Ondansetron 4 Mg Orodispersible Tablets
Out of date information, search anotherPACKAGE LEAFLET: INFORMATION FOR THE USER

PHARMACEUTICALS
Ondansetron 4 mg orodispersible tablets Ondansetron 8 mg orodispersible tablets
Ondansetron
Read all of this leaflet carefully before you start taking this medicine.
In this leaflet:
1. WHAT ONDANSETRON ORODISPERSIBLE TABLETS ARE AND WHAT IT IS USED FOR
2. BEFORE YOU TAKE ONDANSETRON ORODISPERSIBLE TABLETS
3. HOW TO TAKE ONDANSETRON ORODISPERSIBLE TABLETS
• Keep this leaflet. You may need to read it again.
• If you have any further questions, ask your doctor or pharmacist.
• This medicine has been prescribed for you. Do not pass it on to others. It may harm them, even if their symptoms are the same as yours.
• If any of the side effects gets serious, or if you notice any side effects not listed in this leaflet, please tell your doctor or pharmacist.
1. What Ondansetron orodispersible tablets is
and what it is used for
2. Before you take Ondansetron orodispersible tablets
3. How to take Ondansetron orodispersible tablets
4. Possible side effects
5. How to store Ondansetron orodispersible tablets
6. Further information
Ondansetron orodispersible tablet is a fast dissolving tablet when placed on top of the tongue. Ondansetron orodispersible tablets contains ondansetron which belongs to a group of medicines that stop you feeling sick or being sick, so called anti-emetics.
Ondansetron orodispersible tablets may
> stop the effects of nausea and vomiting caused by cytotoxic chemotherapy in children and adults
> prevent post-operative nausea and vomiting in children and adults
> stop the effects of nausea and vomiting caused by radiotherapy in adults
If you are not sure why they have been prescribed for you, ask your doctor.
Do not take Ondansetron orodispersible tablets
- If you are allergic (hypersensitive) to ondansetron, or any of other ingredients in Ondansetron orodispersible tablet or any similar medicines.
Important information about some of the ingredients of Ondansetron orodispersible tablets
Ondansetron orodispersible tablets contain aspartame, a source of phenylalanine. This may be harmful for people with phenylketonuria. Ondansetron orodispersible tablets contain glucose and sorbitol. If you have been told by your doctor that you have intolerance to some sugars, contact your doctor before taking this medicinal product.
Ondansetron orodispersible tablets contain sulphur dioxide which may rarely cause severe hypersensitivity reactions and bronchospasm.
Always take Ondansetron orodispersible tablets exactly as your doctor has told you. You should check with your doctor or pharmacist if you are not sure.
After starting to take your medicine Ondansetron orodispersible tablets should start to work within one or two hours of taking a dose. If you are sick within one hour of taking a dose, then take the same dose again - otherwise do not take more Ondansetron orodispersible tablets than the label says. If you continue to feel sick then tell your doctor.
Treatment and prevention of nausea and vomiting in connection with chemotherapy or radiotherapy
Adults:
8 mg 1 to 2 hours before chemotherapy or radiotherapy followed by 8 mg 12 hours for up to 5 days. Your doctor may decide to give the first dose as an injection.
Elderly:
The same dose as for adults.
Children (age 2 years and up) and adolescents under the age of 18 years:
The dose is individual and depends on the size/surface of the child. Ondansetron Bluefish should not be used for children with a total body surface of less than 0.6 m2.
Children aged over 6 months and adolescents
• The usual dose for a child is up to 4 mg twice a day
• This can be given up to 5 days
Treatment and prevention of post-operative nausea and vomiting
Take special care with Ondansetron orodis-persible tablets
Before treatment with Ondansetron orodispers-ible tablets, tell your doctor
- If you are pregnant or likey to become pregnant soon.
- If you are breast-feeding.
- If you have liver disease.
- If you have a blockage in your gut or if you suffer from severe constipation
- If you have been told by your doctor that you have an intolerance to some sugars, contact your doctor before taking this medicinal product
- If you have phenylketonuria because Ondansetron orodispersible tablets contain a source of phenylalanine. May be harmful for people with phenylketonuria.
- If it is children below the age of 2 years or with a body surface of less than 0.6 m2.
Taking other medicines
Please tell your Doctor or pharmacist if you are taking or have recently taken any other medicines like phenytoin, carbamazepine, rifampi-cin, tramadol and including medicines obtained without a prescription.
Pregnancy
Ask your doctor or pharmacist for advice before taking any medicine.
Breast-feeding
Ondansetron probably passes into the mother's milk. Therefore it is recommended that mothers taking Ondansetron orodispersible tablets do not breast-feed.
Adults, prevention and treatment:
16 mg one hour prior to anesthesia or alternatively, 8 mg administered one hour prior to anesthesia followed by an additional 8 mg after 8 and 16 hours. Your doctor may choose to give you the medicine as injections.
Elderly, prevention and treatment:
There is limited experience with the use of ondansetron to elderly patients. Ondansetron is however tolerated well by patients above 65 years in chemotherapy (please refer to section above).
Patients with moderate or severe liver disease: the total daily dose should not be more than 8 mg.
Patients with poor sparteine/debrisoquine metabolism: No alteration of daily dosage or frequency of dosing is required. Do not remove Ondansetron orodispersible tablets from its blister or pierce the foil until you are ready to take it.
The tablet(s) must be taken as follows:
In order to prevent the breakage of tablets, it is important not to push the tablet out of its recess (Figure A).
The tablets in each strip are separated from each other by perforations. Detach one tablet recess along the perforation (Figure 1). The covering foil should be removed carefully. Start with the corner that is marked with an arrow (Figures 2 and 3).
Driving and using machines
Ondansetron orodispersible tablets has no influence on the ability to drive and use machines.
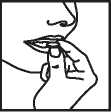
The tablet must be taken from the container with dry hands and placed on the tongue (Figure 4). The tablet will disintegrate, after which it can be swallowed with water.
Strawberry flavour contains sulphur dioxide (E220) which may rarely cause severe hypersensitivity reactions and bronchospasm.
Fig. A. Fig. 1. Fig. 2.
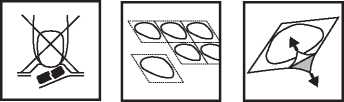
If any of the side effects gets serious, or if you notice any side effects not listed in this leaflet, please tell your doctor or pharmacist.
Fig. 3.

Fig. 4.
5. HOW TO STORE ONDANSETRON ORODISPERSIBLE TABLETS
If you take more Ondansetron orodispers-ible tablets than you should
It is important to stick to the dose on the label. Taking more than this could make you ill. If too much medicine is taken, don't delay; ask your doctor what to do or contact your nearest hospital casualty department.
If you forget to take Ondansetron orodis-persible tablets
Do not take a double dose to make up for a forgotten dose.
If you miss a dose and feel sick or vomit, take an Ondansetron orodispersible tablet as soon as possibe and then carry on as before.
If you miss a dose but do not feel sick take the next dose as on the label.
Keep out of the reach and sight of children. Store in the original package in order to protect from light.
Store below 30°C.
Do not use Ondansetron orodispersible tablets after the expiry date which is stated on the carton or foil after EXP.
Medicines should not be used if signs of deterioration, such as discolouration or broken tablets, are observed.
Medicines should not be disposed of via wastewater or household waste. Ask your pharmacist how to dispose of medicines no longer required. These measures will help to protect the environment.
6. FURTHER INFORMATION
4. POSSIBLE SIDE EFFECTS
Like all medicines, Ondansetron orodispersible tablets can cause side effects, although not everybody gets them.
What Ondansetron orodispersible tablets contains
The active substance is ondansetron. Each orodispersible tablet contains 4 mg or 8 mg ondansetron.
Some side effects are more likely to occur than others. The possible side effects of ondansetron are listed below and can be divided as:
- Very common (affecting not more than 1 in 10 persons)
- Common (affecting less than 1 in 10, but more than 1 in 100 persons)
- Uncommon (affecting less than 1 in 100, but more than 1 in 1,000 persons)
- Rare (affecting less than 1 in 1,000 persons, but more than 1 in 10,000 persons)
- Very rare (affecting less than 1 in 10,000 persons, including isolated reports)
A few people can be allergic to some medicines; if any of the following side effects come on soon after taking Ondansetron orodispersible tablets, stop taking them and tell your doctor immediately:
• Sudden wheeziness and chest pain or chest tightness
• Swelling of eyelids, face, lips, mouth or tongue
• Skin rash - red spots or hives (skin lumps) anywhere on the body
• Collapse
Very common (affecting not more than 1 in 10 persons)
- Headache.
The other ingredients are: Aspartame (E951), Crospovidone type B, Magnesium stearate (E572), Microcrystalline cellulose (E460), Pharmaburst TM C1, containing mannitol (E421), sorbitol (E420), crospovidone (type A) and colloidal silicon dioxide); Strawberrry flavouring, (containing dextrose, maltodextrin, gum araibc E414 2.3% and sulphur dioxide E220); Sodium stearyl fumarate.
What Ondansetron orodispersible tablets looks like and contents of the pack
Orodispersible tablets.
Ondansetron 4 mg and 8 mg orodispersible tablets
White, flat, round, bevel-edged tablet.
Ondansetron orodispersible tablets is provided in packs of 6x1, 10x1, 14x1, 20x1, 30x1, 50x1, 60x1, 100x1 tablets in aluminium/OPA/PVC unit-dose blister pack.
Not all pack sizes may be marketed.
Marketing Authorisation Holder and Manufacturer
Bluefish Pharmaceuticals AB Torsgatan 11 SE-111 23 Stockholm Sweden.
Name of the Member State
Denmark
Finland
Germany
Netherlands
Norway
Poland
Spain
Sweden
United Kingdom
Common (affecting less than 1 in 10, but more than 1 in 100 persons)
- Constipation.
- Sensation of warmth or flushing.
Uncommon (affecting less than 1 in 100, but more than 1 in 1,000 persons)
- Arrhythmias, chest pain with or without ST segment depression, bradycardia.
- Asymptomatic increases in liver function tests.
- Seizures, movement disorders including extrapyramidal reactions such as dystonic reactions, oculogyric crisis and dyskinesia have been observed without definitive evidence of persistent clinical sequelae.
- Hiccups.
- Hypotension.
Rare (affecting less than 1 in 1,000 persons, but more than 1 in 10,000 persons)
- Immediate hypersensitivity reactions sometimes severe, including anaphylaxis.
- Dizziness during i.v. administration, which in most cases is prevented or resolved by lengthening the infusion period.
- Transient visual disturbances (eg. blurred vision) during i.v. administration.
Very rare (affecting less than 1 in 10,000 persons, including isolated reports):
Transient blindness predominantly during intravenous administration
The majority of the blindness cases reported resolved within 20 minutes. Most patients had received chemotherapeutic agents, which included cisplatin. Some cases of transient blindness were reported as cortical in origin.
The package leaflet for the blind and partially sighted in other formats is made available on request.
This medicinal product is authorized in the Member States of the EEA under the following names:
Name of the medicinal product
Ondansetron Bluefish 4 mg/8 mg smeltetabletter Ondansetron Bluefish 4 mg/8 mg tabletti, suussa hajoava
Ondansetron Bluefish 4 mg/8 mg munsonderfal-lande tabletter Ondansetron Bluefish 4 mg/8 mg schmelz-tabletten
Ondansetron Bluefish 4 mg/8 mg smelttabletten Ondansetron Bluefish 4 mg/8 mg smeltetabletter Ondansetron Bluefish Ondansetron Bluefish 4 mg/8 mg comprimidos bucodispersibles EFG Ondansetron Bluefish 4 mg/ 8 mg munsonderfal-lande tabletter Ondansetron 4 mg/ 8 mg orodispersible tablets
This leaflet was last approved in 11/2009.