out of the sachet and put it in your mouth directly on your tongue. It will dissolve rapidly, so that it can be easily swallowed. Water may be used if desired.
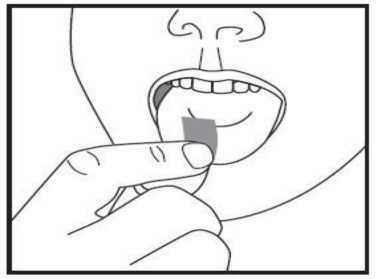
Duration of use
Your doctor or pharmacist will advise you on how long you should continue to take this medicine. You will need to see your doctor regularly to review your treatment and assess your symptoms.
If you take more Donepezil than you should
DO NOT take more than 10 mg donepezil hydrochloride each day.
Call your doctor immediately if you take more than you should. If you cannot contact your doctor, contact the local hospital Accident and Emergency department at once. Always take the orodispersible film, this leaflet and/or the carton with you to the hospital so that the doctor knows what has been taken.
Symptoms of overdosing include feeling and being sick, drooling, sweating, slow heart rate, low blood pressure (light-headedness or dizziness when standing), breathing problems, muscle weakness, losing consciousness and seizures (fits) or convulsions.
If you forget to take Donepezil
If you forget to take an orodispersible film, just take one orodispersible film the following day at the usual time. Do not take a double dose to make up for a forgotten dose.
If you forget to take your medicine for more than one week, call your doctor before taking any more medicine.
|
PIL.1213-14.007A |
28/10/201 |
V010:SPC & PIL update in line with the PRAC |
KP |
|
5 |
recommendations |
|
Sandoz Limited | |
|
Donepezil 5 mg and 10 mg Orodispersible Film |
PL 04416/1213-1214 |
If you stop taking Donepezil
Do not stop taking this medicine even if you are feeling well, unless instructed by your doctor. Positive treatment results may gradually diminish if treatment is stopped.
4. Possible side effects
Like all medicines, this medicine can cause side effects, although not everybody gets them. Serious side effects:
You must tell your doctor immediately if you notice these serious side effects mentioned. You may need urgent medical treatment.
• liver damage e.g. hepatitis. The symptoms of hepatitis are feeling or being sick, loss of appetite, feeling generally unwell, fever, itching, yellowing of the skin and eyes, and dark coloured urine (may affect up to 1 in 1,000 people)
• stomach or duodenal ulcers. The symptoms of ulcers are stomach pain and discomfort (indigestion) felt between the navel and the breast bone (may affect up to 1 in 100 people)
• bleeding in the stomach or intestines. This may cause you to pass black tar like stools or visible blood from the rectum (may affect up to 1 in 100 people)
• seizures (fits) or convulsions (may affect up to 1 in 100 people)
• fever with muscle stiffness, sweating or a lowered level of consciousness, These are the symptoms of a disorder called “Neuroleptic Malignant Syndrome” (may affect up to 1 in 10,000 people).
•muscle weakness, tenderness or pain and particularly, if at the same time, you feel unwell, have a high temperature or have dark urine. They may be caused by an abnormal muscle breakdown which can be life threatening and lead to kidney problems (a condition called rhabdomyolysis).
Very common, may affect more than 1 in 10 people:
• diarrhoea
• nausea (feeling sick)
• headache
Common, may affect 1 to 10 users in 100:
• muscle cramp
• tiredness
• difficulty in sleeping (insomnia)
• the common cold
• loss of appetite
• hallucinations (seeing or hearing things that are not really there) aggressive behavior, agitation, which have resolved on dose reduction or discontinuing of treatment
• unusual dreams and nightmares
fainting
• dizziness, sleeplessness
• stomach feeling uncomfortable, vomiting
|
PIL.1213-14.007A |
28/10/201 |
V010:SPC & PIL update in line with the PRAC |
KP |
|
5 |
recommendations |
|
Sandoz Limited | |
|
Donepezil 5 mg and 10 mg Orodispersible Film |
PL 04416/1213-1214 |
• rash
• itching
• passing urine uncontrollably
• pain, fatigue
• accidents (patients may be more prone to falls and accidental injury)
Uncommon, may affect up to 1 in 100 people:
• slow heart beat
• minor increase of the muscle enzyme creatine kinase in blood tests Rare, may affect up to 1 in 1,000 people:
• shaking, stiffness or uncontrollable movement, especially of the face and tongue but also of the limbs
• cardiac disturbances
Reporting of side effects
If you get any side effects, talk to your <doctor or> <,> <pharmacist> <or nurse>. This includes any possible side effects not listed in this leaflet. You can also report side effects directly via Yellow Card Scheme: www.mhra.gov.uk/yellowcard.
By reporting side effects you can help provide more information on the safety of this medicine.
5. How to storeDONEPEZIL
Keep this medicine out of the sight and reachof children.
Do not use this medicine after the expiry date which is stated on the outer carton and on the sachet. The expiry date refers to the last day of that month.
This medicinal product does not require any special storage condition.
Do not throw away any medicines via wastewater or household waste. Ask your pharmacist how to throw away medicines no longer used. These measures will help to protect the environment.
6. Contents of the pack and other information
What Donepezil contains
The active substance is donepezil hydrochloride.
Each Donepezil 5 mg Orodispersible Film contains 5 mg of donepezil hydrochloride.
Each Donepezil 10 mg Orodispersible Film contains 10 mg of donepezil hydrochloride.
The other ingredients are:
Donepezil 5 mg Orodispersible Films:
Glycerol, hypromellose, levomenthol, maltodextrin, microcrystalline cellulose, peppermint flavour, sucralose, titanium dioxide (E 171), ethanol (alcohol)
|
PIL.1213-14.007A |
28/10/201 |
V010:SPC & PIL update in line with the PRAC |
KP |
|
5 |
recommendations |
|
Sandoz Limited | |
|
Donepezil 5 mg and 10 mg Orodispersible Film |
PL 04416/1213-1214 |
Donepezil 10 mg Orodispersible Films:
Glycerol, hypromellose, levomenthol, maltodextrin, microcrystalline cellulose, peppermint flavour, sucralose, titanium dioxide (E 171), ethanol (alcohol)
What Donepezil looks like and contents of the pack
Donepezil 5 mg Orodispersible Films are white to slightly yellow, rectangular (3 cm2) and flexible.
Donepezil 10 mg Orodispersible Films are white to slightly yellow, rectangular (6 cm ) and flexible.
Donepezil 5 mg and 10 mg Orodispersible Films are available in cartons containing 7, 10, 14, 21, 28, 30, 49, 50, 56, 60, 90, 98, 100 or 120 orodispersible films each individually enclosed in a sealed sachet.
Some packs also contain a plastic-box for temporary storage of a few sachets which is convenient to carry with you compared to the carton.
Not all pack sizes may be marketed.
Marketing Authorisation Holder and Manufacturer
Marketing Authorisation Holder
Sandoz Limited
Frimley Business Park
Frimley
Camberley
Surrey
GU16 7SR
United Kingdom
Manufacturer
Hexal AG, Industriestresse 25, 83607 Holzkirchen, Germany This leaflet was last revised in 10/2015.
|
PIL.1213-14.007A |
28/10/201 |
V010:SPC & PIL update in line with the PRAC |
KP |
|
5 |
recommendations |