Katya 30/75 Coated Tablets
SUMMARY OF PRODUCT CHARACTERISTICS
1 NAME OF THE MEDICINAL PRODUCT
Katya 30/75 coated tablets
2. QUALITATIVE AND QUANTITATIVE COMPOSITION
Active ingredients:
Katya 30/75 coated tablets: Each tablet contains 30 micrograms ethinylestradiol and 75 micrograms gestodene
Excipients:
Katya 30/75 coated tablets contain 38 mg lactose monohydrate and 20 mg sucrose For the full list of excipients, see section 6.1.
3 PHARMACEUTICAL FORM
Coated tablet.
White, round, biconvex sugar coated tablets, both sides are without imprinting.
4.1 Therapeutic indications
Oral contraception.
The decision to prescribe Katya 30/75 coated tablets should take into consideration the individual woman’s current risk factors, particularly those for venous thromboembolism (VTE), and how the risk of VTE with Katya 30/75 coated tablets compares with other Combined Hormonal Contraceptives, CHCs (see sections 4.3 and 4.4).
4.2 Posology and method of administration
How to take Katya 30/75 coated tablets
The tablets should be taken in the order indicated on the package, every day at approximately the same time. One tablet per day should be taken for 21 days. Each subsequent pack should be started after a 7-day tablet-free interval during which time a withdrawal bleeding will occur. This bleeding usually starts on the 2nd or 3rd day after taking the last tablet, and may not stop until the next pack is started.
How to start taking Katya 30/75 coated tablets
If no preceding hormonal contraceptive use in the past month
Taking of the tablets should begin on the first day of the woman’s natural cycle (i.e. on the first day of the woman’s menstrual bleeding). One may begin taking the pills on day 2-5, but in these cases, it is recommended that a barrier method also be used for the first 7 days on which pills are taken during the first cycle.
When replacing another contraceptive pill of the combination type
The woman should start taking Katya 30/75 coated tablets on the next day after taking the last active tablet in her previous package of contraceptive pills - but no later than the day after the usual tablet-free or placebo-tablet period of her previous contraceptive pill.
When changing from progestogen-only preparations (progestogen-only pills, injection, implant, or from a progestogen-releasing intrauterine system (IUS))
The woman may change from progestogen-only pills (POPs) on any day. The first tablet should be taken on the day after any tablet of the POP package. When changing from an implant or the IUS, Katya 30/75 coated tablets should be started on the day the implant is removed. When changing from injections, Katya 30/75 coated tablets should be started when the next injection is due to be given. In all these cases, the woman is advised to also use a barrier method for the first 7 days of taking the pills.
After an abortion in the first trimester
The woman may start taking the pills immediately. If she does so, no further contraceptive steps need be taken.
After delivery or abortion in the second trimester For breastfeeding women - see section 4.6.
The woman should be advised to begin taking the tablets on day 21- 28 after delivery in non-lactating women or after abortion in the second trimester. If she starts later, she should be advised to also use a barrier method during the first 7 days of taking the pills. If she has already had intercourse, the possibility of pregnancy should be excluded before she begins taking the pills, or she should wait for her first menstruation.
Missed tablets
Missing a tablet for less than 12 hours does not diminish the contraceptive protection. The woman should take the tablet as soon as she remembers, and continue taking the rest of the tablets as usual.
Missing a tablet for more than 12 hours can diminish the contraceptive protection. The two following rules may be helpful in dealing with missed tablets.
Taking of the tablets should never be discontinued for longer than 7 days.
It takes 7 days of uninterrupted ingestion of the tablets to achieve sufficient suppression of the hypothalamus-pituitary-ovarian axis.
Thus, the following advice can be given in daily practice:
Week 1
The user should take the last missed tablet as soon as she remembers, even if this means that she needs to take 2 tablets at the same time. From then on, she should continue to take the tablets at the usual time. At the same time, she should use a barrier method, i.e. a condom, for the next 7 days. If she had intercourse during the past 7 days, she should consider the possibility that she might be pregnant. The more tablets have been missed, and the closer this happened to the monthly tablet-free period, the higher the risk of pregnancy.
Week 2
The user should take the last missed tablet as soon as she remembers, even if this means that she needs to take 2 tablets at the same time. From then on, she should continue to take the tablets at the usual time. If the tablets have been taken correctly for the 7 days prior to the missed tablet, it is not necessary to take any additional contraceptive precautions. If this is not the case, however, or if more than 1 tablet has been missed, the woman should use a barrier method, i.e. a condom for the next 7 days.
Week 3
The risk of reduced protection is imminent because of the approaching tablet-free period. The reduced contraceptive protection can be prevented, however, by adjusting the intake of the tablets. By adhering to either of the following two options, it is, therefore, not necessary to take any additional contraceptive precautions, provided that the tablets have been taken correctly for the 7 days prior to the missed tablet. If this is not the case, the woman should be advised to follow the first of the two choices, and at the same time use a barrier method, i.e. a condom for the next 7 days.
The user should take the last missed tablet as soon as she remembers even if this means that she needs to take 2 tablets at the same time. From then on, she should continue to take the tablets at the usual time. She begins the next pack immediately after she took the last tablet from the current package; that means no pause between packages. The user will probably not get her menstruation before the end of the second package, but she may experience spotting or withdrawal bleeding on the days when she takes the tablets.
The woman can also be advised to stop taking tablets from the current package. In that case, she should have a tablet-free period for up to 7 days, including the days when she missed the tablets, and subsequently continue with the next pack.
If the woman missed the tablets, and subsequently did not get her menstruation in the first normal tablet-free period, she should consider the possibility that she may be pregnant.
What to do in case of vomiting/diarrhoea
If vomiting occurs within 3-4 hours after tablet taking, absorption may not be complete. In this case, the advice concerning missed tablets, described above should be followed. Unless Diarrhoea is extremely severe it does not affect the absorption of combined oral contraceptives and therefore use of additional contraception is not necessary. If severe diarrhoea continues for 2 or more days, the procedures for missed pills should be followed. If the woman does not want to change her usual tablet intake, she should take an extra tablet(s) from another blister pack.
How to advance or delay menstruation
To delay menstruation, the woman should continue with another pack of Katya 30/75 coated tablets without a tablet-free period. Menstruation can be delayed as long as is desired up to the end of the second package, but no longer. While menstruation is being delayed, the woman may experience withdrawal bleeding or spotting. Regular intake of Katya 30/75 coated tablets should be resumed after the normal tablet-free period of 7 days.
To move menstruation to another day of the week than the woman is used to with her current tablet schedule, she can be advised to shorten the next tablet-free period by as many days as she wishes. The shorter the interval, the higher the risk that she will not get her menstruation and will have breakthrough bleeding or spotting while she is taking the next pack (just as when menstruation is being delayed).
4.3 Contraindications
Combined hormonal contraceptives (CHCs) should not be used in the following conditions. If such a condition should occur for the first time during use of CHCs, the use must be discontinued immediately:
• Presence or risk of venous thromboembolism (VTE)
o Venous thromboembolism - current VTE (on anticoagulants) or history of (e.g. deep venous thrombosis [DVT] or pulmonary embolism [PE])
o Known hereditary or acquired predisposition for venous
thromboembolism, such as APC- resistance, (including Factor V Leiden), antithrombin-III-deficiency, protein C deficiency, protein S deficiency
o Major surgery with prolonged immobilisation (see section 4.4)
o A high risk of venous thromboembolism due to the presence of multiple risk factors (see section 4.4)
• Presence or risk of arterial thromboembolism (ATE)
o Arterial thromboembolism - current arterial thromboembolism, history of arterial thromboembolism (e.g. myocardial infarction) or prodromal condition (e.g. angina pectoris)
o Cerebrovascular disease - current stroke, history of stroke or prodromal condition (e.g. transient ischaemic attack, TIA)
o Known hereditary or acquired predisposition for arterial thromboembolism, such as hyperhomocysteinaemia and antiphospholipid-antibodies (anticardiolipin-antibodies, lupus anticoagulant).
o History of migraine with focal neurological symptoms.
o A high risk of arterial thromboembolism due to multiple risk factors (see section 4.4) or to the presence of one serious risk factor such as:
• diabetes mellitus with vascular symptoms
• severe hypertension
• severe dyslipoproteinaemia
• Known or suspected sex-steroid influenced malignancies (e.g. of the genital organs or the breast)
• Presence or history of severe hepatic disorders, as long as liver function tests are not normalised
• Presence or history of benign or malignant liver tumours
• Undiagnosed vaginal bleeding
• Hypersensitivity to the active substances or to any of the excipients listed in section 6.1
4.4 Special warnings and precautions for use
If any of the conditions or risk factors mentioned below is present, the suitability of Katya 30/75 coated tablets should be discussed with the woman.
In the event of aggravation, or first appearance of any of these conditions or risk factors, the woman should be advised to contact her doctor to determine whether the use of Katya 30/75 coated tablets should be discontinued.
1. Circulatory disorders
Risk of venous thromboembolism (VTE)
The use of any combined hormonal contraceptive (CHC) increases the risk of venous thromboembolism (VTE) compared with no use. Products that contain levonorgestrel, norgestimate or norethisterone are associated with the lowest risk of VTE. Other products such as Katya 30/75 coated tablets may have up to twice this level of risk. The decision to use any product other than one with the lowest VTE risk should be taken only after a discussion with the woman to ensure she understands the risk of VTE with Katya 30/75 coated tablets, how her current risk factors influence this risk, and that her VTE risk is highest in the first ever year of use. There is also some evidence that the risk is increased when a CHC is re-started after a break in use of 4 weeks or more.
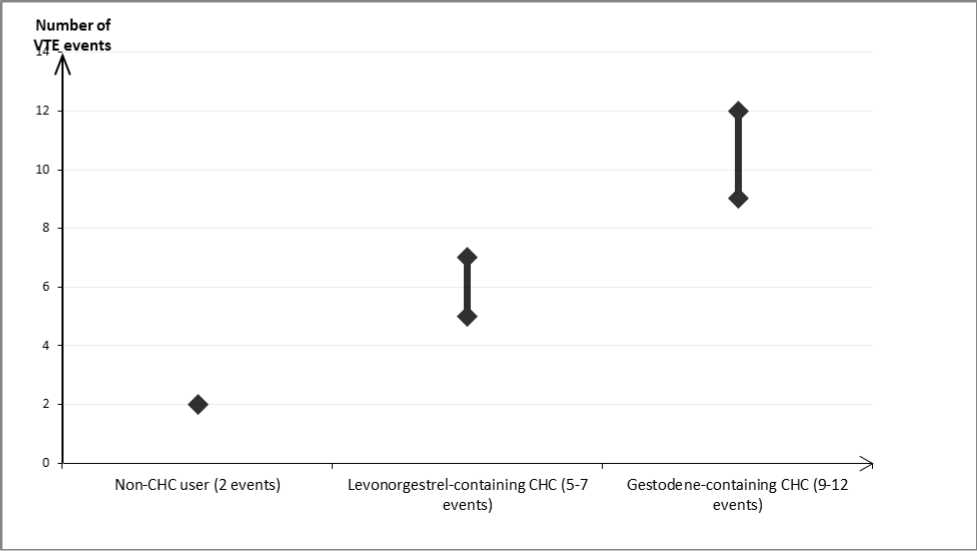
In women who do not use a CHC and are not pregnant about 2 out of 10,000 will develop a VTE over the period of one year. However, in any individual woman the risk may be far higher, depending on her underlying risk factors (see below).
It is estimated1 2 that out of 10,000 women who use a CHC containing gestodene between 9 and 12 women will develop a VTE in one year; this compares with about 6 in women who use a levonorgestrel-containing CHC.
In both cases, the number of VTEs per year is fewer than the number expected during pregnancy or in the postpartum period.
VTE may be fatal in 1-2% of cases.
Number of VTE events per 10,000 women in one year
Extremely rarely, thrombosis has been reported to occur in CHC users in other blood vessels, e.g. hepatic, mesenteric, renal or retinal veins and arteries.
Risk factors for VTE
The risk for venous thromboembolic complications in CHC users may increase substantially in a woman with additional risk factors, particularly if there are multiple risk factors (see table).
Katya 30/75 coated tablets are contraindicated if a woman has multiple risk factors that put her at high risk of venous thrombosis (see section 4.3). If a woman has more than one risk factor, it is possible that the increase in risk is greater than the sum of the individual factors - in this case her total risk of VTE should be considered. If the balance of benefits and risks is considered to be negative a CHC should not be prescribed (see section 4.3).
Table: Risk factors for VTE
|
Risk factor |
Comment |
|
Obesity (body mass index over 30 kg/m2) |
Risk increases substantially as BMI rises. Particularly important to consider if other risk factors also present. |
|
Prolonged immobilisation , major surgery, any surgery to the legs or |
In these situations it is advisable to discontinue use of the patch/pill/ring (in the case of elective surgery |
|
pelvis, neurosurgery, or major trauma Note: temporary immobilisation including air travel >4 hours can also be a risk factor for VTE, particularly |
least four weeks in advance) and not resume until two weeks after complete remobilisation. Another method of contraception should be used to avoid unintentional pregnancy. Antithrombotic treatment should be considered if Katya 30/75 coated tablets have not been discontinued in advance. |
|
Positive family history (venous thromboembolism ever in a sibling or parent especially at a relatively early age e.g. before 50). |
If a hereditary predisposition is suspected, the woman should be referred to a specialist for advice before deciding about any CHC use |
|
Other medical conditions associated with VTE |
Cancer, systemic lupus erythematosus, haemolytic uraemic syndrome, chronic inflammatory bowel disease (Crohn’s disease or ulcerative colitis) and ---ll di----- |
|
Increasing age |
Particularly above 35 years |
There is no consensus about the possible role of varicose veins and superficial thrombophlebitis in the onset or progression of venous thrombosis.
The increased risk of thromboembolism in pregnancy, and particularly the 6 week period of the puerperium, must be considered (for information on “Pregnancy and lactation” see section 4.6).
Symptoms of VTE (deep vein thrombosis and pulmonary embolism)
In the event of symptoms women should be advised to seek urgent medical attention and to inform the healthcare professional that she is taking a CHC.
Symptoms of deep vein thrombosis (DVT) can include:
- unilateral swelling of the leg and/or foot or along a vein in the leg;
- pain or tenderness in the leg which may be felt only when standing or walking,
- increased warmth in the affected leg; red or discoloured skin on the leg.
Symptoms of pulmonary embolism (PE) can include:
- sudden onset of unexplained shortness of breath or rapid breathing;
- sudden coughing which may be associated with haemoptysis;
- sharp chest pain;
- severe light headedness or dizziness;
- rapid or irregular heartbeat.
Some of these symptoms (e.g. “shortness of breath”, “coughing”) are non-specific and might be misinterpreted as more common or less severe events (e.g. respiratory tract infections).
Other signs of vascular occlusion can include: sudden pain, swelling and slight blue discoloration of an extremity.
If the occlusion occurs in the eye symptoms can range from painless blurring of vision which can progress to loss of vision. Sometimes loss of vision can occur almost immediately.
Risk of arterial thromboembolism (ATE)
Epidemiological studies have associated the use of CHCs with an increased risk for arterial thromboembolism (myocardial infarction) or for cerebrovascular accident (e.g. transient ischaemic attack, stroke). Arterial thromboembolic events may be fatal.
Risk factors for ATE
The risk of arterial thromboembolic complications or of a cerebrovascular accident in CHC users increases in women with risk factors (see table). Katya 30/75 coated tablets are contraindicated if a woman has one serious or multiple risk factors for ATE that puts her at high risk of arterial thrombosis (see section
4.3). If a woman has more than one risk factor, it is possible that the increase in risk is greater than the sum of the individual factors - in this case her total risk should be considered. If the balance of benefits and risks is considered to be negative a CHC should not be prescribed (see section 4.3).
Table: Risk factors for ATE
|
Risk factor |
Comment |
|
Increasing age |
Particularly above 35 years |
|
Smoking |
Women should be advised not to smoke if they wish to use a CHC. Women over 35 who continue to smoke should be strongly advised to use a different method of contraception. |
|
Hypertension | |
|
Obesity (body mass index over 30 kg/m2) |
Risk increases substantially as BMI increases. Particularly important in women with additional risk factors |
|
Positive family history (arterial thromboembolism ever in a sibling or parent especially at relatively early age e.g. below 50). |
If a hereditary predisposition is suspected, the woman should be referred to a specialist for advice before deciding about any CHC use |
|
Migraine |
An increase in frequency or severity of migraine during CHC use (which may be prodromal of a cerebrovascular event) may be a reason for immediate discontinuation |
|
Other medical conditions associated with adverse vascular |
Diabetes mellitus, hyperhomocysteinaemia, valvular heart disease and atrial fibrillation, |
dyslipoproteinaemia and systemic lupus erythematosus.
Symptoms of ATE
In the event of symptoms women should be advised to seek urgent medical attention and to inform the healthcare professional that she is taking a CHCs.
Symptoms of a cerebrovascular accident can include:
- sudden numbness or weakness of the face, arm or leg, especially on one side of the body;
- sudden trouble walking, dizziness, loss of balance or coordination;
- sudden confusion, trouble speaking or understanding;
- sudden trouble seeing in one or both eyes;
- sudden, severe or prolonged headache with no known cause;
- loss of consciousness or fainting with or without seizure.
Temporary symptoms suggest the event is a transient ischaemic attack (TIA).
Symptoms of myocardial infarction (MI) can include:
- pain, discomfort, pressure, heaviness, sensation of squeezing or fullness in the chest, arm, or below the breastbone;
- discomfort radiating to the back, jaw, throat, arm, stomach;
- feeling of being full, having indigestion or choking;
- sweating, nausea, vomiting or dizziness;
- extreme weakness, anxiety, or shortness of breath;
- rapid or irregular heartbeats.
2. Tumours:
Cervical cancer
In some epidemiological studies an increased risk of cervical cancer has been reported in long term users of COCs, but it is still not clear to which extent this finding may be influenced by impacts of sexual behaviour and other factors, such as human papilloma virus (HPV).
Breast cancer
A meta-analysis from 54 epidemiological studies reported that there is a slightly increased relative risk (RR=1.24) of having breast cancer diagnosed in woman who are currently using COCs. The excess risk gradually disappears during the course of the 10 years after cessation of COC use. Because breast cancer is rare in women below 40 years of age, the excess number of breast cancer diagnoses in current and recent users of COC is small in relation to the overall risk of breast cancer.
These studies do not provide evidence for causation. The observed pattern of increased risk may be due to an earlier diagnosis of breast cancer in COC users, the biological effects of COCs or a combination of both. The breast cancers diagnosed in users tend to be less advanced clinically than the cancers diagnosed in non-users.
Liver tumours
Benign and malignant liver tumours have been reported in users of COCs. These tumours have, in isolated cases, lead to life threatening, intra-abdominal haemorrhage. A liver tumour must be taken into consideration as a differential diagnosis when severe pain occurs in the upper abdomen, if there is hepatomegaly, or if there are signs of intra-abdominal haemorrhage in women taking COCs.
3. Other conditions
Women with hypertriglyceridaemia, or a family history thereof, may be at increased risk of pancreatitis when taking COCs.
In the case of acute or chronic impairment of liver function, the use of Katya 30/75 coated tablets should be stopped until liver function tests have returned to normal. Steroid hormones may be poorly metabolised in patients with impaired liver function.
Even though slight increases in blood pressure have been reported in many women taking COCs, clinically important increases in blood pressure are rare. If persistent clinical hypertension develops during COC use, intake should be discontinued and the hypertension treated. Use of COCs may be resumed, if appropriate, when normotensive values are reached with antihypertensive therapy.
It has been reported that the following conditions may occur, or worsen both during pregnancy and during use of COCs, but the evidence of a relationship is inconclusive: Jaundice and/or pruritus in connection with cholestasis; development of gallstones; porphyria; systemic lupus erythematosus; haemolytic uraemic syndrome; Sydenham's chorea; herpes gestationis; loss of hearing due to otosclerosis.
COCs may have an influence on peripheral insulin resistance and glucose tolerance. Therefore, diabetics should be closely monitored during COC use.
Katya 30/75 coated tablets contain lactose and sucrose. Patients with rare hereditary problems of galactose intolerance, the Lapp lactase deficiency or glucose-galactose malabsorption or with rare hereditary problems of fructose intolerance should not take this medicinal product.
Worsening of endogenous depression, of epilepsy (see section 4.5 interactions), of Crohn’s disease and of ulcerative colitis has been reported during COC use.
Chloasma may occur, in particular in women with a medical history of chloasma gravidarum. Women with a tendency to chloasma should avoid exposure to sunlight or ultraviolet radiation while taking COCs.
Herbal preparations containing St John’s wort (Hypericum perforatum) should not be used while taking Katya 30/75 coated tablets due to the risk of decreased plasma concentrations and reduced clinical effects of Katya 30/75 coated tablets (see section 4.5).
Reduced efficacy
The efficacy of oral contraceptives may be reduced in the case of missed tablets, severe diarrhoea or vomiting (see section 4.2) or concomitant use of other medicinal products (see section 4.5).
Reduced cycle control
With all combined oral contraceptives, irregular bleeding (spotting or break through bleeding) may occur, especially during the first months. Hence, the evaluation of any irregular bleeding should be considered after a period of adaptation of approximately 3 cycles.
If bleeding irregularities persist COCs with a higher hormonal content may need to be considered. If bleeding irregularities occur after previously regular cycles, then nonhormonal causes should be considered, and adequate diagnostic measures are indicated to exclude malignancy or pregnancy.
Occasionally withdrawal bleeding during the tablet-free interval may not occur at all. If the tablets have been taken according to the instructions described in section 4.2, it is unlikely that the woman is pregnant. However, if the tablets have not been taken according to the instructions, before the first absent withdrawal bleeding, or if two withdrawal bleedings are overdue, pregnancy should be excluded before COC use is continued.
Medical examination/consultation
Prior to the initiation or reinstitution of Katya 30/75 coated tablets a complete medical history (including family history) should be taken and pregnancy must be ruled out. Blood pressure should be measured and a physical examination should be performed, guided by the contra-indications (see section 4.3) and warnings (see section 4.4). It is important to draw a woman’s attention to the information on venous and arterial thrombosis, including the risk of Katya 30/75 coated tablets compared with other CHCs, the symptoms of VTE and ATE, the known risk factors and what to do in the event of a suspected thrombosis.
The woman should also be instructed to carefully read the user leaflet and to adhere to the advice given. The frequency and nature of examinations should be based on established practice guidelines and be adapted to the individual woman.
Women should be advised that oral contraceptives do not protect against HIV infections (AIDS) and other sexually transmitted diseases.
4.5 Interaction with other medicinal products and other forms of interaction
Drug interaction resulting in elevated clearance of sex hormones may cause breakthrough bleeding and contraceptive failure. This has been established with hydantoins, barbiturates, primidone, carbamazepine and rifampicin; oxcarbazepine, topiramate, griseofulvin, felbamate and ritonavir are also suspected. The mechanism of this interaction seems to rest upon the liver enzyme-inducing properties of these medicinal products. Maximal enzyme induction is generally not visible before 2-3 weeks after the start of the treatment, but it may persist for at least 4 weeks after the end of treatment.
Contraceptive failure has also been reported with antibiotics, such as ampicillin and tetracyclins. The mechanism of this action has not been elucidated.
Women undergoing short-term treatment with any of the above mentioned groups, or individual medicinal products, should temporarily use a barrier method along with the contraceptive pills; that means during the time when both this medicinal product and the contraceptive pills are taken, as well as 7 days after the medicinal product is discontinued. Women treated with rifampicin should use a barrier method along with the contraceptive pills during the time when they are treated with rifampicin as well as for 28 days after they stop taking rifampicin. If the intake of another concomitant medicinal product stretches beyond the number of tablets in the contraceptive pill pack, the woman should start the next pack without observing the normal tablet-free period.
For long-term users of medicinal products that induce liver enzymes, use of other contraceptive measures should be advised.
Patients being treated with Katya 30/75 coated tablets should not simultaneously use products/alternative medicinal products containing Hypericum perforatum (St. John’s wort), as this can lead to loss of contraceptive effect. Withdrawal bleeding and undesired pregnancy have been reported.
Hypericum perforatum (St. John’s wort) increases, by enzyme induction, the amount of enzymes that metabolise medicinal products. The effect of the enzyme induction may last for at least 1-2 weeks after the end of treatment with Hypericum.
COC effects on other drugs: oral contraceptives may interfere with the metabolism of other drugs. Accordingly, plasma and tissue concentrations may either increase (e.g. ciclosporin) or decrease (lamotrigine).
Laboratory tests
The use of contraceptive steroids can influence the results of certain laboratory tests, including the biochemical parameters of liver, thyroid, adrenal, and kidney function; plasma levels of (transport) proteins, such as corticosteroid-binding globulin and lipid/lipoprotein fractions; the parameters of carbohydrate metabolism, and the parameter of coagulation and fibrinolysis. The changes usually remain within the normal test ranges.
4.6 Pregnancy and lactation
Katya 30/75 coated tablets are not indicated during pregnancy. If pregnancy occurs during medication with Katya 30/75 coated tablets, the preparation should be withdrawn immediately.
Extensive epidemiological studies have revealed neither an increased risk of birth defects in children born to women who used COCs prior to pregnancy, nor a teratogenic effect when COCs were taken inadvertently during pregnancy.
Contraceptive steroids can influence breastfeeding, as they can lower the amount and change the composition of breast milk. Small amounts of contraceptive steroids and/or their metabolites can be excreted in the milk. The use of contraceptive steroids should, therefore, generally not be advised to a breastfeeding mother before her child is completely weaned.
The increased risk of VTE during the postpartum period should be considered when re-starting Katya 30/75 coated tablets (see section 4.2 and 4.4).
4.7 Effects on ability to drive and use machines
Katya 30/75 coated tablets have no or negligible influence on the ability to drive and use machines.
4.8 Undesirable effects
The most commonly reported adverse reactions are irregular bleeding, nausea, weight increase, breast tenderness and headache. They occur usually at the beginning of therapy and are transient.
Description of selected adverse reactions
An increased risk of arterial and venous thrombotic and thrombo-embolic events, including myocardial infarction, stroke, transient ischemic attacks, venous thrombosis and pulmonary embolism has been observed in women using CHCs, which are discussed in more detail in section 4.4.
|
Organ system class |
Common (>1/100 to < 1/10) |
Uncommon (>1/1,000 to < 1/100) |
Rare (>1/10,000 to < 1,000) |
Very rare (>1/10,000) |
|
Nervous system disorders |
Headache Nervousness |
Chorea | ||
|
Eye disorders |
Ocular irritation when wearing contact lenses Visual disturbances | |||
|
Ear and labyrinth disorders |
Otosclerosis | |||
|
Gastrointestinal disorders |
Nausea |
Vomiting |
Cholelithiasis |
Pancreatitis |
|
Skin and Subcutaneous tissue disorders |
Acne |
Chloasma | ||
|
Metabolism and nutrition disorders |
Hyperlipidaemia | |||
|
Vascular disorders |
Migraine |
Hypertension |
Venous thromboembolism Arterial thromboembolism | |
|
General disorders and administration site conditions |
Weight increase Fluid retention | |||
|
Immune system disorders |
Lupus erythematosus | |||
|
Reproduction system and breast disorders |
Irregular bleeding Amenorrhoea Hypomenorrhoea Breast tenderness |
Changes in vaginal secretion | ||
|
Psychiatric disorders |
Changes in libido Depression Irritability |
The following serious adverse events have been reported in women using COCs, see sections 4.3 and 4.4.
o Venous thromboembolism, i.e. deep leg or pelvic venous thrombosis and pulmonary embolism. o Arterial thromboembolic disorders o Liver tumours
o Skin and subcutaneous disorders: chloasma.
The frequency of diagnosis of breast cancer is very slightly increased among COC-users. As breast cancer is rare in women under 40 years of age the increase is small in relation to the overall risk of breast cancer. Causation with COC use is unknown. For further information, see sections 4.3 and 4.4.
Reporting of suspected adverse reactions
Reporting suspected adverse reactions after authorisation of the medicinal product is important. It allows continued monitoring of the benefit/risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions via the Yellow Card Scheme. Website: www.mhra.gov.uk/yellowcard
4.9 Overdose
No serious harmful effects have been reported with overdoses. Symptoms that can arise in connection with
an overdose are: Nausea, vomiting, and vaginal bleeding. There is no antidote, and further treatment should be symptomatic.
5 PHARMACOLOGICAL PROPERTIES
5.1 Pharmacodynamic properties
Pharmacotherapeutic group: hormonal contraceptives for systemic use ATC code: G03 AA10
The contraceptive effect of contraceptive pills rests on the interaction of various factors, the most important of which are inhibition of ovulation and changes in the endometrium. Along with protecting against pregnancy, COCs have several positive properties which, next to the negative properties (see 4.8 Warnings, Undesirable effects), can be useful in deciding on the method of birth control. The menstrual cycle is more regular and the menstruation is often less painful, and bleeding is lighter. The latter may result in a decrease in the occurrence of iron deficiency.
Gestodene
Absorption
Gestodene, when taken orally, is absorbed quickly and completely. Following a single dose the maximum serum concentration of 4 ng/ml is reached in approximately one hour. Bioavailability is approximately 99%.
Distribution
Gestodene is bound to serum albumin and to sex hormone binding globulin (SHBG). Only 1-2% of the total amount of gestodene in serum is found as free steroid, while 5070% is specifically bound to SHBG. The ethinylestradiol-induced increase in SHBG influences the distribution of serum proteins, which causes an increase of the SHBG-bound fraction, and a decrease of the albumin-bound fraction. The apparent distribution volume of gestodene is 0.7 l/kg.
Metabolism
Gestodene is metabolised completely via the known pathways of steroid metabolism. The metabolic clearance rate from serum is 0.8 ml/min/kg. No interaction occurs when gestodene is taken together with ethinylestradiol.
Elimination
Serum level of gestodene is reduced at 2 rates. The last rate is characterised by a half-life of 12 - 15 hours.
Gestodene is not excreted unchanged. Its metabolites are excreted in urine and in bile at a ratio of 6:4.
The half-life of metabolite excretion is approximately 1 day.
Steady-state
Pharmacokinetics of gestodene is influenced by the levels of SHBG in serum, which increase to triple values with ethinylestradiol. Upon daily intake, the level of gestodene in serum increases till approximately four times the single dose value, and reaches steady-state within the second half of the treatment cycle.
Ethinylestradiol
Absorption
Ethinylestradiol, taken orally, is absorbed quickly and completely. Maximal serum concentration of about 80 pg/ml is reached within 1-2 hours. Complete bioavailability, resulting from pre-systemic conjugation and first-pass metabolism, is approximately 60%.
Distribution
During lactation, 0.02% of the daily maternal dose passes into breast milk. Ethinylestradiol is predominantly bound non-specifically to albumin (approx. 98.5%), and causes increase in serum concentration of SHBG. The apparent distribution volume is found to be approximately 5 l/kg.
Metabolism
Ethinylestradiol undergoes pre-systemic conjugation both in the mucosa of the small intestine, and in the liver. Ethinylestradiol is primarily metabolised by aromatic hydroxylation, but many different hydroxylated and methylated metabolites are formed,
and found as free metabolites and as glucuronide and sulphate conjugates. The metabolic clearance rate is approximately 5 ml/min/kg.
Elimination
Serum level of ethinylestradiol is reduced at 2 rates, the last one with a half-life of 24 hours. Unchanged Ethinylestradiol is not excreted, but its metabolites are excreted in urine and in bile at a ratio of 4:6. The half-life of metabolite excretion is approximately 1 day.
Steady-state
Steady-state occurs after 3-4 days, and the serum levels of ethinylestradiol are 30-40% higher than at single dose.
5.3 Preclinical safety data
Ethinylestradiol and gestodene are not genotoxic. Carcinogenicity studies with ethinylestradiol alone or in combination with various progestogens do not indicate any particular carcinogenic hazard to women when used as indicated for contraception. However it should be noted that sex hormones can advance the growth of certain hormone-dependent tissues and tumours.
Reproductive toxicity studies on fertility, development of the fetus or reproductive ability with ethinylestradiol alone or in combination with progestogens revealed no undesirable effects for humans when used as recommended.
6 PHARMACEUTICAL PARTICULARS
6.1 List of excipients
Tablet core: Magnesium stearate Povidone K-25 Maize starch Lactose monohydrate
Tablet coating: Povidone K-90 Macrogol 6000 Talc
Calcium carbonate Sucrose
Wax montan glycol
Not applicable.
6.3 Shelf life
3 years
6.4 Special precautions for storage
Do not store above 30 °C.
6.5 Nature and contents of container
Blister: PVC/aluminium.
Pack sizes: 1 x 21 tablets; 3 x 21 tablets, 6 x 21 tablets Not all pack sizes may be marketed.
6.6 Special precautions for disposal
No special requirements.
7 MARKETING AUTHORISATION HOLDER
Stragen UK Ltd Castle Court 41 London Road Reigate Surrey RH2 9RJ
8
9
MARKETING AUTHORISATION NUMBER(S)
PL 21844/0011
DATE OF FIRST AUTHORISATION/RENEWAL OF THE AUTHORISATION
20/04/2012
10
DATE OF REVISION OF THE TEXT
07/08/2014
These incidences were estimated from the totality of the epidemiological study data, using relative risks for the different products compared with levonorgestrel-containing CHCs.
Mid-point of range of 5-7 per 10,000 WY, based on a relative risk for CHCs containing levonorgestrel versus nonuse of approximately 2.3 to 3.6