Salbulin Mdpi Novolizer 100 Micrograms / Dose Inhalation Powder
#
#
PACKAGE LEAFLET: INFORMATION FOR THE USER
SALBULIN® MDPI NOVOLIZER®
100 micrograms/dose inhalation powder
Salbutamol
DO NOT take Salbulin MDPI Novolizer if:
• you are allergic (hypersensitive) to salbutamol, lactose or milk protein
Warnings and precautions Talk to your doctor before using Salbulin MDPI Novolizer if you can answer 'yes' to any of the following questions:
• do you have any heart or circulatory problems, particularly a recent heart attack or angina?
• do you suffer from high blood pressure?
• have you been told you have an aneurysm (a swelling or dilation of a blood vessel)?
• do you have an overactive thyroid gland?
• do you suffer from diabetes which is difficult to control?
• do you have a tumour near your kidney (a phaeochromocytoma)?
• are you about to have an operation requiring a general anaesthetic?
• are you taking any other medicines and/or inhalers for your asthma either prescribed by a doctor or bought from a pharmacy?
Seek medical advice if you experience chest pain or other symptoms of worsening of your heart disease.
Your doctor may wish to alter your treatment or monitor your condition more closely if any of the above apply to you.
Care should be taken when treating acute asthma attacks or exacerbation of severe asthma as increased serum lactate levels, and rarely, lactic acidosis have been reported after the use of high doses of salbutamol. This is reversible on reducing the dose of salbutamol.
Read all of this leaflet carefully before you start using this medicine because it contains important information for you.
• Keep this leaflet. You may need to read it again.
• If you have any further questions, ask your doctor or pharmacist.
• This medicine has been prescribed for you only. Do not pass it on to others. It may harm them, even if their signs of illness are the same as yours.
• If you get any side effects, talk to your doctor or pharmacist. This includes any possible side effects not listed in this leaflet. See section 4.
What is in this leaflet
1. WHAT SALBULIN MDPI NOVOLIZER IS AND WHAT IT IS USED FOR.
2. WHAT YOU NEED TO KNOW BEFORE USING SALBULIN MDPI NOVOLIZER.
3. HOW TO USE SALBULIN MDPI NOVOLIZER.
4. POSSIBLE SIDE-EFFECTS.
5. HOW TO STORE SALBULIN MDPI NOVOLIZER.
6. CONTENTS OF THE PACK AND OTHER INFORMATION.
7. INSTRUCTIONS FOR USING THE NOVOLIZER DEVICE.
1. WHAT SALBULIN MDPI NOVOLIZER IS AND WHAT IT IS USED FOR
Salbulin MDPI Novolizer is indicated in adults, adolescents and children aged 6 to 12 years.
Salbulin MDPI Novolizer is a powder for inhalation containing the active substance salbutamol.
Salbutamol belongs to a group of anti-asthmatic drugs known as bronchodilators which are used to relieve symptoms such as chest tightness, wheezing or cough caused by asthma or other conditions. Salbutamol can also be used to prevent asthma symptoms brought on by exercise and other 'triggers' such as pollen.
For instructions for using the Novolizer Inhaler Device, see section 7 on the reverse side of this leaflet.
2. WHAT YOU NEED TO KNOW BEFORE YOU USE SALBULIN MDPI NOVOLIZER
Other medicines and Salbulin MDPI Novolizer
Treatment with salbutamol may cause hypokalaemia (low potassium in your blood) which can be aggravated by some other drugs. These include other drugs for asthma, such as xanthine derivatives (e.g. theophylline) or steroids (e.g. prednisolone), or drugs for other conditions such as water tablets (e.g. frusemide) or digoxin. You must mention to your doctor if you are taking any of these drugs, as your doctor may want to take a blood sample to monitor your potassium levels.
If you are taking beta-blockers (e.g. atenolol) for high blood pressure or angina, or antidepressants (e.g. moclobemide, phenelzine, amitriptyline, clomipramide or imipramine) you should also tell your doctor.
Some general anaesthetics may interact with salbutamol to cause heart problems and decrease blood pressure. Therefore, if you are having an operation, advise hospital staff that you are taking Salbulin MDPI Novolizer.
Tell you doctor or pharmacist if you are taking, have recently taken or might take any other medicines including inhalers and even those medicines not prescribed by your doctor and bought without a prescription.
Salbulin MDPI Novolizer with food and drink
Food and drink are not known to alter the effectiveness of Salbulin MDPI Novolizer
Pregnancy, breast feeding and fertility
If you are pregnant or breast feeding, think you may be pregnant or are planning to have a baby, ask your doctor or pharmacist for advice before taking this medicine.
If taken during pregnancy salbutamol will pass through the placenta and may cause the heart rate of the foetus (unborn baby) to increase. However, there is no evidence that salbutamol causes any malformations. Salbutamol also passes into a mother's milk and may cause some side effects in a breast-fed infant (see section 4 below).
Driving and using machines
No studies on the ability to drive and use machines have been performed.
Salbulin MDPI Novolizer contains milk sugar (lactose).
You should not take Salbulin MDPI Novolizer if you are allergic to lactose.
Normally, the content of lactose in a single dose does not cause any problems in persons with lactose intolerance.
If you are concerned that you may have an intolerance, you must talk to your doctor.
3. HOW TO USE SALBULIN MDPI NOVOLIZER
Always use this medicine exactly as your doctor has told you. Check with your doctor or pharmacist if you are unsure.
The label should remind you of how many actuations (puffs) to take and how often you should take them. Do not take more than your doctor has told you to. Your doctor or nurse should also have shown you how to use the Novolizer Device, but please read the instructions for using this device carefully before taking this medicine. These instructions can be found on the reverse of this leaflet.
The following information about the use of Salbulin MDPI Novolizer applies unless your doctor has instructed otherwise.
Please also note that if you are changing from another salbutamol inhaler to Salbulin MDPI Novolizer you may find that you have to adjust your dose.
Use in adults and adolescents above 12 years of age
For relief of symptoms: Start with 1 puff; if needed the dose may be increased to 2 puffs. You should leave at least 1 minute between each puff.
A further dose should not be taken within 4 hours.
For prevention of exercise-induced asthma and wheezing before contact with a 'trigger': 2 puffs should be taken 10-15 minutes before exercise or contact with the 'trigger'.
Maximum daily dose: 8 puffs
Use in children 6 -12 years of age
For relief of symptoms: Start with 1 puff; if needed the dose may be increased to 2 puffs. You should leave at least 1 minute between each puff.
A further dose should not be taken within 4 hours.
For prevention of exercise-induced asthma and wheezing before contact with a 'trigger': 1 puff should be taken 10-15 minutes before exercise or contact with the 'trigger'. If needed this may be increased to 2 puffs.
Maximum daily dose: 4 puffs
Children below 6 years of age
Salbulin MDPI Novolizer is not recommended for use in children below the age of 6 because there is insufficient information on safety and effectiveness in this age group.
You must not take more puffs than your doctor advises. If your asthma is getting worse you should discuss your treatment with your doctor, as you may need additional medicine for your chest condition. The following may indicate your asthma is getting worse:
• You find that you need more puffs than usual
• Relief of your chest tightness is not sufficient
• You notice that the relief is not lasting as long
IF YOUR BREATHING SUDDENLY BECOMES WORSE OR YOU ARE UNABLE TO TRIGGER THE NOVOLIZER DEVICE YOU MUST CONTACT YOUR DOCTOR IMMEDIATELY.
If you have asthma your doctor may have instructed you on the use of a peak-flow meter. It is important to monitor your asthma and the effectiveness of the asthma treatments you have been given.
If you inhale more doses than you should
If you take more puffs than recommended you may notice that your heart beats faster than usual and you may feel shaky. These effects usually wear off in a few hours, but you should contact your doctor immediately.
If you need to see another doctor or go to hospital
If you see any other doctor or have to go to hospital for any reason, you should take all your inhalers and any other medicines with you including those bought without a prescription. If you have any further questions on the use of this product, ask your doctor or pharmacist.
4. POSSIBLE SIDE EFFECTS
Like all medicines, Salbulin MDPI Novolizer can cause side effects, although not everybody gets them.
The common side effects (may affect up to 1 in 10 people) you are most likely to notice when using Salbulin MDPI Novolizer include: unpleasant or unusual taste, irritation of the throat and mouth causing a cough, shakiness (usually in the hands), dizziness, restlessness, headache, sweating and feeling sick. Muscle cramp can also occur, but this is rare (may affect up to 1 in 1,000 people). These effects, if noticed will usually wear off within 2 weeks of treatment.
Very rarely (may affect up to 1 in 10,000 people) inflammation of the kidney and a drop in blood platelets can occur. Sometimes some people may notice their heart beating faster or unevenly (palpitations), or feeling faint. These occur only rarely with inhaled salbutamol, but if you are affected tell your doctor.
Very rarely, some people may experience chest pain (due to heart problems such as angina). Tell your doctor as soon as possible, but do not stop using this medicine unless told to do so.
As with all inhalers it is possible for the powder to make you wheezy immediately after inhalation. This occurs rarely. If this happens you must call your doctor immediately. Allergic reactions to salbutamol, although very rare, have been reported. These include: rash, skin problems, swelling of the face, lips, eyes and throat, wheeziness, low blood pressure and collapse. If you notice any of these reactions contact your doctor immediately.
Children using this medicine may rarely become hyperactive or excitable. They may also suffer sleep disturbance or hallucinations, although this is very rare. Lactose-monohydrate contains small amounts of milk proteins and can therefore cause allergic reactions.
Reporting of side effects
If you get any side effects, talk to your doctor, pharmacist or nurse. This includes any possible side effects not listed in this leaflet. You can also report side effects directly via the Yellow Card Scheme at: www.mhra.gov.uk/yellowcard.
By reporting side effects you can help provide more information on the safety of this medicine.
5. HOW TO STORE SALBULIN MDPI NOVOLIZER
Keep this medicine out of the sight and reach of children.
Do not use this medicine after the expiry date which is stated on the label, the carton and the cartridge container. The expiry date refers to the last day of that month. The cartridges containing Salbulin MDPI Novolizer should be kept in their sealed outer container until required for use. When the Novolizer device is in-use it should be stored in a dry place to ensure that the powder cartridge inside is protected from moisture. Regardless of whether the cartridge is empty or not after 6 months a new cartridge should be inserted. It is recommended that you keep a record of when you replace the cartridges to help you remember when you will need to insert a new one. The Novolizer device should be replaced after 2000 inhalations. Therefore, a maximum of 10 cartridges should be used with one device. In any event it is recommended that you replace this device at least once a year.
Do not store above 30°C.
Store in original package.
Protect from moisture.
Do not throw away any medicines via wastewater or household waste. Ask your pharmacist how to throw away medicines you no longer use. These measures will help to protect the environment.
6. CONTENTS OF THE PACK AND OTHER INFORMATION
What Salbulin MDPI Novolizer contains
Salbulin MDPI Novolizer is a dry powder for inhalation. The active substance is salbutamol. Each actuation (puff) contains 100 micrograms of salbutamol (as sulphate). The other ingredient is Lactose monohydrate.
What Salbulin MDPI Novolizer looks
like and contents of pack
The following packs may be available:
An Original sales pack which contains a Salbulin MDPI Novolizer dry powder inhaler device and a Salbulin MDPI Novolizer drug cartridge sealed in a plastic container with aluminium foil.
Each cartridge contains 200 inhalations (puffs).
A Refill pack which contains either 1 or 2 Salbulin MDPI Novolizer drug cartridges sealed in a plastic container with aluminium foil. These are for use with the Salbulin MDPI Novolizer dry powder inhaler device. Each refill cartridge contains 200 inhalations (puffs).
A Hospital pack with pack of 10 of
original sales packs
Not all pack sizes may be marketed.
The Manufacturer of this product is:
Meda Pharma GmbH & Co. KG BenzstraGe 1
D-61352 Bad Homburg, Germany or
Vemedia Manufacturing B.V.
Verrijn Stuartweg 60
NL-1112 AX Diemen, The Netherlands
or
Meda Manufacturing GmbH Neurather Ring 1 D-51063 Cologne, Germany
The Marketing Authorisation Holder is:
Meda Pharmaceuticals Ltd Skyway House, Parsonage Road, Takeley, Bishop's Stortford, CM22 6PU United Kingdom
This medicinal product is authorised in the Member States of the EEA under the following names:
United Kingdom: Salbulin® MDPI Novolizer®, 100 micrograms/dose inhalation powder Germany: Salbutamol MDPI Novolizer®,
100 Mikrogramm/Dosis Pulver zur Inhalation.
pharma
Cartridge
Dosage Counter
Lid

Cartridge box
Button
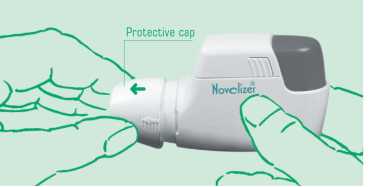
Protective cap

Control window
The Novolizer Device is a breath-actuated dry powder inhaler. It helps to make inhaling simple and reliable. It includes safety features to ensure that you take the correct dose and it is also environmentally friendly as it does not use chlorofluorocarbon (CFC) propellants. The following instructions for using the Novolizer Device are brief and easy to understand, but it is recommended that children use this device under the supervision of an adult.
Please note that the Novolizer Device supplied for use with Salbulin MDPI Novolizer must only be used with this medicine.
Inserting a cartridge
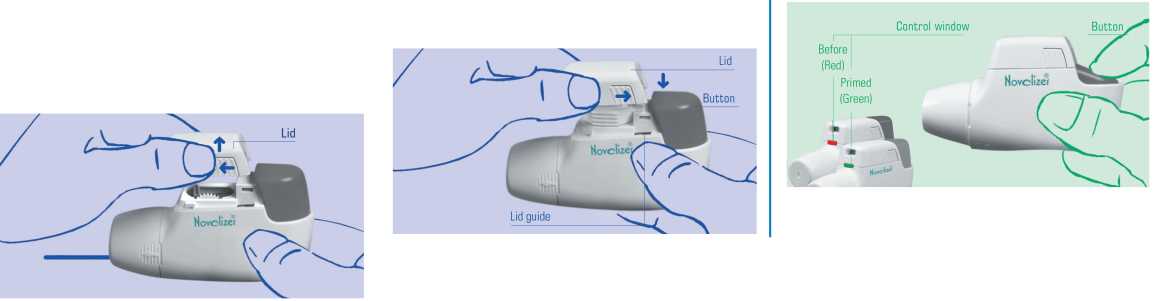
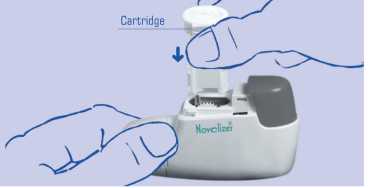
1. When you first get the Novolizer Device you will need to put the cartridge containing the medicine into the device. To do this, gently squeeze the ribbed surfaces on both sides of the lid. Move the lid forwards (^-) and off (^).
1. When using the Novolizer Device always keep it horizontal. Remove the protective cap, by lightly squeezing the sides and sliding forwards (^).
2. To 'prime' the Novolizer Device push down hard on the large coloured button until it stops (40 (see picture). You will hear a loud double click and the colour of the control window will change from red to green. You can now release the large button. The colour green in the control window indicates that the Novolizer Device is now primed and ready to use. The large button should only be pressed immediately before inhaling.
4.The loud click and the colour change from green to red in the control window indicate that you have inhaled correctly. The figures in the top window show the number of inhalations (puffs) remaining. Each inhalation is counted, but please note that figures in the top window count down in steps of 20 until 60 puffs remain and then in steps of 10 when it is below 60.
This cartridge MUST be replaced when either:
• '0' appears in the middle of the counter window or
• every 6 months, even if the cartridge is not empty.
Please note: if there is no click and the colour in the control window does not change please repeat the procedure as described above. If you cannot manage to inhale correctly after several attempts, please consult your doctor, nurse or pharmacist .
4. Replace the lid into the lid guides from above (+) and push down flat towards the dosage button (^) until it clicks into place.
2. Remove the protective aluminium foil from the cartridge box and take out the new cartridge. Only do this just before you need to use the cartridge.
5. The Novolizer Device is now filled and ready for use. You can leave the cartridge in the Novolizer Device until '0' appears in the middle of the counter window; however, you must replace it with a new cartridge within 6 months of taking the cartridge out of its sealed outer container.

3. Insert the cartridge into the Novolizer Device with the dosage counter facing the mouthpiece (^). Do not press the dosage button while inserting the cartridge.
6. When the cartridge needs replacing remove the lid as described in step 1 and lift out the used cartridge. Insert a new cartridge following steps 2 to 5.
3. Breathe out deeply, but not into the Novolizer Device. Put your lips around the mouthpiece. Inhale the powder with a strong deep breath. During this breath you should hear a loud click. This click indicates that you have breathed in strongly enough for the Novolizer Device to work. Continue inhaling as strongly and deeply as you can. Hold your breath for a few seconds before breathing out slowly through your nose. If you need to take another dose repeat steps 2 and 3. Replace the protective cap on the mouthpiece.

Cleaning
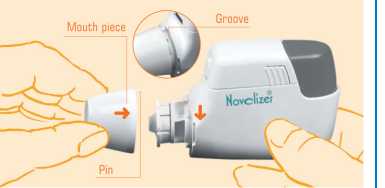
1. You should clean your Novolizer Device regularly, or at least whenever you change the cartridge. To do this first remove the protective cap. Then twist the mouthpiece anticlockwise slightly (^). It will become loose and you can remove it (^).

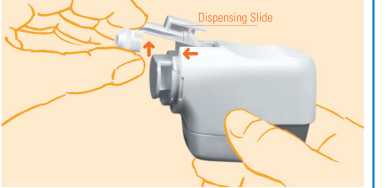
2. Now turn the Novolizer Device upside down. Loosen the dispensing slide and move it forwards (4-) and upwards (^). Any remaining powder can be removed from the dispensing slide by tapping it lightly. Clean the mouthpiece, the dispensing slide and the powder inhaler with a soft and dry lint-free cloth.
DO NOT use water or detergent.
1. The Novolizer Device can be refilled using new cartridges containing Salbulin MDPI Novolizer. It is ideal for long-term usage, but you must remember to replace your Novolizer Device after 2000 puffs (10 cartridges). In any event you must replace it within a year. Make sure your Novolizer Device is protected from moisture.
Remember to replace the cartridge every 6 months even if it is not empty.
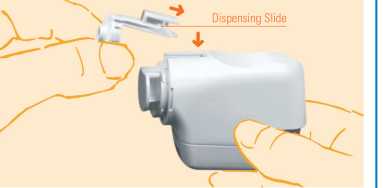
3. After cleaning, re-insert the dispensing slide by sliding down at an angle (il) and press back into position (40. You will hear a click as it fits into position.
4. Insert the mouthpiece so that the small pin fits into the groove on the Novolizer Device just below the colour control window (^). Twist the mouthpiece clockwise (40 so that it clicks into place.
Finish by replacing protective cap.
This leaflet was last revised in January 2015
neDQ
® Meda GmbH & Co. KG © Meda Pharmaceuticals Ltd.
T5007592_p01.indd 2 11.02.15 08:41