Vallclara 0.02 Mg/3 Mg Film-Coated Tablets
Package leaflet: Information for the patient Vallclara 0.02 mg/3 mg film-coated tablets (ethinylestradiol/drospirenone)
Read all of this leaflet carefully before you start taking this medicine because it contains important information for you.
• Keep this leaflet. You may need to read it again.
• If you have any further questions, ask your doctor, pharmacist or nurse.
• This medicine has been prescribed for you only. Do not pass it on to others. It may harm them even if their signs of illness are the same as yours.
• If you get any side effects, talk to your doctor, pharmacist or nurse. This includes any possible side effects not listed in this leaflet. See section 4.
Important things to know about combined hormonal contraceptives (CHCs):
• They are one of the most reliable reversible methods of contraception if used correctly
• They slightly increase the risk of having a blood clot in the veins and arteries, especially in the first year or when restarting a combined hormonal contraceptive following a break of 4 or more weeks
• Please be alert and see your doctor if you think you may have symptoms of a blood clot (see section 2 “Blood clots”)
What is in this leaflet:
1. What Vallclara is and what it is used for
2. What you need to know before you take Vallclara
3. How to take Vallclara
4. Possible side effects
5. How to store Vallclara
6. Contents of the pack and other information
1. What Vallclara is and what it is used for
• Vallclara is a contraceptive pill and is used to prevent pregnancy.
• Each tablet contains a small amount of two different female hormones, namely drospirenone and ethinylestradiol.
• Contraceptive pills that contain two hormones are called “combination” pills.
2. What you need to know before you take Vallclara
General notes
Before you start using Vallclara you should read the information on blood clots in section 2. It is particularly important to read the symptoms of a blood clot (see section 2 “Blood clots”).
Before you can begin taking Vallclara, your doctor will ask you some questions about your personal health history and that of your close relatives. The doctor will also measure your blood pressure, and, depending upon your personal situation, may also carry out some other tests.
In this leaflet, several situations are described where you should stop using Vallclara, or where the reliability of Vallclara may be decreased.
In such situations you should either not have sex or you should take extra non-hormonal contraceptive precautions, e.g. use a condom or another barrier method. Do not use rhythm or temperature methods. These methods can be unreliable because Vallclara alters the monthly changes of body temperature and of the cervical mucus.
Vallclara, like other hormonal contraceptives, does not protect against HIV infection (AIDS) or any other sexually transmitted disease.
Do not take Vallclara:
You should not use Vallclara if you have any of the conditions listed below. If you do have any of
the conditions listed below, you must tell your doctor. Your doctor will discuss with you what other
form of birth control would be more appropriate.
• if you have (or have ever had) a blood clot in a blood vessel of your legs (deep vein thrombosis, DVT), your lungs (pulmonary embolus, PE) or other organs;
• if you know you have a disorder affecting your blood clotting - for instance, protein C deficiency, protein S deficiency, antithrombin-III deficiency, Factor V Leiden or antiphospholipid antibodies;
• if you need an operation or if you are off your feet for a long time (see section 2 “Blood clots”);
• if you have ever had a heart attack or a stroke;
• if you have (or have ever had) angina pectoris (a condition that causes severe chest pain and may be a first sign of a heart attack) or transient ischaemic attack (TIA - temporary stroke symptoms);
• if you have any of the following diseases that may increase your risk of a clot in the arteries:
- severe diabetes with blood vessel damage
- very high blood pressure
- a very high level of fat in the blood (cholesterol or triglycerides)
- a condition known as hyperhomocysteinaemia
• if you have (or have ever had) a type of migraine called ‘migraine with aura’;
• if you have (or have ever had) a liver disease and your liver function is still not normal
• if your kidneys are not working well (renal failure)
• if you have (or have ever had) a tumour in the liver
• if you have (or have ever had) or if you are suspected of having breast cancer or cancer of the genital organs
• if you have any unexplained bleeding from the vagina
• if you are allergic to ethinylestradiol or drospirenone, or any of the other ingredients of this medicine (listed in section 6). This may cause itching, rash or swelling.
Warnings and precautions
When to take special care with Vallclara
When should you contact your doctor?
Seek urgent medical attention
- if you notice possible signs of a blood clot that may mean you are suffering from a blood clot in the leg (i.e. deep vein thrombosis), a blood clot in the lung (i.e. pulmonary embolism), a heart attack or a stroke (see “Blood clot” section below).
For a description of the symptoms of these serious side effects please go to “How to recognise a blood clot”.
Tell your doctor if any of the following conditions apply to you.
If the condition develops, or gets worse while you are using Vallclara, you should also tell your doctor.
In some situations you need to take special care while using Vallclara or any other combination pill, and your doctor may need to examine you regularly.
Talk to your doctor or pharmacist before taking Vallclara:
• if a close relative has or has ever had breast cancer
• if you have cancer
• if you have a disease of the liver (such as obstruction in bile duct which may cause jaundice and symptoms like itching) or the gallbladder (such as gallstones)
• if you have other kidney problems and are taking medicines which increase potassium levels in the blood
• if you have diabetes
• if you have depression
• if you have Crohn’s disease or ulcerative colitis (chronic inflammatory bowel disease)
• if you have systemic lupus erythematosus (SLE - a disease affecting your natural defence system)
• if you have haemolytic uraemic syndrome (HUS - a disorder of blood clotting causing failure of the kidneys)
• if you have sickle cell anaemia (an inherited disease of the red blood cells)
• if you have elevated levels of fat in the blood (hypertriglyceridaemia) or a positive family history for this condition. Hypertriglyceridaemia has been associated with an increased risk of developing pancreatitis (inflammation of the pancreas)
• if you need an operation, or you are off your feet for a long time (see in section 2 “Blood clots”)
• if you have just given birth you are at an increased risk of blood clots. You should ask your doctor how soon after delivery you can start taking Vallclara
• if you have an inflammation in the veins under the skin (superficial thrombophlebitis);
• if you have varicose veins.
• if you have epilepsy (see "Other medicines and Vallclara")
• if you have a disease that first appeared during pregnancy or earlier use of sex hormones (for example, hearing loss, a blood disease called porphyria, skin rash with blisters during pregnancy (gestational herpes), a nerve disease causing sudden movements of the body (Sydenham's chorea))
• if you have high blood pressure during therapy which is not controlled by treatment with medicine.
• if you have or have ever had chloasma (a discolouration of the skin especially of the face or neck known as “pregnancy patches”). If so, avoid direct sunlight or ultraviolet light while taking this medicine.
• if you have hereditary angioedema, products containing oestrogens may cause or worsen symptoms. You should see your doctor immediately if you experience symptoms of angioedema such as swollen face, tongue and/or throat and/or difficulty swallowing or hives together with difficulty breathing.
BLOOD CLOTS
Using a combined hormonal contraceptive such as Vallclara increases your risk of developing a blood clot compared with not using one. In rare cases a blood clot can block blood vessels and cause serious problems.
Blood clots can develop
■ in veins (referred to as a ‘venous thrombosis’, ‘venous thromboembolism’ or VTE)
■ in the arteries (referred to as an ‘arterial thrombosis’, ‘arterial thromboembolism’ or ATE).
Recovery from blood clots is not always complete. Rarely, there may be serious lasting effects or, very rarely, they may be fatal.
It is important to remember that the overall risk of a harmful blood clot due to Vallclara is small.
HOW TO RECOGNISE A BLOOD CLOT
Seek urgent medical attention if you notice any of the following signs or symptoms.
|
Are you experiencing any of these signs? |
What are you possibly suffering from? |
|
• swelling of one leg or along a vein in the leg or foot especially when accompanied by: • pain or tenderness in the leg which may be felt only when standing or walking • increased warmth in the affected leg • change in colour of the skin on the leg e.g. turning pale, red or blue |
Deep vein thrombosis |
|
• sudden unexplained breathlessness or rapid breathing; • sudden cough without an obvious cause, which may bring up blood; • sharp chest pain which may increase with deep breathing; • severe light headedness or dizziness; • rapid or irregular heart beat • severe pain in your stomach; If you are unsure, talk to a doctor as some of these symptoms such as coughing or being short of breath may be mistaken for a milder condition such as a respiratory tract infection (e.g. a ‘common cold’). |
Pulmonary embolism |
|
Symptoms most commonly occur in one eye: • immediate loss of vision or • painless blurring of vision which can progress to loss of vision |
Retinal vein thrombosis (blood clot in the eye) |
|
• chest pain, discomfort, pressure, heaviness • sensation of squeezing or fullness in the chest, arm or below the breastbone; • fullness, indigestion or choking feeling; • upper body discomfort radiating to the back, jaw, throat, arm and stomach; • sweating, nausea, vomiting or dizziness; • extreme weakness, anxiety, or shortness of breath; • rapid or irregular heart beats |
Heart attack |
|
Are you experiencing any of these signs? |
What are you possibly suffering from? |
|
• sudden weakness or numbness of the face, arm or leg, especially on one side of the body; • sudden confusion, trouble speaking or understanding; • sudden trouble seeing in one or both eyes; • sudden trouble walking, dizziness, loss of balance or | |
|
coordination; | |
|
• sudden, severe or prolonged headache with no known |
Stroke |
|
cause; | |
|
• loss of consciousness or fainting with or without seizure. | |
|
Sometimes the symptoms of stroke can be brief with an almost immediate and full recovery, but you should still seek urgent medical attention as you may be at risk of another stroke. | |
|
• swelling and slight blue discolouration of an extremity; |
Blood clots blocking other |
|
• severe pain in your stomach (acute abdomen) |
blood vessels |
BLOOD CLOTS IN A VEIN
What can happen if a blood clot forms in a vein?
• The use of combined hormonal contraceptives has been connected with an increase in the risk of blood clots in the vein (venous thrombosis). However, these side effects are rare. Most frequently, they occur in the first year of use of a combined hormonal contraceptive.
• If a blood clot forms in a vein in the leg or foot it can cause a deep vein thrombosis (DVT).
• If a blood clot travels from the leg and lodges in the lung it can cause a pulmonary embolism
(PE).
• Very rarely a clot may form in a vein in another organ such as the eye (retinal vein thrombosis).
When is the risk of developing a blood clot in a vein highest?
The risk of developing a blood clot in a vein is highest during the first year of taking a combined hormonal contraceptive for the first time. The risk may also be higher if you restart taking a combined hormonal contraceptive (the same product or a different product) after a break of 4 weeks or more
After the first year, the risk gets smaller but is always slightly higher than if you were not using a combined hormonal contraceptive.
When you stop Vallclara your risk of a blood clot returns to normal within a few weeks.
What is the risk of developing a blood clot?
The risk depends on your natural risk of VTE and the type of combined hormonal contraceptive you are taking.
The overall risk of a blood clot in the leg or lung (DVT or PE) with Vallclara is small.
- Out of 10,000 women who are not using any combined hormonal contraceptive and are not pregnant, about 2 will develop a blood clot in a year.
- Out of 10,000 women who are using a combined hormonal contraceptive that contains levonorgestrel, norethisterone, or norgestimate about 5-7 will develop a blood clot in a year.
- Out of 10,000 women who are using a combined hormonal contraceptive that contains drospirenone, such as Vallclara, between about 9 and 12 women will develop a blood clot in a year.
- The risk of having a blood clot will vary according to your personal medical history (see “Factors that increase your risk of a blood clot” below).
|
Risk of developing a blood clot in a year | |
|
Women who are not using a combined hormonal pill/patch/ring and are not pregnant |
About 2 out of 10,000 women |
|
Women using a combined hormonal contraceptive pill containing levonorgestrel, norethisterone or norgestimate |
About 5-7 out of 10,000 women |
|
Women using Vallclara |
About 9-12 out of 10,000 women |
Factors that increase your risk of a blood clot in a vein
The risk of a blood clot with Vallclara is small but some conditions will increase the risk. Your risk
is higher:
• if you are very overweight (body mass index or BMI over 30kg/m2);
• if one of your immediate family has had a blood clot in the leg, lung or other organ at a young age (e.g. below the age of about 50). In this case you could have a hereditary blood clotting disorder;
• if you need to have an operation, or if you are off your feet for a long time because of an injury or illness, or you have your leg in a cast. The use of Vallclara may need to be stopped several weeks before surgery or while you are less mobile. If you need to stop Vallclara ask your doctor when you can start using it again.
• as you get older (particularly above about 35 years);
• if you gave birth less than a few weeks ago
The risk of developing a blood clot increases the more conditions you have.
Air travel (>4 hours) may temporarily increase your risk of a blood clot, particularly if you have
some of the other factors listed.
It is important to tell your doctor if any of these conditions apply to you, even if you are unsure. Your doctor may decide that Vallclara needs to be stopped.
If any of the above conditions change while you are using Vallclara, for example a close family member experiences a thrombosis for no known reason; or you gain a lot of weight, tell your doctor.
BLOOD CLOTS IN AN ARTERY
What can happen if a blood clot forms in an artery?
Like a blood clot in a vein, a clot in an artery can cause serious problems. For example, it can cause a heart attack or a stroke.
Factors that increase your risk of a blood clot in an artery
It is important to note that the risk of a heart attack or stroke from using Vallclara is very small but can increase:
• with increasing age (beyond about 35 years);
• if you smoke. When using a combined hormonal contraceptive like Vallclara you are advised to stop smoking. If you are unable to stop smoking and are older than 35 your doctor may advise you to use a different type of contraceptive;
• if you are overweight;
• if you have high blood pressure;
• if a member of your immediate family has had a heart attack or stroke at a young age (less than about 50). In this case you could also have a higher risk of having a heart attack or stroke;
• if you, or someone in your immediate family, have a high level of fat in the blood (cholesterol or triglycerides);
• if you get migraines, especially migraines with aura;
• if you have a problem with your heart (valve disorder, disturbance of the rhythm called atrial fibrillation)
• if you have diabetes.
If you have more than one of these conditions or if any of them are particularly severe the risk of developing a blood clot may be increased even more.
If any of the above conditions change while you are using Vallclara, for example you start smoking, a close family member experiences a thrombosis for no known reason; or you gain a lot of weight, tell your doctor.
Vallclara and cancer
Breast cancer has been observed slightly more often in women using combination pills, but it is not known whether this is caused by the treatment. For example, it may be that more tumours are detected in women on combination pills because they are examined by their doctor more often. The occurrence of breast tumours becomes gradually less after stopping the combination hormonal contraceptives. It is important to regularly check your breasts and you should contact your doctor if you feel any lump.
In rare cases, benign liver tumours, and in even fewer cases malignant liver tumours have been reported in pill users. Contact your doctor if you have unusually severe abdominal pain, abdominal swelling (which may be due to enlargement of liver) or bleeding from your stomach.
Bleeding between periods
During the first few months that you are taking Vallclara you may have unexpected bleeding (bleeding outside the gap week). If this bleeding occurs for more than a few months, or if it begins after some months, your doctor must find out what is wrong.
What to do if no bleeding occurs during the gap week
If you have taken all the tablets correctly, have not had vomiting or severe diarrhoea and you have not taken any other medicines, it is highly unlikely that you are pregnant.
If the expected bleeding does not happen twice in succession, you may be pregnant. Contact your doctor immediately. Do not start the next strip until you are sure that you are not pregnant.
Other medicines and Vallclara
Tell your doctor which medicines or herbal products you are taking, have recently taken or might take. Also tell any other doctor or dentist who prescribes another medicine (or the pharmacist) that you use Vallclara. They can tell you if you need to take additional contraceptive precautions (for example condoms) and if so, for how long._
Some medicines can have an influence on the blood levels of Vallclara and can make it less effective in preventing pregnancy, or can cause unexpected bleeding. These include:
• medicines used for the treatment of
o epilepsy (e.g. primidone, phenytoin, barbiturates, carbamazepine, oxcarbazepine, felbamate, topiramate)
o tuberculosis (e.g. rifampicin)
o HIV and Hepatitis C virus infections (so-called protease inhibitors and non-nucleoside reverse transcriptase inhibitors, such as ritonavir, nevirapine, efavirenz) o fungal infections (griseofulvin or ketoconazole) o arthritis, arthrosis (etoricoxib)
o high blood pressure in the blood vessels in the lungs (bosentan)
• the herbal remedy St John’s wort
If you are taking any of the above mentioned medicines along with Vallclara you should take additional contraceptive precautions (for example condoms) during and for 28 days after the therapy.
Vallclara may influence the effect of other medicines, e.g.
• medicines containing ciclosporin
• the anti-epileptic lamotrigine (this could lead to an increased frequency of seizures)
• theophylline (used to treat breathing problems) • tizanidine (used to treat muscle pain and/or muscle cramps)
Y our doctor may monitor your blood potassium levels if you are taking certain medicines to treat heart problems (such as water tablets).
Laboratory tests
If you need a blood test, tell your doctor or the laboratory staff that you are taking the pill, because hormone contraceptives can affect the results of some tests.
Pregnancy and breast-feeding
Pregnancy
If you are pregnant, do not take Vallclara. If you become pregnant while taking Vallclara stop immediately and contact your doctor. If you want to become pregnant, you can stop taking Vallclara at any time (see also “If you want to stop taking Vallclara”).
Breast-feeding
Use of Vallclara is generally not advisable when a woman is breast-feeding. If you want to take the pill while you are breast-feeding you should contact your doctor.
Driving and using machines
There is no information suggesting that use of Vallclara affects driving or use of machines. Vallclara contains lactose.
If you have been told by your doctor that you have an intolerance to some sugars, contact your doctor before taking this medicine.
3. How to take Vallclara
Always take this medicine exactly as your doctor or pharmacist has told you. Check with your doctor or pharmacist if you are not sure.
Take one tablet of Vallclara every day, if necessary with a small amount of water. You may take the tablets with or without food, but you should take the tablets every day around the same time.
The strip contains 21 tablets. Next to each tablet is printed the day of the week that it should be taken. If, for example you start on a Wednesday, take a tablet with "WED" next to it. Follow the direction of the arrow on the strip until all 21 tablets have been taken.
Then take no tablets for 7 days. In the course of these 7 tablet-free days (otherwise called a stop or gap week) bleeding should begin. This so-called “withdrawal bleeding”, usually starts on the 2nd or 3rd day of the gap week.
On the 8th day after the last Vallclara tablet (that is, after the 7-day gap week), you should start with the following strip, whether your bleeding has stopped or not. This means that you should start every strip on the same day of the week and that the withdrawal bleed should occur on the same days each month.
If you use Vallclara in this manner, you are also protected against pregnancy also during the 7 days when you are not taking a tablet.
When can you start with the first strip?
• If you have not used a contraceptive with hormones in the previous month
Begin with Vallclara on the first day of your usual cycle (that is, the first day of your period). If you start Vallclara on the first day of your period you are immediately protected against pregnancy. You may also begin on day 2-5 of the cycle, but then you must use extra protective measures (for example, a condom) for the first 7 days.
• Changing from a combination hormonal contraceptive, or combination contraceptive vaginal ring or patch
You should start taking Vallclara preferably on the day after the last active tablet (the last tablet containing active substances) of your previous pill, but at the latest on the day after the tablet-free days of your previous pill (or after the last inactive tablet of your previous pill). When changing from a combination contraceptive vaginal ring or patch, you should start taking Vallclara preferably on the day of removal, but at the latest when the next application would have been due.
• Changing from a progestogen-only-method (progestogen-only pill, injection, implant or a progestogen-releasing IUD)
You may switch any day from the progestogen-only pill however, (from an implant or an IUD you should switch on the day of its removal, from an injectable when the next injection would be due) but in all of these cases use extra protective measures (for example, a condom) for the first 7 days of tablet-taking.
• After a miscarriage or termination of your pregnancy Follow the advice of your doctor.
• After having a baby
You can start Vallclara between 21 and 28 days after having a baby. If you start later than day 28, use a so-called barrier method of contraception (for example, a condom) during the first 7 days of Vallclara use.
If, after having a baby, you have had sex before starting Vallclara, be sure that you are not pregnant or wait until your next period.
• If you are breast-feeding and want to start Vallclara (again) after having a baby Read the section on "Breast-feeding".
Ask your doctor what to do if you are not sure when to start.
If you take more Vallclara than you should
There are no reports of serious harmful results of taking too many Vallclara tablets.
If you take several tablets at once then you may have signs of overdose such as nausea or vomiting. Young girls may have bleeding from the vagina.
If you have taken too many Vallclara tablets, or you discover that a child has taken some, ask your doctor or pharmacist for advice.
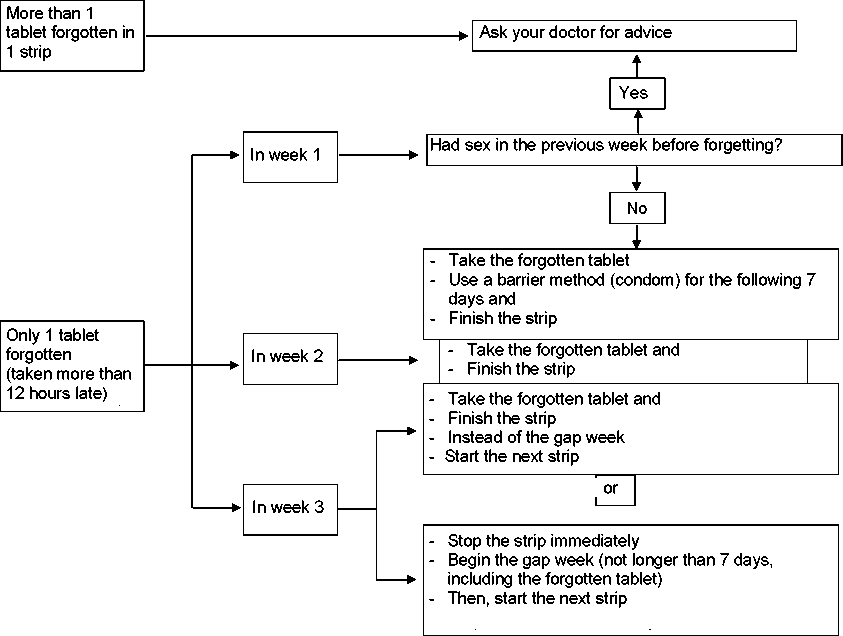
If you forget to take Vallclara
• If you are less than 12 hours late taking a tablet, the protection against pregnancy is not reduced. Take the tablet as soon as you remember and then take the following tablets again at the usual time.
• If you are more than 12 hours late taking a tablet, the protection against pregnancy may be reduced. The greater the number of tablets that you have forgotten, the greater is the risk of becoming pregnant.
The risk of incomplete protection against pregnancy is greatest if you forget a tablet at the beginning or at the end of the strip. Therefore, you should keep to the following rules (see the diagram below):
• More than one tablet forgotten in this strip
Contact your doctor.
• One tablet forgotten in week 1
Take the forgotten tablet as soon as you remember, even if that means you have to take two tablets at the same time. Continue taking the tablets at the usual time and use extra precautions for the next 7 days, for example, a condom. If you have had sex in the week before forgetting the tablet you should contact your doctor as there is a chance you may have become pregnant.
• One tablet forgotten in week 2
Take the forgotten tablet as soon as you remember, even if that means you have to take two tablets at the same time. Continue taking the tablets at the usual time. The protection against pregnancy is not reduced, and you do not need to take extra precautions.
• One tablet forgotten in week 3
You can choose between two possibilities:
1. Take the forgotten tablet as soon as you remember, even if that means that you have to take two tablets at the same time. Continue taking the tablets at the usual time. Instead of taking the tablet-free period start the next strip.
Most likely, you will have a period at the end of the second strip but you may have light or menstruation-like bleeding during the second strip.
2. You can also stop the strip and go directly to the tablet-free period of 7 days (including the days you missed tablets, record the day on which you forgot your tablet). If you want to start a new strip on the day you always start, make the tablet-free period less than 7 days.
If you follow one of these two recommendations, you will remain protected against pregnancy.
If you have forgotten any of the tablets in a strip, and you do not have a bleeding during the first tablet-free period, you may be pregnant. Contact your doctor before you start the next strip.
The following diagram describes how to proceed if you forget to take your tablet(s):
What to do in the case of vomiting or severe diarrhoea
If you vomit within 3-4 hours after taking a tablet or you have severe diarrhoea, there is a risk that the active substances in the pill will not be fully taken up by your body in which case you may need to use extra protection (such as a condom) to avoid pregnancy. The situation is almost the same as forgetting a tablet. After vomiting or diarrhoea, take another tablet from a reserve strip as soon as possible. If possible take it within 12 hours of when you normally take your pill. If that is not possible or 12 hours have passed, you should follow the advice given under "If you forget to take Vallclara".
Delaying your period: what you need to know
Even though it is not recommended, you can delay your period by going straight to a new strip of Vallclara instead of the tablet-free period and finishing it. You may experience light or menstruation-like bleeding while using this second strip. After the second strip is finished, take the usual tablet-free period of 7 days, and start the next strip.
You should ask your doctor for advice before deciding to delay your menstrual period.
Changing the first day of your period: what you need to know
If you take the tablets according to the instructions, then your period will begin during the tablet-free week. If you have to change this day, reduce the number of tablet-free days (but never increase them - 7 is the maximum!). For example, if your tablet-free days normally begin on a Friday, and you want to change this to a Tuesday (3 days earlier) start a new strip 3 days earlier than usual. If you make the tablet-free interval very short (for example, 3 days or less) you may not have any bleeding during these days. You may then experience light or menstruation-like bleeding.
If you are not sure what to do, consult your doctor.
If you stop taking Vallclara
You can stop taking Vallclara whenever you want. If you do not want to become pregnant, ask your doctor for advice about other reliable methods of birth control. If you want to become pregnant, stop taking Vallclara and wait for a period before trying to become pregnant. You will be able to calculate the expected delivery date more easily.
If you have any further questions on the use of this medicine, ask your doctor or pharmacist.
4. Possible side effects
Like all medicines, this medicine can cause side effects, although not everybody gets them. If you get any side effects, particularly if severe and persistent, or have any change to your health that you think may be due to Vallclara, please talk to your doctor.
An increased risk of blood clots in your veins (venous thromboembolism (VTE)) or blood clots in your arteries (arterial thromboembolism (ATE)) is present for all women taking combined hormonal contraceptives. For more detailed information on the different risks from taking combined hormonal contraceptives please see section 2 “What you need to know before you take Vallclara”.
If any of the following side effects happen, you may need urgent medical attention. Stop taking Vallclara and contact a doctor or go to the nearest hospital immediately.
Uncommon (may affect up to 1 in 100 people)
- sudden swelling of the skin and/or mucous membranes (e.g. tongue or throat), and/or difficulty swallowing or hives together with difficulty breathing (angioedema)
- harmful blood clots in a lung (i.e. pulmonary embolism (PE))
Rare (may affect up to 1 in 1,000 people)
- harmful blood clots in a vein or artery for example:
o in a leg or foot (i.e. deep vein thrombosis (DVT))
o heart attack
o stroke
o mini-stroke or temporary stroke-like symptoms, known as a transient ischaemic attack
(TIA)
o blood clots in the liver, stomach/intestine, kidneys or eye.
The chance of having a blood clot may be higher if you have any other conditions that increase this risk (See section 2 for more information on the conditions that increase risk for blood clots and the symptoms of a blood clot)
Conditions that may occur or worsen during pregnancy or previous use of the pill include:
- systemic lupus erythematosus (SLE, a disease affecting the immune system)
- inflammation of the colon or other parts of the intestine (with signs such as bloody diarrhoea, pain when passing stools, pain in the abdomen) (Crohn’s disease and ulcerative colitis)
- epilepsy
- uterine myoma (non-cancerous tumour that grows within the muscle tissue of the uterus)
- a blood pigment disorder (porphyria)
- blister-like rash (herpes gestationis) whilst pregnant
- Sydenham’s chorea (a disease of the nerves in which sudden movements of the body occur)
- a certain blood disorder that causes kidney damage (haemolytic uraemic syndrome, with signals such as decreased urine output, blood in the urine, low red blood cells, nausea, vomiting, confusion and diarrhoea)
- yellowing of the skin or white of the eyes due to obstruction in the bile duct (chloestatic jaundice)
Also, breast cancer (see section 2 “Vallclara and cancer”) and non-cancerous (benign) and cancerous (malignant) liver tumours (with signs such as swollen abdomen, weight loss, abnormal liver function which may be seen in blood tests) and chloasma (yellow brown patches on the skin and particularly the face, so called “pregnancy patches”), which may be permanent, especially in women who have previously had chloasma during pregnancy, have been observed.
Other possible side effects
Common (may affect up to 1 in 10 people):
- mood swings
- headache
- abdominal pain (stomach ache)
- acne
- breast pain, breast enlargement, breast tenderness, painful or irregular periods
- weight gain
Uncommon (may affect up to 1 in 100 people):
- Candida (fungal infection)
- cold sores (herpes simplex)
- allergic reactions
- increased appetite
- depression, nervousness, sleep disorder
- feeling of “pins and needles”, giddiness (vertigo)
- problems with vision
- irregular heart beat or unusually fast heart rate
- high blood pressure, low blood pressure, migraine, varicose veins
- sore throat
- nausea, vomiting, inflammation of stomach and/or intestine, diarrhoea, constipation
- hair loss (alopecia), eczema, itching, rashes, dry skin, oily skin disorders (seborrheic dermatitis)
- neck pain, limb pain, muscle cramps
- bladder infection
- breast lump (benign), milk production while not pregnant (galactorrhea), ovarian cysts, hot flushes, absence of periods, very heavy periods, vaginal discharge, vaginal infection or inflammation, vaginal dryness, lower abdominal (pelvic) pain, abnormal cervical smear (Papanicolaou or Pap smear), decreased interest in sex
- fluid retention, lack of energy, excessive thirst, increased sweating
- weight loss
- general pain
Rare (may affect up to 1 in 1,000 people):
- asthma
- hearing impairment
- erythema nodosum (characterised by painful reddish skin nodules)
- erythema multiforme (rash with target-shaped reddening or sores)
Reporting of side effects
If you get any side effects, talk to your doctor or pharmacist. This includes any possible side effects not listed in this leaflet. You can also report side effects directly via the Yellow Card Scheme, at www.mhra.gov.uk/yellowcard. By reporting side effects you can help provide more information on the safety of this medicine.
5. How to store Vallclara
Keep this medicine out of the sight and reach of children.
This medicine does not require any special storage conditions.
Do not use this medicine after the expiry date which is stated on the blister/carton after "Do not use after:" or "EXP:". The expiry date refers to the last day of that month.
Do not throw away any medicines via wastewater or household waste. Ask your pharmacist how to throw away medicines you no longer use. These measures will help protect the environment.
6. Contents of the pack and other information What Vallclara contains
The active substances are ethinylestradiol 0.02 mg and drospirenone 3 mg.
The other ingredients are:
Tablet core: lactose monohydrate, pregelatinised starch (maize), povidone, croscarmellose sodium, polysorbate 80, magnesium stearate.
Coating: Polyvinyl alcohol partial hydrolysed, titanium dioxide (E171), macrogol 3350, talc, yellow iron oxide (E172), red iron oxide (E172), black iron oxide (E172).
What Vallclara looks like and contents of the pack
Tablets are pink, round film-coated tablets.
Vallclara is available in boxes of 1, 2, 3, 6 and 13 blisters, each one containing 21 tablets.
Not all pack sizes may be marketed
Marketing Authorisation Holder
DB Ashbourne Limited
The Rectory, Braybrooke Road
Arthingworth, Market Harborough
LE16 8JT, United Kingdom Telephone: (01858) 525643 Fax:(01858)525383 Email: enquiries@medinformation.co.uk
Manufacturer
Laboratorios Leon Farma, S.A.
Pol. Ind. Navatejera.
C/ La Vallina s/n
24008 - Villaquilambre, Leon.
Spain
This medicinal product is authorised in the Member States of the EEA under the following names:
France
Germany
Italy
Norway
Sweden
The Netherlands United Kingdom
DROSPIRENONE / ETHINYLESTRADIOL MYLAN 3 mg/0,02 mg, comprime pellicule
Georgette-20 3 mg/0,02 mg Filmtabletten ETINILESTRADIOLO E DROSPIRENONE DOC Iren
Vallclara 0,02 mg/3 mg filmdragerade tabletter Vallclara 0,02 mg/3 mg, filmomhulde tabletten Vallclara 0.02 mg/3 mg film-coated tablets
This leaflet was last revised in: December 2015
18