Ropivacaine 2 Mg/Ml Solution For Injection
• If too much Ropivacaine is given into the spinal fluid, the whole body may become numbed (anaesthetised).
Children
In children, the side effects are the same as in adults except for low blood pressure which happens less often in children (affecting less than 1 in 10 children) and being sick which happens more often in children (affecting more than 1 in 10 children).
If any of the side effects get serious, or if you notice any side effects not listed in this leaflet, please tell your doctor or pharmacist.
5. HOW TO STORE ROPIVACAINE
Keep this medicine out of the reach and sight of children.
Do not use Ropivacaine after the expiry date which is stated on the ampoule or carton box. The expiry date refers to the last day of that month.
Do not freeze.
Do not use Ropivacaine if you notice any precipitation in the solution for injection.
Your doctor or the hospital will normally store Ropivacaine and they are responsible for the quality of the product when it has been opened if it is not used immediately. They are also responsible for disposing of any unused Ropivacaine correctly.
Medicines should not be disposed of via wastewater or household waste. Your doctor, nurse or pharmacist will dispose of medicines no longer required. These measures will help to protect the environment.
6. FURTHER INFORMATION What Ropivacaine contains
- The active substance is ropivacaine hydrochloride 2 mg/ml. Each 10 ml polypropylene ampoule contains 20 mg ropivacaine (as hydrochloride).
Each 20 ml polypropylene ampoule contains 40 mg ropivacaine, (as hydrochloride).
- The other ingredients are sodium chloride, sodium hydroxide (for pH adjustment) and water for injections.
What Ropivacaine looks like and contents of the pack
Ropivacaine solution for injection is a clear, colourless, sterile, isotonic, isobaric aqueous solution for injection.
Ropivacaine 2 mg/ml solution for injection is available in 10 ml and 20 ml transparent polypropylene ampoules.
Pack sizes:
10 sterile ampoules in plastic overwrap
Marketing Authorisation Holder and Manufacturer
Mercury Pharmaceuticals Ltd No. 1 Croydon,
12-16 Addiscombe Road,
Croydon,
CR0 0XT, UK
101950/LF/2
This leaflet was last approved in July 2012.
cc
CO
Infants and children aged 1-12 years:
The proposed ropivacaine hydrochloride doses for peripheral block in infants and children provide guidelines for use in children without severe disease. More conservative doses and close monitoring are recommended for children with severe disease.
Single injections for peripheral nerve block (e.g. ilioinguinal nerve block, brachial plexus block) should not exceed 2,5-3,0 mg/kg.
Continuous infusion for peripheral nerve block are recommended at 0,2-0,6 mg/kg/h (0,10,3 ml/kg/h) up to 72 h.
The use of ropivacaine hydrochloride in premature children has not been documented.
Method of administration Epidural administration by injection.
Careful aspiration before and during injection is recommended to prevent intravascular injection. The patient’s vital functions should be observed closely during the injection. If toxic symptoms occur, the injection should be stopped immediately.
A single caudal epidural injection of ropivacaine hydrochloride 2 mg/ml produces adequate postoperative analgesia below T12 in the majority of patients when a dose of 2 mg/kg is used in a volume of 1 ml/kg. The volume of the caudal epidural injection may be adjusted to achieve a different distribution of sensory block, as recommended in standard textbooks. In children above 4 years of age, doses up to 3 mg/kg of a concentration of ropivacaine hydrochloride 3 mg/ml have been studied. However, this concentration is associated with a higher incidence of motor block.
Fractionation of the calculated local anaesthetic dose is recommended, whatever route of administration.
In case infusion of ropivacaine hydrochloride is recommended, Ropivacaine solution for infusion can be used.
Incompatibilities
In the absence of compatibility studies, this medicinal product must not be mixed with other medicinal products.
In alkaline solutions precipitation may occur as ropivacaine hydrochloride shows poor solubility at pH > 6.0.
Disposal
Any unused product or waste material should be disposed of in accordance with local requirements.
PACKAGE LEAFLET: INFORMATION FOR THE USER
Ropivacaine 2 mg/ml solution for injection
Ropivacaine hydrochloride
Read all of this leaflet carefully before you start using this medicine.
- Keep this leaflet. You may need to read it again.
- If you have any further questions, ask your doctor or pharmacist.
- This medicine has been prescribed for you. Do not pass it on to others. It may harm them, even if their symptoms are the same as yours.
- If any of the side effects gets serious, or if you notice any side effects not listed in this leaflet, please tell your doctor or pharmacist.
In this leaf et:
1. What Ropivacaine is and what it is used for
2. Before you use Ropivacaine
3. How to use Ropivacaine
4. Possible side effects
5. How to store Ropivacaine
6. Further information
This product will be referred to as Ropivacaine from here on.
1. WHAT ROPIVACAINE IS AND WHAT IT IS USED FOR
Ropivacaine contains the active substance ropivacaine hydrochloride which is a type of medicine called local anaesthetic.
Ropivacaine 2 mg/ml solution for injection is used in adults and children of all ages for acute pain management. It numbs (anaesthetises) parts of the body e.g. after surgery.
2. BEFORE YOU USE ROPIVACAINE You must not be given Ropivacaine
- if you are allergic (hypersensitive) to ropivacaine hydrochloride, other so called local anaesthetics of the amide type or any of the other ingredients of Ropivacaine,
- if you have a decrease in blood volume (hypovolaemia). This is measured by healthcare personnel.
- for injection into a blood vessel to numb a specific area of your body,
- for injection into the neck of the womb to relieve pain during childbirth.
Take special care with Ropivacaine
- In newborn children as they are more susceptible to Ropivacaine.
- In children < 12 years as some injections of Ropivacaine in order to numb parts of the body are not established in younger children.
Special care should be taken to avoid any injection of Ropivacaine directly into a blood vessel to prevent any immediate toxic effects. Injection should not be performed in inflamed areas.
Please tell your doctor:
- if you are in a poor general conditon due to your age or other factors.
- if you have heart problems (partial or complete heart conduction block)
- if you have advanced liver problems
- if you have severe kidney problems.
Tell your doctor if you have any of these problems because your doctor may need to adjust the dose of Ropivacaine.
Please tell your doctor:
- if you suffer from acute porphyria (problems with building up red blood pigment, sometimes resulting in neurological symptoms).
Tell your doctor if you or somebody in your family have porphyria because your doctor may need to use another anaesthetic.
The following information is intended for medical or healthcare professionals only:
Handling
Ropivacaine should only be used by, or under the supervision of, clinicians experienced in regional anaesthesia (see section 3).
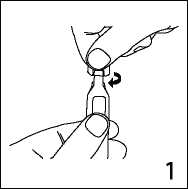
1. Hold ampoule upright and flick the neck to remove any solution.
Open by sharply twisting the top of the ampoule.
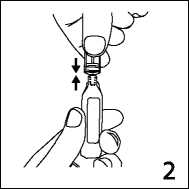
2. The ampoule can then be attached directly to the syringe as shown.
The ampoule fits both Luerfit and LuerLock syringes.
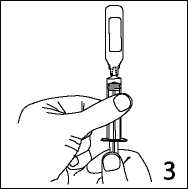
3. Hold the syringe with ampoule uppermost. Without squeezing the ampoule, withdraw the solution. Maintain downward pressure on the syringe piston after the solution has been withdrawn until the empty ampoule has been removed.
Shelf life
Shelf-life before opening 2 years
Shelf-life after opening
From a microbiological point of view, the product should be used immediately. If not used immediately, in-use storage times and conditions prior to use are the responsibility of the user and would normally not be longer than 24 hours at 2 to 8 °C.
Ropivacaine products are preservative free and are intended for single use only. Discard any unused solution.
The medicinal product should be visually inspected prior to use. The solution should only be used if it is clear, practically free from particles and if the container is undamaged.
The intact container must not be re-autoclaved.
Posology
Adults and children above 12 years of age
The following table is a guide to dosage for the more commonly used blocks. The smallest dose required to produce an effective block should be used. The clinician’s experience and knowledge of the patient’s physical status are of importance when deciding the dose.
In general, surgical anaesthesia (e.g. epidural administration) requires the use of the higher concentrations and doses. The Ropivacaine 10 mg/ml formulation is recommended for epidural anaesthesia in which a complete motor block is essential for the surgery. For analgesia (e.g. epidural administration for acute pain management) the lower concentrations and doses are recommended.
The evaluation of side effects is based on the following frequencies:
|
very common: |
affects more than 1 user in 10 |
|
common: |
affects 1 to 10 users in 100 |
|
uncommon: |
affects 1 to 10 users in 1,000 |
|
rare: |
affects 1 to 10 users in 10,000 |
|
very rare: |
affects less than 1 user in 10,000 |
|
not known: |
frequency cannot be estimated from the available data |
Taking other medicines
Please tell your doctor or healthcare personnel if you are taking or have recently taken any other medicines, including medicines obtained without a prescription.
Caution should be exercised if you are receiving:
- Other local anaesthetics (e.g. lidocaine) or agents structurally related to amide-type local anaesthetics, e.g. certain medicines used to treat an irregular heart beat (arrhythmia), such as mexiletine or amiodarone
- General anaesthetics or opioids, such as morphine or codeine
- Medicines used to treat depression (e.g. fluvoxamine)
- Certain antibiotics (e.g. enoxacin)
Pregnancy and breast-feeding
Before you are given Ropivacaine please tell your doctor if you are pregnant, planning to get pregnant, or if you are breast-feeding. It is not known if ropivacaine hydrochloride affects pregnancy or passes into breast milk.
Ask your doctor or pharmacist for advice before taking any medicine.
Driving and using machines
Ropivacaine may make you feel sleepy and affect the speed of your reactions. After you have been given Ropivacaine, you should not drive or use any tools or machines until the next day.
Discuss with your doctor or pharmacist if you are unsure about anything.
Important information about some of the ingredients of Ropivacaine
This medicinal product contains 0.147 mmol (or 3.39 mg) sodium per ml. To be taken into consideration by patients on a controlled sodium diet.
3. HOW TO USE ROPIVACAINE
Method of administration
Your doctor will administer Ropivacaine to you. It is administered by injection.
Dosage
The dose used will depend on what it is being used for and also on your health, age and weight. The smallest dose that can produce effective numbing (anaesthesia) of the required area should be used.
The usual dose
- for adults and adolescents older than 12 years of age is between 2 mg and 300 mg of
ropivacaine hydrochloride.
- in infants and children (0 up to and including 12 years of age) is 1-3 mg for each kilogram of body weight.
Duration of treatment
Administration of ropivacaine hydrochloride usually takes between 2 to 10 hours in case of anaesthesia prior to certain surgeries and can take up to 72 hours in case of pain relief during or after surgery.
If you are given more Ropivacaine than you should be
The first symptoms of being given too much ropivacaine hydrochloride are usually problems with
- hearing and sight,
- numbness around the mouth,
- dizziness or light-headedness,
- tingling,
- speech disorder characterised by poor articulation (dysarthria),
- muscular stiffness, muscular twitching, fits (convulsions),
- low blood pressure,
- slow or irregular heart beat.
These symptoms may precede to cardiac arrest, breathing arrest or severe fits.
If you experience any of these symptoms or think you may have received too much Ropivacaine, tell your doctor or healthcare personnel immediately.
In case of acute toxicity, appropriate corrective actions will be taken immediately by the healthcare personnel.
If you have any further questions on the use of this product, ask your doctor or pharmacist.
4. POSSIBLE SIDE EFFECTS
Like all medicines, Ropivacaine can cause side effects, although not everybody gets them.
Important side effects to look out for:
Sudden life-threatening allergic reactions (such as anaphylaxis, angioneurotic oedema and urticaria) are rare. Possible symptoms include
- sudden onset of rash,
- itching or lumpy rash (hives),
- swelling of the face, lips, tongue or other parts of the body,
- and shortness of breath, wheezing or difficulty breathing.
If you think that Ropivacaine is causing an allergic reaction, tell your doctor or healthcare personnel immediately.
Other possible side effects:
Very common
• Low blood pressure (hypotension). This might make you feel dizzy or light-headed.
• Feeling sick (nausea)
Common
• Headache, pins and needles (paraesthesia), feeling dizzy
• Slow or fast heart beat (bradycardia, tachycardia)
• High blood pressure (hypertension)
• Being sick (vomiting)
• Difficulty in passing urine (urinary retention)
• Back pain, increased temperature, muscular stiffness (rigor)
Uncommon
• Anxiety
• Some symptoms can happen if the injection was given into a blood vessel by mistake, or if you have been given too much Ropivacaine (see also section 3 “If you are given more Ropivacaine than you should be” above). These include fits (convulsions, seizures), feeling dizzy or light-headed, numbness of the lips and around the mouth, numbness of the tongue, hearing problems, problems with your sight (vision), problems with your speech (dysarthria), muscular twitching and trembling, reduced sense of touch (hypoaesthesia)
• Fainting (syncope)
• Difficulty breathing (dyspnoea)
• Low body temperature
Rare
• Cardiac arrest, irregular heart beat (cardiac arrhythmias)
Possible side effects seen with other local anaesthetics which might also be caused by Ropivacaine include:
• Numbness, due to nerve irritation caused by the needle or the injection. This does not usually last for long.
• Damaged nerves. Rarely, this may cause permanent problems.
|
Concentration Volume mg/ml ml |
Dose Onset mg minutes |
Duration hours | |||
|
Lumbar Epidural Administration Bolus |
2.0 |
10-20 |
20-40 |
10-15 |
0.5-1.5 |
|
Intermittent injections (top-up) (e.g. labour pain management) Continuous infusion e.g. Labour pain |
2.0 2.0 |
10-15 20-30 (minimum interval 30 minutes) 6-10 ml/h 12-20 mg/h |
- |
- | |
|
Postoperative pain management |
2.0 |
6-14 ml/h 12-28 mg/h |
- |
- | |
|
Thoracic Epidural Administration | |||||
|
Continuous infusion (postoperative pain management) |
2.0 |
6-14 ml/h 12-28 mg/h |
- |
- | |
|
Field Block (e.g. minor nerve blocks and infiltration) |
2.0 |
1-100 |
2.0-200 |
1-5 |
2-6 |
|
Peripheral nerve block (Femoral or interscalene block) | |||||
|
Continuous infusion or intermittent injections (e.g. postoperative pain management) |
2.0 |
5-10 ml/h 10-20 mg/h | |||
The doses in the table are those considered to be necessary to produce a successful block and should be regarded as guidelines for use in adults. Individual variations in onset and duration occur. The figures in the column ‘Dose’ refect the expected average dose range needed. Standard textbooks should be consulted for both factors affecting specific block techniques and individual patient requirements.
Method of administration
Perineural and epidural administration by injection.
Careful aspiration before and during injection is recommended to prevent intravascular injection. When a large dose is to be injected, a test dose of 3-5 ml lidocaine 2% (lignocaine) with adrenaline (epinephrine) 1:200.000 is recommended. An inadvertent intravascular injection may be recognised by a temporary increase in heart rate and an accidental intrathecal injection by signs of a spinal block.
Aspiration should be performed prior to and during administration of the main dose, which should be injected slowly or in incremental doses, at a rate of 25-50 mg/min, while closely observing the patient’s vital functions and maintaining verbal contact. If toxic symptoms occur, the injection should be stopped immediately.
In epidural block for surgery, single doses of up to 250 mg ropivacaine hydrochloride have been used and well tolerated.
In brachial plexus block a single dose of 300 mg has been used in a limited number of patients and was well tolerated.
When prolonged blocks are used, either through continuous infusion or through repeated bolus administration, the risks of reaching a toxic plasma concentration or inducing local neural injury must be considered. Cumulative doses up to 675 mg ropivacaine hydrochloride for surgery and postoperative analgesia administered over 24 hours were well tolerated in adults, as were postoperative continuous epidural infusions at rates up to 28 mg/hour for 72 hours. In a limited number of patients higher doses of up to 800 mg/day have been administered with relatively few adverse reactions.
For treatment of postoperative pain, the following technique can be recommended: Unless preoperatively instituted, an epidural block with Ropivacaine 7.5 mg/ml is induced via an epidural catheter. Analgesia is maintained with Ropivacaine 2 mg/ml infusion. Infusion rates of 6-14 ml (12-28 mg), per hour provide adequate analgesia with only slight and non-progressive motor block in most cases of moderate to severe postoperative pain. The maximum duration of epidural block is 3 days. However, close monitoring of analgesic effect should be performed in order to remove the catheter as soon as the pain condition allows it. With this technique a significant reduction in the need for opioids has been observed.
In clinical studies an epidural infusion of ropivacaine hydrochloride 2 mg/ml alone or mixed with fentanyl 1-4 pg/ml has been given for postoperative pain management for up to 72 hours. The combination of ropivacaine hydrochloride and fentanyl provided improved pain relief but caused opioid side effects. The combination of ropivacaine hydrochloride and fentanyl has been investigated only for ropivacaine hydrochloride 2 mg/ml.
When prolonged peripheral nerve blocks are applied, either through continuous infusion or through repeated injections, the risks of reaching a toxic plasma concentration or inducing local neural injury must be considered. In clinical studies, femoral nerve block was established with 300 mg ropivacaine hydrochloride 7.5 mg/ml and interscalene block with 225 mg ropivacaine hydrochloride 7.5 mg/ml, respectively, before surgery. Analgesia was then maintained with ropivacaine hydrochloride 2 mg/ml. Infusion rates or intermittent injections of 10-20 mg per hour for 48 hours provided adequate analgesia and were well tolerated.
Paediatric patients up to and including 12 years of age
|
Concentration |
Volume |
Dose | |
|
mg/ml |
ml/kg |
mg/kg | |
|
Single Caudal Epidural Block |
2.0 |
1 |
2 |
|
Blocks below T12, in children with a body weight up to 25 kg |
Continuous epidural infusion
In children with a body weight up to 25 kg
0 up to 6 months
|
Bolus dosea |
2.0 |
0.5-1 |
1-2 |
|
Infusion up to 72 hours |
2.0 |
0.1 ml/kg/h |
0.2 mg/kg/h |
|
6 up to 12 months | |||
|
Bolus dosea |
2.0 |
0.5-1 |
1-2 |
|
Infusion up to 72 hours |
2.0 |
0.2 ml/kg/h |
0.4 mg/kg/h |
|
1 to 12 years | |||
|
Bolus doseb |
2.0 |
1 |
2 |
|
Infusion up to 72 hours |
2.0 |
0.2 ml/kg/h |
0.4 mg/kg/h |
The dose in the table should be regarded as guidelines for use in paediatrics. Individual variations occur. In children with a high body weight a gradual reduction of the dosage is often necessary and should be based on the ideal body weight. The volume for single caudal epidural block and the volume for epidural bolus doses should not exceed 25 ml in any patient. Standard textbooks should be consulted for factors affecting specific block techniques and for individual patient requirements.
a Doses at the low end of the dose intervals are recommended for thoracic epidural blocks while doses in the high end are recommended for lumbar or caudal epidural blocks.
b Recommended for lumbar epidural blocks. It is good practice to reduce the bolus dose for thoracic epidural analgesia.