Qlaira
Out of date information, search anotherPACKAGE LEAFLET: INFORMATION FOR THE USER
Qlaira®
(estradiol valerate/dienogest)
Read all of this leaflet carefully before you start taking this medicine because it contains important information for you.
• Keep this leaflet, you may need to read it again.
• If you have any further questions, ask your doctor or pharmacist.
• This medicine has been prescribed for you only, do not pass it on to others. It may harm them.
• If you get any side effects, talk to your doctor or pharmacist. This includes any possible side effects not listed in this leaflet.
What is in this leaflet:
1. What Qlaira is and what it is used for
2. What you need to know before you take Qlaira When not to take Qlaira
Warnings and precautions Qlaira and venous/arterial blood clots Qlaira and cancer Bleeding between periods
What to do if no bleeding occurs on day 26 or the following day(s)
Other medicines and Qlaira
Qlaira with food and drink
Laboratory tests
Pregnancy and breast-feeding
Driving and using machines
Qlaira contains lactose
3. How to take Qlaira Preparation of the wallet
When can you start with the first wallet?
If you take more Qlaira than you should If you forget to take Qlaira Use in children
What to do if you vomit or have severe diarrhoea If you want to stop taking Qlaira
4. Possible side effects
5. How to store Qlaira
6. Contents of the pack and other information
1. What Qlaira is and what it is used for
► Qlaira is a contraceptive pill and is used to prevent pregnancy.
► Qlaira is used for the treatment of heavy menstrual bleeding (not caused by any disease of the womb) in women who wish to use oral contraception.
► Each coloured, active tablet contains a small amount of female hormones, either estradiol valerate, or estradiol valerate combined with dienogest.
► The 2 white tablets contain no active substances and are called inactive tablets.
► Contraceptive pills that contain two hormones are called “combined pills”.
2. What you need to know before you take Qlaira General notes
Before you can begin taking Qlaira, your doctor will ask you some questions about your personal health history and that of your close relatives. The doctor will also measure your blood pressure and, depending upon your personal situation, may also carry out some other tests.
In this leaflet, several situations are described where you should stop using Qlaira, or where the reliability of Qlaira may be decreased. In such situations you should either not have sex or you should take extra non-hormonal contraceptive precautions, e.g. use a condom or another barrier method. Do not use rhythm or temperature methods. These methods can be unreliable because Qlaira alters the monthly changes of body temperature and cervical mucus.
Qlaira, like other hormonal contraceptives, does not protect against HIV infection (AIDS) or any other sexually transmitted disease.
When not to take Qlaira
Do not take Qlaira:
► if you have (or have ever had) a blood clot in a blood vessel of the leg (thrombosis), of the lung (pulmonary embolism) or other organs
► if you have (or have ever had) a heart attack or stroke
► if you have (or have ever had) a disease that can be an indicator (i) of a future heart attack (for example, angina pectoris which causes severe chest pain) or (ii) of a stroke (for example, a minor stroke with no residual effects)
► if you have (or have ever had) a certain kind of migraine (with so-called focal neurological symptoms)
► if you have a disease that may increase the risk of a clot in the arteries. This applies to the following diseases:
- diabetes with damaged blood vessels
- very high blood pressure - a very high level of blood fats (cholesterol or triglycerides)
► if you have a disturbance of blood clotting (for example, protein C deficiency)
► if you have (or have ever had) inflammation of the pancreas
(pancreatitis)
► if you have (or have ever had) liver disease and your liver function is still not normal
► if you have (or have ever had) a tumour of the liver
► if you have (or have ever had) cancer or suspected cancer of the breast or genital organs
► if you have any unexplained bleeding from the vagina
► if you are allergic (hypersensitive) to estradiol valerate or dienogest, or any of the other ingredients of this medicine (listed in section 6). This may cause itching, rash or swelling
Warnings and precautions
In some situations you need to take special care while taking Qlaira or any other combined pill, and your doctor may need to examine you regularly. Consult your doctor before starting to use Qlaira if any of the following conditions apply to you or if any of them develop or worsen while you are taking Qlaira:
► if a close relative has or has ever had breast cancer
► if you have a disease of the liver or gall bladder
► if you have jaundice
► if you have diabetes
► if you have depression
► if you have Crohn’s disease or inflammatory bowel disease (ulcerative colitis)
► if you have a blood disease called haemolytic uraemic syndrome (HUS) that causes kidney damage
► if you have sickle cell disease
► if you have epilepsy (see “Other medicines and Qlaira”')
► if you have a disease of the immune system called systemic lupus erythematosus (SLE)
► if you have a disease that first appeared during pregnancy or earlier use of sex hormones, for example, hearing loss, porphyria (a disease of the blood), gestational herpes (skin rash with blisters during pregnancy), Sydenham’s chorea (a nerve disease causing sudden movements of the body)
► if you have (or have ever had) golden brown pigment patches so-called “pregnancy patches” especially on the face (Chloasma). If this is the case, avoid direct exposure to sunlight or ultraviolet light
► if you have hereditary angioedema. Consult your doctor immediately if you experience symptoms of angioedema such as swollen face, tongue and/or throat and/or difficulty swallowing or hives, together with difficulty breathing. Products containing oestrogens may induce or worsen symptoms of angioedema
► if you have cardiac or renal insufficiency
Talk to your doctor before taking Qlaira.
Qlaira and venous/arterial blood clots
The following warnings and precautions are derived from studies with combined pills containing ethinylestradiol. It is not yet known whether or not they apply to Qlaira:
The use of any combined pill (including Qlaira) increases a woman’s risk of developing a venous blood clot (venous thrombosis) compared with women who do not take any contraceptive pill.
Very occasionally, blood clots may cause serious permanent disabilities or may even be fatal.
The main locations of blood clots are, for example, the veins of the legs (venous thrombosis), the arteries of the heart (heart attack), the lungs (pulmonary embolism) or the brain (stroke).
The risk of venous blood clots in users of combined pills increases:
► with increasing age
► if you are overweight
► if one of your close relatives had a blood clot in the leg, lung (pulmonary embolism), or other organ at a young age
► if you need surgery, if you have had a serious accident or if you have any prolonged period of immobilisation. It is important to tell your doctor that you are using Qlaira as the treatment may have to be stopped.
Your doctor will tell you when to start Qlaira again. This is usually about two weeks after you have recovered.
The use of combined pills has been linked to an increased risk of an arterial blood clot (arterial thrombosis), for example, in the blood vessels of the heart (heart attack) or the brain (stroke).
The risk of arterial blood clots in users of combined pills increases:
► with increasing age
► if you smoke. You are strongly advised to stop smoking when you use the pill, especially if you are over 35 years old
► if you have a high level of blood cholesterol or triglycerides
► if you are overweight
► if one of your close relatives ever had a heart attack or stroke at a young age
► if you have high blood pressure
► if you suffer from migraine
► if you have a problem with your heart (valve disorder, a disturbance of the heart rhythm)
Stop taking Qlaira and contact your doctor immediately if you notice possible signs of a blood clot or a stroke such as:
► severe pain and/or swelling in one of your legs
► sudden severe pain in the chest which may spread to the left arm
► sudden breathlessness
► sudden cough with no obvious cause
► any unusual, severe or long-lasting headache, or worsening of migraines
► partial or complete blindness, or double vision
► difficulty in speaking, or inability to speak
► giddiness or fainting
► weakness, strange feeling, or numbness in any part of your body
► sudden severe stomachache
Directly after giving birth, women are at an increased risk of blood clots so you should ask your doctor how soon after delivery you can start taking a combined pill.
Qlaira and cancer
Breast cancer has been observed slightly more often in women using combined pills, but it is not known whether this is caused by the treatment itself. For example, it may be that more tumours are detected in women on combined pills because they are examined by their doctor more often. The risk of breast tumours becomes gradually less after stopping the combination hormonal contraceptives. It is important to regularly check your breasts and you should contact your doctor if you feel any lump.
In rare cases, benign liver tumours, and in even fewer cases malignant liver tumours have been reported in contraceptive pill users. In isolated cases, these tumours have led to lifethreatening internal bleeding. Contact your doctor if you have unusually severe abdominal pain.
Some studies suggest that long-term use of the pill increases a woman's risk of developing cervical cancer. However, it is not clear to what extent sexual behaviour or other factors such as Human Papilloma Virus (HPV) increases this risk.
Bleeding between periods
During the first few months of taking Qlaira, you may have unexpected bleeding. Usually bleeding starts on day 26, the day you take the second dark red tablet, or the following day(s). The information provided by women in the diaries they kept during a clinical study of Qlaira shows that it is not unusual to experience unexpected bleeding in a given cycle (10-18% of users). If unexpected bleeding occurs more than 3 months in a row, or if it begins after some months, your doctor will have to investigate the cause.
What to do if no bleeding occurs on day 26 or the following day(s)
The information provided by women in the diaries they kept during a clinical study of Qlaira shows that it is not unusual to miss your regular bleeding after day 26 (observed in about 15% of cycles).
If you have taken all the tablets correctly, have not had any vomiting or severe diarrhoea and you have not taken any other medicines, it is highly unlikely that you are pregnant.
If the expected bleeding does not happen twice in a row or you have taken the tablets incorrectly, you may be pregnant. Contact your doctor immediately. Do not start the next wallet until you are sure that you are not pregnant.
Other medicines and Qlaira
Always tell your doctor which medicines or herbal products you are already using. Also tell any other doctor or dentist who prescribes another medicine (or the pharmacist from whom you got the medicine) that you take Qlaira. They can tell you if you need to take additional contraceptive precautions (for example condoms) and if so, for how long.
Some medicines can make Qlaira less effective in preventing pregnancy, or can cause unexpected bleeding. These include:
► medicines used for the treatment of:
- epilepsy (e.g. primidone, phenytoin, barbiturates, carbamazepine, oxcarbazepine, topiramate, felbamate)
- tuberculosis (e.g. rifampicin)
- HIV infections (e.g. ritonavir, nevirapine) other infections (antibiotics such as penicillins, tetracyclines, griseofulvin).
► the herbal remedy St. John's wort
Some medicines can increase the levels of the active substances of
Qlaira in the blood. Inform your doctor if you are using:
► anti-fungal medicines containing ketoconazole
► antibiotics containing erythromycin
► Qlaira may influence the effect of other medicines, e.g.
- medicines containing ciclosporin
- the anti-epileptic lamotrigine (this could lead to an increased frequency of seizures).
Ask your doctor or pharmacist for advice before taking any medicine. Your doctor or pharmacist may advise on extra protective measures while you are taking other medication together with Qlaira.
Qlaira with food and drink
Qlaira may be taken with or without food, if necessary with a small amount of water.
Laboratory tests
If you need a blood test or other laboratory tests tell your doctor or the laboratory staff that you are taking the pill because oral contraceptives can affect the results of some tests.
Pregnancy and breast-feeding
Do not take Qlaira if you are pregnant. If you become pregnant while taking Qlaira, stop taking it immediately and contact your doctor. If you want to become pregnant, you can stop taking Qlaira at any time (see also “If you stop taking Qlaira’").
In general you should not take Qlaira while you are breast-feeding.
If you want to take the pill while you are breast-feeding you should contact your doctor.
Ask your doctor or pharmacist for advice before taking any medicine when you are pregnant or breast-feeding.
Driving and using machines
There is nothing to suggest that the use of Qlaira affects driving or use of machines.
Qlaira contains lactose
If you have been told by your doctor that you have an intolerance to some sugars, contact your doctor before taking Qlaira.
3. How to take Qlaira
Each wallet contains 26 coloured active tablets and 2 white inactive tablets.
Take one tablet of Qlaira every day, if necessary with a small amount of water. You may take the tablets with or without food, but you should take the tablets at around the same time every day.
Preparation of the wallet
To help you keep track, there are 7 weekday sticker strips marked with the 7 days of the week.
Choose the weekday sticker strip that starts with the day you begin taking the tablets. For example, if you start on a Wednesday, use the weekday sticker strip that starts with “WED”.
Stick the weekday sticker strip along the top of the Qlaira wallet where it reads “Place weekday sticker strip here”, so that the first day is above the tablet marked “1”.
There is now a day shown above every tablet and you can see whether you have taken a pill on a particular day. Follow the direction of the arrow on the wallet until all 28 tablets have been taken.
Usually, so-called withdrawal bleeding starts when you are taking the second dark red tablet or the white tablets and may not have finished before you start the next wallet. Some women still experience bleeding after taking the first tablets of the new wallet.
Start the following wallet without a gap, in other words the day after you have finished your current wallet, even if the bleeding has not stopped. This means that you should start your following wallet on the same day of the week as the current wallet and that the withdrawal bleed should occur on the same weekdays each month.
If you use Qlaira in this manner, you are protected against pregnancy even during the 2 days when you take inactive tablets.
When can you start with the first wallet?
► If you have not used a contraceptive with hormones during the previous month.
Start taking Qlaira on the first day of the cycle (that is, the first day of your period).
► Changing from another combined hormonal contraceptive pill, or combined contraceptive vaginal ring or patch.
Start Qlaira the day after taking the last active tablet (the last tablet containing the active substances) of your previous pill. When changing from a combined contraceptive vaginal ring or patch, start using Qlaira on the day of removal or, follow the advice of your doctor.
*
Missed more than 1 coloured pill
Forgot to start a new wallet
Day
1-9
Day
10-17
Only
missed 1
pill (more than 12 hours late)
► Changing from a progestogen-only-method (progestogen-only pill, injection, implant or a progestogen-releasing ‘IUS', intrauterine system). You may switch from the progestogen-only pill any day (from an implant or the IUS on the day of its removal, from an injectable when the next injection would be due) but in all of these cases you must use extra protective measures (for example, a condom) during the first 9 days of Qlaira use.
► After a miscarriage.
Follow the advice of your doctor.
► After having a baby.
You can start Qlaira between 21 and 28 days after having a baby. If you start later than day 28, use a barrier method (for example, a condom) during the first 9 days of Qlaira use.
If, after having a baby, you have had sex before re-starting Qlaira, be sure that you are not pregnant or wait until the next menstrual period.
If you want to start Qlaira after having a baby and are breastfeeding, read the section on “Pregnancy and breast-feeding”.
Ask your doctor what to do if you are not sure when to start.
If you take more Qlaira than you should
There are no reports of serious harmful effects of taking too many Qlaira tablets.
If you take several active tablets at once, you may feel sick or throw up. Young girls may have bleeding from the vagina.
If you have taken too many Qlaira tablets, or you discover that a child has taken some, ask your doctor or pharmacist for advice.
If you forget to take Qlaira
Inactive tablets: If you miss a white tablet (2 tablets at the end of the wallet), you do not need to take it later because they do not contain any active substances. However, it is important that you discard the missed white tablet(s) to make sure that the number of days when you take inactive tablets is not increased as this would increase the risk of pregnancy. Continue with the next tablet at the usual time.
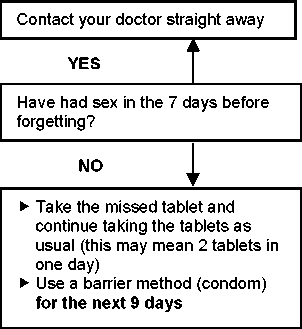
Active tablets: Depending on the day of the cycle on which one active tablet has been missed, you may need to take additional contraceptive precautions, for example a barrier method such as a condom. Take the tablets according to the following principles. See also the ‘missed pill chart’ for details.
► If you are less than 12 hours late when taking a tablet, the protection against pregnancy is not reduced. Take the tablet as soon as you remember and then continue taking the tablets again at the usual time.
► If you are more than 12 hours late taking a tablet, the protection against pregnancy may be reduced. Depending on the day of the cycle on which one tablet has been missed, use additional contraceptive precautions e.g. a barrier method such as a condom. See also the ‘missed pill chart’ for details.
► More than one tablet forgotten in this wallet Contact your doctor.
Do not take more than 2 active tablets on a given day.
If you have forgotten to start a new wallet, or if you have missed one or more tablets during days 3 - 9 of your wallet, there is a risk that you are already pregnant (if you had sex in the 7 days before forgetting the tablet). In that case, contact your doctor.
The more tablets you have forgotten (especially those on days 3 - 24) and the closer they are to the inactive tablet phase, the greater the risk that the protection from pregnancy is reduced.
See also the ‘missed pill chart’ for details.
If you have forgotten any of the active tablets in a wallet, and you have no bleeding at the end of a wallet, you may be pregnant. Contact your doctor before you start the next wallet.
>
>
|
► Do not take the missed tablet | ||
|
Day |
—► |
► Start immediately with the next |
|
18-24 |
wallet | |
|
► Use a barrier method (condom) | ||
|
for the next 9 days | ||
|
► Take the missed tablet and | ||
|
Day |
—► |
continue taking the tablets as |
|
25-26 |
usual (this may mean 2 tablets in | |
|
one day) | ||
|
► No additional contraception | ||
|
necessary | ||
|
► Discard the missed tablet and | ||
|
Day |
—► |
continue taking the tablets as |
|
27-28 |
usual | |
|
► No additional contraception | ||
|
necessary | ||
Use in children
No data available in adolescents below 18 years.
What to do if you vomit or have severe diarrhoea
If you throw up within 3-4 hours of taking an active tablet or you have severe diarrhoea, there is a risk that the active substances in the pill are not fully absorbed by your body.
The situation is almost the same as forgetting a tablet. After throwing up or having diarrhoea, take the next tablet as soon as possible. If possible, take it within 12 hours of when you normally take your pill. If this is not possible or 12 hours have passed, you should follow the advice given under “If you forget to take Qlaira”. If you do not want to change your normal tablet taking pattern take the corresponding tablet from another wallet.
If you stop taking Qlaira
You can stop taking Qlaira at any time. If you do not want to become pregnant, ask your doctor for advice about other reliable methods of birth control. If you want to become pregnant, stop taking Qlaira and wait for a menstrual period before starting to try to become pregnant. You will be able to calculate the expected delivery date more easily.
If you have any further questions on the use of this product, ask your doctor or pharmacist.
4. Possible side effects
Like all medicines, Qlaira can cause side effects, although not everybody gets them.
Serious side effects
Serious reactions associated with the use of the pill, as well as the related symptoms, are described in the following sections: “Qlaira and venous/arterial blood clots” and “Qlaira and cancer”.
Please read these sections carefully and consult your doctor at once where appropriate.
Other possible side effects
The following side effects have been linked with the use of Qlaira:
Common side effects (between 1 and 10 in every 100 users may be affected):
► headache
► abdominal pain, nausea
► acne
► no periods, breast discomfort, painful periods, irregular bleeding (heavy irregular bleeding)
POM
► weight gain
Uncommon side effects (between 1 and 10 in every 1,000 users may be
affected):
► fungal infections, fungal infection of the vulva and vagina, vaginal infection
► increased appetite
► depression, depressed mood, emotional disorder, problems sleeping, decreased interest in sex, mental disorder, mood swings
► dizziness, migraine
► hot flush, high blood pressure
► diarrhoea, vomiting
► increased liver enzymes
► hair loss, excessive sweating (hyperhidrosis), itching, rash
► muscle cramps
► swollen breasts, lumps in the breast, abnormal cell growth on the neck of the womb (cervical dysplasia), dysfunctional genital bleeding, pain with intercourse, fibrocystic breast disease, heavy periods, menstrual disorders, ovarian cyst, pelvic pain, premenstrual syndrome, growth in the uterus, contractions of the uterus, uterine/vaginal bleeding incl. spotting, vaginal discharge, vulvovaginal dryness
► fatigue, irritability, swelling of parts of your body, e.g. ankles (oedema)
► weight loss, blood pressure changes.
Rare side effects (between 1 and 10 in every 10,000 users may be
affected):
► Candida infection, oral herpes, pelvic inflammatory disease, a vessel disease of the eye resembling a fungal infection (presumed ocular histoplasmosis syndrome), a fungal infection of the skin (tinea versicolor), urinary tract infection, bacterial inflammation of the vagina
► fluid retention, increase in certain blood fats (triglycerides)
► aggression, anxiety, feelings of unhappiness, increased interest in sex, nervousness, night mare, restlessness, problems sleeping, stress
► reduced attention, “pins and needles”, giddiness
► contact lens intolerance, dry eye, eye swelling
► heart attack (myocardial infarction), palpitations
► bleeding in a varicose vein, low blood pressure, inflammation of superficial veins, painful veins
► constipation, dry mouth, indigestion, heartburn
► liver nodules (focal nodular hyperplasia), chronic inflammation of gallbladder
► allergic skin reactions, golden brown pigment patches (chloasma) and other pigmentation disorders, male pattern hair growth, excessive hair growth, skin conditions such as dermatitis and neurodermatitis, dandruff and oily skin (seborrhoea) and other skin disorders.
► back pain, pain in jaw, sensation of heaviness
► urinary tract pain
► abnormal withdrawal bleeding, benign breast nodules, breast cancer in early stage, breast cysts, breast discharge, polyp on the neck of the womb, reddening on the neck of the womb, bleeding during intercourse, spontaneous milk flow, genital discharge, lighter periods, delayed periods, rupture of an ovarian cyst, vaginal odour, burning sensation in the vulva and vagina, vulvovaginal discomfort
► swollen lymph nodes
► asthma, difficulty in breathing, nose bleeding
► chest pain, tiredness and feeling generally unwell, fever
► abnormal smear from the neck of the womb
Further information (taken from the diaries women kept during a Qlaira clinical trial) on the possible side effects “irregular bleeding (heavy irregular bleeding)” and “no periods” is given in the sections “Bleeding between periods” and “What to do if no bleeding occurs on day 26 or the following davsfsl”.
In addition to the above mentioned side effects, the skin disorders erythema nodosum, erythema multiforme, as well as breast discharge and hypersensitivity have occurred in women using combined pills containing ethinylestradiol. Although these symptoms were not reported during the clinical studies performed with Qlaira, the possibility that they might also occur under treatment cannot be ruled out.
If any of the side effects gets serious or if you notice any side effects not listed in this leaflet, please tell your doctor or pharmacist.
Reporting of side effects
If you get any side effects, talk to your doctor, pharmacist or nurse. This includes any possible side effects not listed in this leaflet. You can also report side effects directly via the Yellow Card Scheme at: www.mhra.aov.uk/vellowcard.
By reporting side effects, you can help provide more information on the safety of this medicine.
5. HOW TO STORE QLAIRA
Keep out of the sight and reach of children.
This medicinal product does not require any special storage conditions.
Do not use Qlaira after the expiry date which is stated on the wallet after EXP. The expiry date refers to the last day of that month.
Medicines should not be disposed of via wastewater or household waste. Ask your pharmacist how to dispose of medicines no longer required. These measures will help to protect the environment.
If the tablets show any signs of discolouration or deterioration consult your pharmacist for advice.
6. Contents of the pack and other information What Qlaira contains
The active substances are estradiol valerate, or estradiol valerate combined with dienogest.
Each wallet (28 film-coated tablets) of Qlaira contains 26 active tablets in
4 different colours in rows 1,2, 3 and 4, as well as 2 white inactive tablets in row 4.
Composition of the coloured tablets containing one or two active substances:
2 dark yellow tablets each containing 3 mg estradiol valerate
5 medium red tablets each containing 2 mg estradiol valerate and 2 mg dienogest
17 light yellow tablets each containing 2 mg estradiol valerate and 3 mg dienogest
2 dark red tablets each containing 1 mg estradiol valerate Composition of the white inactive tablets:
These tablets do not contain any active substances.
Other ingredients in the coloured active tablets are:
Tablet core: lactose monohydrate, maize starch, pregelatinised maize starch, providone K25 (E1201), magnesium stearate (E572)
Tablet film-coating: hypromellose type 2910 (E464), macrogol 6000, talc (E553b), titanium dioxide (E171), iron oxide yellow (E172) and/or iron oxide red (E172)
Other ingredients in the white inactive tablets are:
Tablet core: lactose monohydrate, maize starch, providone K25 (E1201), magnesium stearate (E572).
Tablet film-coating: hypromellose type 2910 (E464), talc (E553b), titanium dioxide (E171)
What Qlaira looks like and content of the pack
Qlaira tablets are film-coated tablets; the core of the tablet is covered with a coating.
Each wallet (28 film-coated tablets) contains 2 dark yellow tablets in row 1,5 medium red tablets in row 1,17 light yellow tablets in rows 2, 3 and 4, 2 dark red tablets in row 4 as well as 2 white tablets in row 4.
The dark yellow round tablets are marked ‘DD’ in a hexagon on one side and plain on the reverse
The medium red round tablets are marked ‘DJ in a hexagon on one side and plain on the reverse
The light yellow round tablets are marked ‘DH’ in a hexagon on one side and plain on the reverse
The dark red round tablets are marked ‘DN’ in a hexagon on one side and plain on the reverse
The white inactive tablet are round tablets marked ‘DT’ in a hexagon on one side and plain on the reverse.
Available in packs containing 3 wallets each containing 28 tablets.
PL 20774/1258 Qlaira
Manufactured by: Bayer Weimer GmbH und Co. KG, 99427, Weimar, Germany or Bayer Pharma AG, 13342, Berlin, Germany. Procured from within the EU. Product Licence Holder: Quadrant Pharmaceuticals Ltd, Lynstock House, Lynstock Way, Lostock, Bolton, BL6 4SA.
Qlaira is a registered trademark of Bayer Schering Pharma.
Leaflet revision date: 23rd December 2013
PP3/1258/V1