Trusopt 20Mg/Ml Eye Drops Solution
S1585 LEAFLET Trusopt 20160104
PACKAGE LEAFLET: INFORMATION FOR THE USER TRUSOPT® 20 mg/ml EYE DROPS SOLUTION (dorzolamide hydrochloride)
Your medicine is called as Trusopt 20 mg/ml Eye Drops Solution but will be referred to as Trusopt throughout the following leaflet. Read all of this leaflet carefully before you start using this medicine because it contains important information for you.
- Keep this leaflet. You may need to read it again.
- If you have any further questions, ask your doctor or pharmacist.
- This medicine has been prescribed for you only. Do not pass it on to others. It may harm them, even if their signs of illness are the same as yours.
- If you get any side effects, talk to your doctor or pharmacist. This includes any possible side effects not listed in this leaflet. See section 4.
What is in this leaflet:
1. What Trusopt is and what it is used for
2. What you need to know before you use Trusopt
3. How to use Trusopt
4. Possible side effects
5. How to store Trusopt
6. Contents of the pack and other information
1. WHAT TRUSOPT IS AND WHAT IT IS USED FOR
Trusopt contains dorzolamide which belongs to a group of medicines called “carbonic anhydrase inhibitors”.
This medicine is prescribed to lower raised pressure in the eye and to treat glaucoma. This medicine can be used alone or in addition to other medicines which lower the pressure in the eye (so-called beta-blockers).
2. WHAT YOU NEED TO KNOW BEFORE YOU USE TRUSOPT
Do not use Trusopt:
- if you are allergic to dorzolamide hydrochloride or any of the other ingredients of this medicine (listed in section 6).
- if you have severe kidney impairment or problems, or a prior history of kidney stones.
Warnings and precautions
Talk to your doctor or pharmacist before using Trusopt.
Tell your doctor or pharmacist about any medical problems you have now or have had in the past, including eye problems and eye surgeries, and about any allergies to any medications.
If you develop any eye irritation or any new eye problems such as redness of the eye or swelling of the eyelids, contact your doctor immediately.
If you suspect that Trusopt is causing an allergic reaction (for example, skin rash, severe skin reaction or itching), stop using this medicine and contact your doctor immediately.
Use in children
Trusopt has been studied in infants and children less than 6 years of age who have raised pressure in the eye(s) or have been diagnosed with glaucoma. For more information, talk to your doctor.
Use in elderly
In studies with Trusopt, the effects of this medicine were similar in both elderly and younger patients.
Use in patients with liver impairment
Tell your doctor about any liver problems you now have or have suffered from in the past.
Other medicines and Trusopt
Tell your doctor or pharmacist if you are using, have recently used or might use any other medicines (including eye drops). This is particularly important if you are taking another carbonic anhydrase inhibitor such as acetazolamide, or a sulpha drug.
Pregnancy and breast-feeding
Ask your doctor or pharmacist for advice before taking any medicine.
Use in pregnancy
You should not use this medicine during pregnancy. Tell your doctor if you are pregnant or intend to become pregnant.
Use in breast-feeding
If treatment with this medicine is required, breast-feeding is not recommended. Tell your doctor if you are breast-feeding or intend to breast-feed.
Driving and using machines
No studies on the effects on the ability to drive or use machines have been performed. There are side effects associated with Trusopt, such as dizziness and blurred vision, which may affect your ability to drive and/or operate machinery. Do not drive or operate machinery until you feel well or your vision is clear.
Trusopt contains benzalkonium chloride
Trusopt contains the preservative benzalkonium chloride. This preservative may be deposited in soft contact lenses and may possibly discolour the lenses. If you wear contact lenses, you should consult your doctor before using this medicine.
3. HOW TO USE TRUSOPT
Always use this medicine exactly as your doctor has told you. Check with your doctor or pharmacist if you are not sure. The appropriate dosage and duration of treatment will be established by your doctor.
When this medicine is used alone, the recommended dose is one drop in the affected eye(s) in the morning, in the afternoon and in the evening.
If your doctor has recommended you use this medicine with a beta-blocker eye drop to lower eye pressure, then the recommended dose is one drop of Trusopt in the affected eye(s) in the morning and in the evening.
If you are using Trusopt with another eye drop, the drops should be instilled at least 10 minutes apart.
Do not allow the tip of the container to touch the eye or areas around the eye. It may become contaminated with bacteria that can cause eye infections leading to serious damage of the eye, even loss of vision. To avoid possible contamination, wash your hands before using this medicine and keep the tip of the container away from contact with any surface. If you think your medication may be contaminated, or if you develop an eye infection, contact your doctor immediately concerning continued use of this bottle. Instructions for use:
1. Before using the medication for the first time, be sure the Safety Strip on the front of the bottle is unbroken. A gap between the bottle and the cap is normal for an unopened bottle.
Opening Arrows Safety Strip

2. First wash your hands, then tear off the Safety Strip to break the seal.
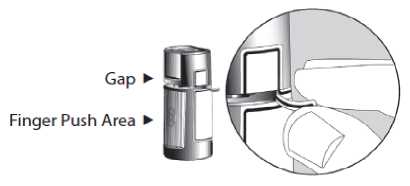
3. To open the bottle, unscrew the cap by turning as indicated by the arrows on the top of the cap. Do not pull the cap directly up and away from the bottle. Pulling the cap directly up will prevent your dispenser from operating properly.
4.
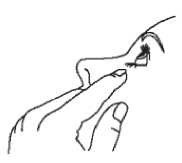
Tilt your head back and pull your lower eyelid down slightly to form a pocket between your eyelid and your eye.
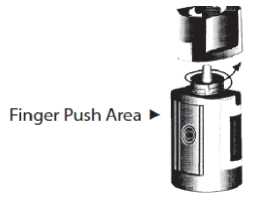
5. Invert the bottle, and press lightly with the thumb or index finger over the “Finger Push Area” (as shown) until a single drop is dispensed into the eye as directed by your doctor.
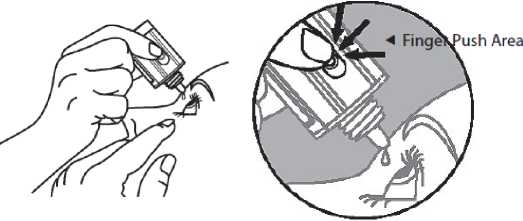
DO NOT TOUCH YOUR EYE OR EYELID WITH THE DROPPER
TIP.
6. If drop dispensing is difficult after opening for the first time, replace the cap on the bottle and tighten (Do not overtighten) and then remove by turning the cap in the opposite directions as indicated by the arrows on the top of the cap.
7. Repeat steps 4 & 5 with the other eye if instructed to do so by your doctor.
8. Replace the cap by turning until it is firmly touching the bottle. The arrow on the left side of the cap must be aligned with the arrow on the left side of the bottle label for proper closure. Do not overtighten or you may damage the bottle and cap.
9. The dispenser tip is designed to provide a single drop; therefore, do NOT enlarge the hole of the dispenser tip.
10. After you have used all doses, there will be some medicine left in the bottle. You should not be concerned since an extra amount of medicine has been added and you will get the full amount of medicine that your doctor prescribed. Do not attempt to remove the excess medicine from the bottle.
If you use more T rusopt than you should
If you put too many drops in your eye or swallow any of the contents of the container, you should contact your doctor immediately.
If you forget to use Trusopt
It is important to take this medicine as prescribed by your doctor. If you miss a dose, take it as soon as possible. However, if it is almost time for the next dose, skip the missed dose and go back to your regular dosing schedule.
Do not take a double dose to make up for the forgotten dose.
If you stop using Trusopt
If you want to stop using this medicine talk to your doctor first. If you have any further questions on the use of this product, ask your doctor or pharmacist.
4. POSSIBLE SIDE EFFECTS
Reporting of side effects
If you get any side effects, talk to your doctor, pharmacist or nurse. This includes any possible side effects not listed in this leaflet. You can also report side effects directly via the yellow card scheme at www.mhra.gov.uk/yellowcard
By reporting side effects you can help provide more information on the safety of this medicine.
5. HOW TO STORE TRUSOPT
• Keep out of the sight and reach of children.
• Store in the original package to protect from light.
• Do not use Trusopt after the expiry date which is stated on the carton and bottle. The expiry date refers to the last day of that month.
• Trusopt should be used within 28 days after the bottle is opened.
• If the medicine becomes discoloured or shows any other signs of deterioration, you should seek the advice of your pharmacist who will tell you what to do.
• Medicines should not be disposed of via wastewater or household waste. Ask your pharmacist how to dispose of medicines no longer required. These measures will help to protect the environment.
6. CONTENTS OF THE PACK AND OTHER INFORMATION
What Trusopt contains
• Each 1ml solution contains 20mg of active ingredient dorzolamide (as 22.26mg dorzolamide hydrochloride).
• The other ingredients are hydroxyethyl cellulose, mannitol, sodium citrate, sodium hydroxide and water for injection. Benzalkonium chloride is added as a preservative.
What Trusopt looks like and contents of the pack
Trusopt is a clear, colourless to nearly colourless slightly viscous solution. The Ocumeter Plus Ophthalmic Dispenser dropper bottle contains 5ml of solution in single packs. Tamper evidence is provided by a safety strip located on the container label.
Product Licence holder
Procured from within the EU and repackaged by the Product Licence holder: S&M Medical Ltd, Chemilines House, Alperton Lane, Wembley, HA0 1DX.
Manufacturer
This product is manufactured by Laboratoires Merck Sharp & Dohme-Chibret, Mirabel Plant, Route de Marsat, Riom 63963 Clermont Ferrand Cedex 9, France.
Like all medicines, this medicine can cause side effects, although not everybody gets them.
If you develop allergic reactions including hives, swelling of the face, lips, tongue, and/or throat which may cause difficulty in breathing or swallowing, you should stop using this medicine and seek immediate medical advice.
The following side effects have been reported with Trusopt either during clinical trials or during post-marketing experience:
Very Common side effects: (affects more than 1 user in 10) Burning and stinging of the eyes.
Common side effects: (affects 1 to 10 users in 100)
Disease of the cornea with sore eye and blurred vision (superficial punctuate keratitis), discharge with itching of the eyes (conjunctivitis), irritation/inflammation of the eyelid, blurred vision, headache, nausea, bitter taste, and fatigue.
Uncommon side effects: (affects 1 to 10 users in 1,000) Inflammation of the iris.
Rare side effects: (affects 1 to 10 user in 10,000)
Tingling or numbness of the hands or feet, temporary shortsightedness which may resolve when treatment is stopped, development of fluid under the retina (choroidal detachment, following filtration surgery), eye pain, eyelid crusting, low pressure in the eye, swelling of the cornea (with symptoms of visual disturbances), eye irritation including redness, kidney stones, dizziness, nose bleed, throat irritation, dry mouth, localized skin rash (contact dermatitis), severe skin reactions, allergic type reactions such as rash, hives, itching, in rare cases possible swelling of the lips, eyes and mouth, shortness of breath, and more rarely wheezing.
I POM | PL 19488/1585 Trusopt 20 mg/ml Eye Drops Solution
Leaflet revision date: 04 January 2016
Trusopt is a registered trade mark of Merck & Co., Inc., USA
S1585 LEAFLET Trusopt 20160104
S1585 LEAFLET Dorzolamide 20160104
PACKAGE LEAFLET: INFORMATION FOR THE USER DORZOLAMIDE 20 mg/ml EYE DROPS SOLUTION (dorzolamide hydrochloride)
Your medicine is called as Dorzolamide 20 mg/ml Eye Drops Solution but will be referred to as Dorzolamide solution throughout the following leaflet.
Read all of this leaflet carefully before you start using this medicine because it contains important information for you.
- Keep this leaflet. You may need to read it again.
- If you have any further questions, ask your doctor or pharmacist.
- This medicine has been prescribed for you only. Do not pass it on to others. It may harm them, even if their signs of illness are the same as yours.
- If you get any side effects, talk to your doctor or pharmacist. This includes any possible side effects not listed in this leaflet. See section 4.
What is in this leaflet:
1. What Dorzolamide solution is and what it is used for
2. What you need to know before you use Dorzolamide solution
3. How to use Dorzolamide solution
4. Possible side effects
5. How to store Dorzolamide solution
6. Contents of the pack and other information
1. WHAT DORZOLAMIDE SOLUTION IS AND WHAT IT IS USED FOR
Dorzolamide solution contains dorzolamide which belongs to a group of medicines called “carbonic anhydrase inhibitors”.
This medicine is prescribed to lower raised pressure in the eye and to treat glaucoma. This medicine can be used alone or in addition to other medicines which lower the pressure in the eye (so-called beta-blockers).
2. WHAT YOU NEED TO KNOW BEFORE YOU USE DORZOLAMIDE SOLUTION
Do not use Dorzolamide solution:
- if you are allergic to dorzolamide hydrochloride or any of the other ingredients of this medicine (listed in section 6).
- if you have severe kidney impairment or problems, or a prior history of kidney stones.
Warnings and precautions
Talk to your doctor or pharmacist before using Dorzolamide solution.
Tell your doctor or pharmacist about any medical problems you have now or have had in the past, including eye problems and eye surgeries, and about any allergies to any medications.
If you develop any eye irritation or any new eye problems such as redness of the eye or swelling of the eyelids, contact your doctor immediately.
If you suspect that Dorzolamide solution is causing an allergic reaction (for example, skin rash, severe skin reaction or itching), stop using this medicine and contact your doctor immediately.
Use in children
Dorzolamide solution has been studied in infants and children less than 6 years of age who have raised pressure in the eye(s) or have been diagnosed with glaucoma. For more information, talk to your doctor.
Use in elderly
In studies with Dorzolamide solution, the effects of this medicine were similar in both elderly and younger patients.
Use in patients with liver impairment
Tell your doctor about any liver problems you now have or have
suffered from in the past.
Other medicines and Dorzolamide solution
Tell your doctor or pharmacist if you are using, have recently used or might use any other medicines (including eye drops). This is particularly important if you are taking another carbonic anhydrase inhibitor such as acetazolamide, or a sulpha drug.
Pregnancy and breast-feeding
Ask your doctor or pharmacist for advice before taking any medicine.
Use in pregnancy
You should not use this medicine during pregnancy. Tell your doctor if you are pregnant or intend to become pregnant.
Use in breast-feeding
If treatment with this medicine is required, breast-feeding is not recommended. Tell your doctor if you are breast-feeding or intend to breast-feed.
Driving and using machines
No studies on the effects on the ability to drive or use machines have been performed. There are side effects associated with Dorzolamide solution, such as dizziness and blurred vision, which may affect your ability to drive and/or operate machinery. Do not drive or operate machinery until you feel well or your vision is clear. Dorzolamide solution contains benzalkonium chloride Dorzolamide solution contains the preservative benzalkonium chloride. This preservative may be deposited in soft contact lenses and may possibly discolour the lenses. If you wear contact lenses, you should consult your doctor before using this medicine.
3. HOW TO USE DORZOLAMIDE SOLUTION
Always use this medicine exactly as your doctor has told you. Check with your doctor or pharmacist if you are not sure. The appropriate dosage and duration of treatment will be established by your doctor.
When this medicine is used alone, the recommended dose is one drop in the affected eye(s) in the morning, in the afternoon and in the evening.
If your doctor has recommended you use this medicine with a beta-blocker eye drop to lower eye pressure, then the recommended dose is one drop of Dorzolamide solution in the affected eye(s) in the morning and in the evening.
If you are using Dorzolamide solution with another eye drop, the drops should be instilled at least 10 minutes apart.
Do not allow the tip of the container to touch the eye or areas around the eye. It may become contaminated with bacteria that can cause eye infections leading to serious damage of the eye, even loss of vision. To avoid possible contamination, wash your hands before using this medicine and keep the tip of the container away from contact with any surface. If you think your medication may be contaminated, or if you develop an eye infection, contact your doctor immediately concerning continued use of this bottle. Instructions for use:
1. Before using the medication for the first time, be sure the Safety Strip on the front of the bottle is unbroken. A gap between the bottle and the cap is normal for an unopened bottle.
Opening Arrows Safety Strip

2. First wash your hands, then tear off the Safety Strip to break the seal.
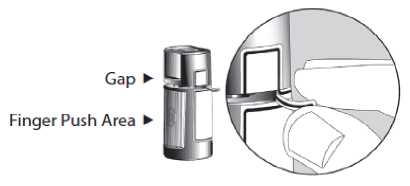
3. To open the bottle, unscrew the cap by turning as indicated by the arrows on the top of the cap. Do not pull the cap directly up and away from the bottle. Pulling the cap directly up will prevent your dispenser from operating properly.
Tilt your head back and pull your lower eyelid down slightly to form a pocket between your eyelid and your eye.
4.
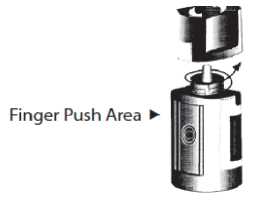
5. Invert the bottle, and press lightly with the thumb or index finger over the “Finger Push Area” (as shown) until a single drop is dispensed into the eye as directed by your doctor.
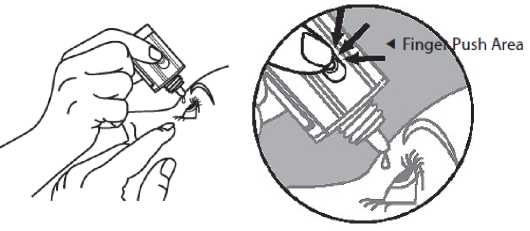
DO NOT TOUCH YOUR EYE OR EYELID WITH THE DROPPER
TIP.
6. If drop dispensing is difficult after opening for the first time, replace the cap on the bottle and tighten (Do not overtighten) and then remove by turning the cap in the opposite directions as indicated by the arrows on the top of the cap.
7. Repeat steps 4 & 5 with the other eye if instructed to do so by your doctor.
8. Replace the cap by turning until it is firmly touching the bottle. The arrow on the left side of the cap must be aligned with the arrow on the left side of the bottle label for proper closure. Do not overtighten or you may damage the bottle and cap.
9. The dispenser tip is designed to provide a single drop; therefore, do NOT enlarge the hole of the dispenser tip.
10. After you have used all doses, there will be some medicine left in the bottle. You should not be concerned since an extra amount of medicine has been added and you will get the full amount of medicine that your doctor prescribed. Do not attempt to remove the excess medicine from the bottle.
If you use more Dorzolamide solution than you should
If you put too many drops in your eye or swallow any of the contents of the container, you should contact your doctor immediately.
If you forget to use Dorzolamide solution
It is important to take this medicine as prescribed by your doctor. If you miss a dose, take it as soon as possible. However, if it is almost time for the next dose, skip the missed dose and go back to your regular dosing schedule.
Do not take a double dose to make up for the forgotten dose.
If you stop using Dorzolamide solution
If you want to stop using this medicine talk to your doctor first. If you have any further questions on the use of this product, ask your doctor or pharmacist.
Rare side effects: (affects 1 to 10 user in 10,000)
Tingling or numbness of the hands or feet, temporary shortsightedness which may resolve when treatment is stopped, development of fluid under the retina (choroidal detachment, following filtration surgery), eye pain, eyelid crusting, low pressure in the eye, swelling of the cornea (with symptoms of visual disturbances), eye irritation including redness, kidney stones, dizziness, nose bleed, throat irritation, dry mouth, localized skin rash (contact dermatitis), severe skin reactions, allergic type reactions such as rash, hives, itching, in rare cases possible swelling of the lips, eyes and mouth, shortness of breath, and more rarely wheezing.
Reporting of side effects
If you get any side effects, talk to your doctor, pharmacist or nurse. This includes any possible side effects not listed in this leaflet. You can also report side effects directly via the yellow card scheme at www.mhra.gov.uk/yellowcard
By reporting side effects you can help provide more information on the safety of this medicine.
5. HOW TO STORE DORZOLAMIDE SOLUTION
• Keep out of the sight and reach of children.
• Store in the original package to protect from light.
• Do not use Dorzolamide solution after the expiry date which is stated on the carton and bottle. The expiry date refers to the last day of that month.
• Dorzolamide solution should be used within 28 days after the bottle is opened.
• If the medicine becomes discoloured or shows any other signs of deterioration, you should seek the advice of your pharmacist who will tell you what to do.
• Medicines should not be disposed of via wastewater or household waste. Ask your pharmacist how to dispose of medicines no longer required. These measures will help to protect the environment.
6. CONTENTS OF THE PACK AND OTHER INFORMATION
What Dorzolamide solution contains
• Each 1ml solution contains 20mg of active ingredient dorzolamide (as 22.26mg dorzolamide hydrochloride).
• The other ingredients are hydroxyethyl cellulose, mannitol, sodium citrate, sodium hydroxide and water for injection. Benzalkonium chloride is added as a preservative.
What Dorzolamide solution looks like and contents of the pack Dorzolamide solution is a clear, colourless to nearly colourless slightly viscous solution. The Ocumeter Plus Ophthalmic Dispenser dropper bottle contains 5ml of solution in single packs. Tamper evidence is provided by a safety strip located on the container label.
Product Licence holder
Procured from within the EU and repackaged by the Product Licence holder: S&M Medical Ltd, Chemilines House, Alperton Lane, Wembley, HA0 1DX.
4. POSSIBLE SIDE EFFECTS
Like all medicines, this medicine can cause side effects, although not everybody gets them.
If you develop allergic reactions including hives, swelling of the face, lips, tongue, and/or throat which may cause difficulty in breathing or swallowing, you should stop using this medicine and seek immediate medical advice.
The following side effects have been reported with Dorzolamide solution either during clinical trials or during post-marketing experience:
Very Common side effects: (affects more than 1 user in 10) Burning and stinging of the eyes.
Common side effects: (affects 1 to 10 users in 100)
Disease of the cornea with sore eye and blurred vision (superficial punctuate keratitis), discharge with itching of the eyes (conjunctivitis), irritation/inflammation of the eyelid, blurred vision, headache, nausea, bitter taste, and fatigue.
Uncommon side effects: (affects 1 to 10 users in 1,000) Inflammation of the iris.
Manufacturer
This product is manufactured by Laboratoires Merck Sharp & Dohme-Chibret, Mirabel Plant, Route de Marsat, Riom 63963 Clermont Ferrand Cedex 9, France.
| POM | PL 19488/1585 Dorzolamide 20 mg/ml Eye Drops Solution
Leaflet revision date: 04 January 2016
S1585 LEAFLET Dorzolamide 20160104