Salamol Steri-Neb 2.5Mg/2.5Ml Nebuliser Solution
•— 5 ro c
i— _o ;±3 o
a> 41 3 's
O o ts C ^ E I ru ^
-S E 8P 00 r\j 3 2.
Read all of this leaflet carefully before you start taking this medicine because it contains important information for you.
Keep this leaflet. You may need to read it again.
If you have any further questions, ask your doctor or pharmacist.
This medicine has been prescribed for you only. Do not pass it on to others. It may harm them, even if their signs of illness are the same as yours.
If you get any side effects, talk to your doctor or pharmacist.
This includes any possible side effects not listed in this leaflet. See section 4.
What is in this leaflet:
1. What Salamol Steri-Neb is and what it is used for
2. What you need to know before you take Salamol Steri-Neb
3. How to take Salamol Steri-Neb
4. Possible side effects
5. How to store Salamol Steri-Neb
6. Contents of the pack and other information
1. What Salamol Steri-Neb is and what it is used for
Your medicine is called Salamol Steri-Neb 2.5mg/2.5ml Nebuliser Solution which is a beta2-agonist which helps to manage asthma by dilating (opening up) the airways (or air passages) in the lungs.
Salamol is used to treat:
• acute severe asthma
• long-term bronchospasm (narrowing of the tubes to the lungs caused by muscle spasm)
that doesn't respond to conventional treatment.
2. What you need to know before you take Salamol Steri-Neb
Do not take Salamol Steri-Neb:
• if you are allergic to salbutamol sulfate or any of the other ingredients of this medicine (listed in section 6).
• for the management of premature labour. Warnings and precautions
Talk to your doctor or pharmacist before you start to take this medicine:
• if you suffer from thyroid problems
• if you have high blood pressure
• if you are diabetic
• if you have a history of heart disease, irregular or fast heartbeat or angina
• if you have a condition known as glaucoma (raised pressure in your eyes)
• if you suffer from hypoxia (deficiency of oxygen in the tissues).
Taking your medicine may lead to low potassium levels in your blood, so your doctor may want you to have regular blood tests to monitor this.
Other medicines and Salamol Steri-Neb
Please tell your doctor or pharmacist if you are taking or have recently taken any other medicines, including medicines obtained without a prescription.
Do not take Salamol Steri-Neb if you are taking the following:
• propranolol or any other non selective beta blocking drug
Talk to your doctor or pharmacist if you are taking any of the following:
• steroids such as prednisolone
• diuretics (water tablets usually used to treat high blood pressure)
• other sympathomimetic drugs such as Metaraminol or Xanthine derivatives such as aminophylline or theophylline (drugs used to improve breathing for people with asthma)
• you are using nebulised anticholinergic drugs (such as Ipratropium bromide) to treat your asthma; make sure the mist does not get in your eyes. This can be prevented by using a mouth piece instead of a face mask or goggles.
Salamol Steri-Neb with food, drink and alcohol
• You can use Salamol Steri-Neb at any time of day, with or without food.
Pregnancy, breast-feeding and fertility
• If you are pregnant, planning to become pregnant or are breast-feeding, ask your doctor for advice before taking any medicine.
Driving and using machines
• Salamol Steri-Neb is not expected to affect your ability to drive or operate machinery.
3. How to take Salamol Steri-Neb
Always take Salamol Steri-Neb exactly as your doctor or pharmacist has told you. You should check with your doctor or pharmacist if you are not sure.
You must only use Salamol Steri-Neb in a power-operated nebuliser. The nebuliser turns the solution into a fine mist that you can breathe in. Delivery of the aerosol may be by face mask or “T” piece.
Read the manufacturer's instructions and make sure you know how to use the nebuliser before you take your medicine.
Use in children and adolescents
The recommended dose is:
• Adults including the Elderly and children/ infants over 18 months
2.5mg taken up to three or four times a day.
Your doctor may increase this to 5mg up to three or four times a day.
Children should be supervised by an adult when they are using a nebuliser.
• Infants under 18 months
1.25mg (0.25mg/kg) up to four times a day. Your
doctor may increase this to 2.5mg up to four times a day. Your doctor may consider giving the infant oxygen therapy.
The nebuliser should be operated by an adult.
What to do if you think your medicine is not working
You should contact your doctor if the dose seems less effective or is short lasting. You should not change the dose you take, or how often you take your medicine without seeking medical advice.
How to use your Salamol Steri-Neb Nebuliser
• Prepare your nebuliser for use according to the manufacturer's instructions.
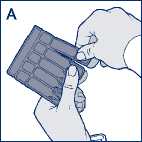
• Remove a Steri-Neb from the labelled strip by twisting and pulling (diagram A).
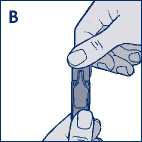
• Hold the Steri-Neb upright and twist off the cap (diagram B).
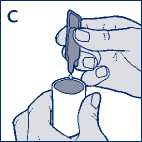
• Squeeze the contents into the reservoir of your nebuliser (diagram C).
• Following the manufacturer's instructions, use your nebuliser.
• After you have used your nebuliser, throw away any solution that is left in the reservoir.
• Clean your nebuliser thoroughly. Do not get the solution or mist in your eyes.
Do not dilute the solution or mix it with other medicines, unless your doctor tells you to.
Do not swallow the solution or use it in injections.
If you take more Salamol Steri-Neb than you should
If you (or someone else) swallow or take a lot of the solution all together, or if you think a child has swallowed or taken any of the solution; contact your nearest hospital casualty department or your doctor immediately. An overdose is likely to cause a very rapid heart beat and feeling shaky.
Please take this leaflet, any remaining solution and the container with you to the hospital or doctor so that they know which solution has been consumed. If you forget to take Salamol Steri-Neb If you forget to take a dose, take one as soon as you remember, unless it is nearly time to take the next one. Do not take a double dose to make up for a forgotten dose.
If you stop taking Salamol Steri-Neb
Do not stop taking your medicine without talking to your doctor first even if you feel better.
If you have any further questions on the use of this product, ask your doctor or pharmacist
4. Possible side effects
Like all medicines, this medicine can cause side effects, although not everybody gets them.
If the following happens, stop taking the solution and tell your doctor immediately or go to the casualty department at your nearest hospital:
• an allergic reaction (swelling of the lips, face or neck leading to severe difficulty in breathing; skin rash or hives)
• a sudden increase in breathlessness or wheezing, which may be due to paradoxical bronchospasm (narrowing of the air passage or tightening of the chest), low blood pressure
• angioedema, a serious allergic reaction causing swelling of the face or throat.
These are very serious but rare side effects.
You may need urgent medical attention or hospitalisation.
Talk to your doctor as soon as possible:
• if you feel your heart is beating faster or stronger than usual (palpitations). This is usually harmless, and usually stops after you have used the medicine for a while
• if you may feel your heartbeat is uneven or it gives an extra beat
Do not stop using this medicine unless told to do so.
Tell your doctor if you have any of the following side effects which may also happen with this medicine:
Common (may affect up to 1 in 10 people)
• Feeling shaky
• Headache
Uncommon (may affect up to 1 in 100 people)
• Mouth and throat irritation
• Muscle cramps
Rare (may affect up to 1 in 1,000 people)
• A low level of potassium in your blood
• Increased blood flow to your extremities (peripheral dilatation)
Very rare (may affect up to 1 in 10,000 people))
• Changes in sleep patterns and changes in behaviour, such as restlessness and excitability
Not known (frequency cannot be estimated from the available data)
• Chest pain, due to heart problems such as angina. Tell your doctor or pharmacist if this occurs. Do not stop using this medicine unless told to do so
• A condition known as lactic acidosis which may cause stomach pain, hyperventilation, shortness of breath, cold feet and hands, irregular heartbeat or thirst.
If you think this medicine is not working well enough for you
If your medicine does not seem to be working as well as usual, talk to your doctor as soon as possible. Your chest problem may be getting worse and you may need a different medicine.
Do not take extra Salamol Steri-Neb unless your doctor tells you to.
Reporting of side effects If you get any side effects, talk to your doctor or pharmacist. This includes any possible side effects not listed in this leaflet. You can also report side effects directly via the Yellow Card Scheme at: www.mhra.gov.uk/yellowcard.
By reporting side effects you can help provide more information on the safety of this medicine.
5. How to store Salamol Steri-Neb
Keep this medicine out of the sight and reach of children.
Do not store above 25°C. Do not refrigerate or freeze. Keep container in outer carton to protect from light.
For single use only. Use immediately. Discard any unused portion.
Do not use this medicine after the expiry date that is stated on the outer packaging. The expiry date refers to the last day of that month.
Do not use Salamol Steri-Neb if you notice the solution is cloudy. Never give it to anyone else.
It may harm them even if they seem to have the same symptoms as you have.
Do not throw away any medicines via wastewater or household waste. Ask your pharmacist how to throw away medicines you no longer use. These measures will help protect the environment.
6. Contents of the pack and other information
What Salamol Steri-Neb solution contains:
• The active ingredient is salbutamol sulfate.
• The other ingredients are sodium chloride, dilute sulfuric acid, and water for injections.
What Salamol Steri-Neb solution looks like and contents of the pack:
• Each Steri-Neb contains 2.5ml of a solution that
is equivalent to 2.5mg of Salbutamol (1mg/ml). There are 20 Steri-Nebs of your medicine in this pack.
• Strips of 5 Steri-Nebs are packed into foil laminate pouches, which in turn are packed into boxes containing 20 and 100 (ampoules).
Not all pack sizes may be marketed.
Marketing Authorisation Holder and Manufacturer
Marketing Authorisation holder:
Norton Healthcare Ltd T/A IVAX Pharmaceuticals UK,
Ridings Point, Whistler Drive, Castleford, West Yorkshire, United Kingdom, WF10 5HX.
Company responsible for manufacture:
IVAX Pharmaceuticals UK, Aston lane North, Whitehouse Vale Industrial Estate, Preston Brook, Runcorn, Cheshire, United Kingdom, WA7 3FA.
The leaflet was last updated:
January 2016 PL 00530/0690 23-30-11045-H
Package Leaflet: Information for the User

