Vancomycin 1000Mg Powder For Concentrate For Solution For Infusion
Out of date information, search another111111111

Vancomycin 500mg and 1000mg
Powder for Concentrate for Solution for Infusion
Read all of this leaflet carefully before you start using this
• Keep this leaflet. You may need to read it again.
• If you have any further questions, ask your doctor or pharmacist.
• This medicine has been prescribed for you. Do not pass it on to others. It may harm them, even if their symptoms are the same as yours.
• If any of the side effects get serious, or if you notice any side effects not listed in this leaflet, please tell your doctor or pharmacist.
• The full name of this medicine is Vancomycin 500mg and 1000mg Powder for Concentrate for Solution for Infusion but within the leaflet it will be referred to as Vancomycin powder.
What Vancomycin powder is and what it is used for
Read this before you are given Vancomycin powder
How Vancomycin powder is stored
Q What Vancomycin powder is and what it is used for
Vancomycin powder is one of a group of medicines called glycopeptide antibiotics. These are used to treat infections caused by bacteria. Vancomycin comes in the form of a powder which is made into a solution using sterile water (for injections).
This solution is given to you as an infusion, a slow injection, by means of a drip. It will only be given to you through a vein. It can also be given by mouth.
What Vancomycin powder is used for
This medicine is used for severe infections caused by bacteria which can resist other antibiotics. It is used in patients who have not responded to treatment with, or have had a bad reaction to, other antibiotics.
It is used to treat various severe infections of the lining or valves of the heart, lungs, bone or soft tissue (flesh). It can also be given to you before some surgical procedures to prevent infections.
0 Read this before you are given Vancomycin powder
Do not have Vancomycin powder if you
• are allergic to vancomycin hydrochloride or to any of the other ingredients of Vancomycin powder (See Section 6 for a list of these).
Tell your doctor if you have had any problems with this medicine or any other in the past.
Take special care with Vancomycin powder if you
• suffer from loss of hearing
• have kidney problems
• are elderly (65 years old and over)
Rapid injection of Vancomycin may cause low blood pressure, shock and rarely cardiac arrest. Stopping the infusion usually results in a prompt cessation of the reactions.
Injection site pain, inflammation of the vein wall and blood clotting can occur and is occasionally severe, slow administration also reduces these side effects.
If you are allergic to another antibiotic called teicoplanin you may also be allergic to vancomycin. Please tell your doctor.
If you suffer from kidney failure or receive concomitant treatment with other substances toxic to kidney the possibility of developing toxic effects is much higher.
Your doctor may perform several tests to see if your kidneys and liver are working properly.
If you are elderly or have kidney problems your doctor may also perform regular tests on your hearing and measure the amount of vancomycin in your blood.
Deafness, transitory or permanent, which may be preceded by noises in ears, can occur in patients with prior deafness, who have received excessive doses, or who receive treatment with another substance toxic to hearing. To reduce this risk, blood levels should be checked periodically and periodic testing of hearing function is recommended.
Prolonged use of Vancomycin powder may result in the overgrowth of resistant organisms; your doctor will monitor this.
Taking other medicines with Vancomycin powder
Tell your doctor if you are also taking:
• gentamycin (antibiotic)
• amphotericin B (antibiotic)
• streptomycin (antibiotic)
• neomycin (antibiotic)
• kanamycin (antibiotic)
• amikacin (antibiotic)
• tobramycin (antibiotic)
• bacitracin (antibiotic)
• polymixin B (antibiotic)
• colistin (antibiotic)
• viomycin (antibiotic)
• cisplatin (medicinal product used to treat some types of cancer)
The following can also react with vancomycin if taken at the same time:
• anaesthetic agents (if you are going to have a general anaesthetic)
• muscle relaxants (used sometimes during a general anaesthetic)
Please tell your doctor or nurse, if you are taking or have taken recently any other medicines, including medicines obtained without a prescription.
Ask your doctor for advice before taking any medicine.
Tell your doctor if you are pregnant or think you may be pregnant. Your doctor will then decide whether you should receive Vancomycin powder.
Tell your doctor if you are breast-feeding, because Vancomycin passes into breast milk. A decision will be made as to whether you breast-feed or are treated with Vancomycin powder.
Vancomycin powder has very little influence on the ability to drive or to use machinery.
Important information about some of the ingredients of Vancomycin powder
This medicinal product contains less than 1mmol sodium (23mg) per vial, i.e. essentially 'sodium-free'.
Continued top of next column
AAAG0244 Continued on next page
The following information is intended for medical or healthcare professionals only:
Vancomycin powder for concentrate for infusion is for single use only and any unused solution should be discarded.
For intravenous use the powder must be reconstituted and the resulting concentrate must then be immediately diluted further prior to use.
For oral use the powder must be reconstituted, the resulting concentration may be stored in a refrigerator (2 - 8°C) for 96 hours. Discard any unused solution.
Preparation of the reconstituted concentrate
Dissolve the content of each 500mg vial in 10ml of sterile water for injections.
Dissolve the content of each 1000mg vial in 20ml of sterile water for injections.
One ml of reconstituted solution contains 50mg of vancomycin. pH of the reconstituted solution is 2.5 to 4.5. The solution should be clear colourless to pale yellow and free from fibre and visible particulate matters.
Preparation of final diluted Solution for infusion
Reconstituted concentrate containing 50mg/ml of vancomycin should be further diluted depending on the method of administration. Suitable diluents are: 5% Glucose Injection or 0.9% Sodium Chloride Injection.
Intermittent infusion:
Reconstituted concentrate containing 500mg of vancomycin (50mg/ml) must be diluted further with at least 100ml diluent. Reconstituted concentrate containing 1000mg vancomycin (50mg/ml) must be diluted further with at least 200ml diluent.
The concentration of vancomycin in Solution for infusion should not exceed 5mg/ml.
The desired dose should be administered slowly by intravenous infusion at a rate of no more than 10mg/minute, for at least 60 minutes or even longer.
Before administration, the reconstituted and diluted solutions should be inspected visually for particulate matter and discoloration. Only clear and colourless to pale yellow solution free from particles should be used.
AAAG0244
0 How to use Vancomycin powder
Vancomycin powder is given to you by hospital staff, using an infusion, (a slow injection by means of a drip). Each infusion will be given slowly, usually lasting for at least one hour. It may also be given to you to drink or via a tube in your nose.
The dose of Vancomycin powder your doctor gives you will depend on your age, weight, general health condition, the severity of the infection, whether you need certain other medicines and how well you respond to the treatment.
For patients whose kidneys are working normally Adults and children above 12 years of age: the usual dose is 2000mg daily in two or four doses (or 30mg per kilogram of bodyweight, per day either 500mg every 6 hours or 1g every 12 hours).
Children (from one month to 12 years of age): the usual intravenous dosage is 10mg/kg per dose given every 6 hours (total daily dosage 40mg/kg of body weight).
New-born babies (born full term):
• 0 - 7 days old: a starting dose of 15mg for each kilogram of the child's weight, followed by 10mg per kilogram, every 12 hours.
• 7 - 30 days old: a starting dose of 15mg for each kilogram of the child's weight, followed by 10mg per kilogram, every 8 hours.
For patients whose kidneys are not working normally
The doctor will reduce the dose or extend the interval between two doses.
Special tests will be carried out and the dose will be adjusted to meet the results of the tests.
If you are elderly, (65 years of age and over), your doctor will also consider how well your kidneys are likely to be working.
For patients whose liver is not working normally
If you have severe liver damage, special tests will be carried out and the dose will be adjusted to meet the results obtained from the tests.
For patients whose kidneys do not work at all
The starting dose is 15mg for each kilogram of bodyweight, followed by a maintenance dose of approximately 1.9mg for each kilogram of bodyweight, over each 24 hours.
Adults and elderly: 500mg a day in divided doses for 7-10 days Children: 40mg for each kilogram of bodyweight, in 3-4 doses for 7-10 days
Maximum daily dose of2000mg
Your doctor will decide when your treatment should end.
If you receive too much Vancomycin
Your doctor monitors the amount of Vancomycin you receive. If the regular blood tests and other tests show that you have too much in your body, the amount of Vancomycin will be reduced or infusion will be stopped. The level remaining in your blood will be lowered.
If you have any further questions about receiving this medicine, please ask your doctor.
Like all medicines, Vancomycin powder can cause side effects, although
not everybody gets them.
• Contact your doctor at once if the following effects happen:swelling of the face or throat, difficulty in breathing, feeling faint, itchy skin or hives. The consequences could become very serious so, tell your doctor or nurse immediately. Infusion will be halted.
• Vancomycin may cause a serious or life-threatening allergic reaction that may affect your skin or other parts of your body such as your liver or blood cells (DRESS syndrome). Symptoms include skin rash, fever, swollen glands that do not go away, swelling of your lip and tongue, yellowing of your skin or of the whites of the eyes, unusual bruising or bleeding, severe fatigue or weakness, unexpected muscle pain or frequent infections
• During or shortly after rapid infusion low blood pressure, difficulty breathing, itchy skin rash, redness of the skin of the upper body, pain and cramp in chest or back muscle can occur. Vancomycin is given slowly (for more than 60 minutes) to avoid these reactions.
Tell your doctor or pharmacist if the following side effects occur or get
worse:
Common side effects (affect 1 to 10 users in 100):
• shortness of breath; noisy breathing (stridor)
• low blood pressure
• vein wall inflammation including blood clotting (thrombophlebitis)
• kidney problems
• skin reactions such as rashes, swelling, itching or hives
• redness, a burning sensation, swelling of a vein and the area around it
• redness and soreness at the point where the infusion goes into your body
• redness of the upper body and the face
• pain and spasm of the chest and back muscles Uncommon side effects (affect 1 to 10 users in 1000):
• temporary or permanent loss of hearing
Rare side effects (affect 1 to 10 users in 10,000):
• anaphylactic reactions, allergic reactions
• a ringing or buzzing in your ears
• dizziness
• feeling sick
• diarrhoea
• raised temperature or shivering
• changes to the number of various types of white blood cells in the blood - an increase or decrease
• a decrease in the number of platelets (a type of blood cell important in blood clotting) in the blood
• inflammation of the kidneys
• acute kidney failure
Very rare side effects (affect less than 1 user in 10,000):
• cardiac arrest
• inflammation of the bowel which causes abdominal pain or bloody diarrhoea
• severe skin reactions such as red and scaly or blistering skin, lesions and flu-like symptoms
• inflammation of the blood vessels
0 How Vancomycin powder is stored
Keep out of the reach and sight of children.
Do not use Vancomycin powder after the expiry date which is stated on the carton and vial after EXP. The expiry date refers to the last day of that month.
Keep the vial in the outer carton in order to protect from light. Store below 25°C.
Medicines should not be disposed of via wastewater or household waste. Ask your pharmacist how to dispose of medicines no longer required. These measures will help to protect the environment.
What Vancomycin powder contains
• The active substance is vancomycin . Each vial contains either 500mg vancomycin (equivalent to 525,000 IU) (as vancomycin hydrochloride) or 1000mg vancomycin (equivalent to 1,050,000 lU) (as vancomycin hydrochloride).
• The other ingredients are sodium hydroxide, hydrochloric acid.
What Vancomycin powder looks like and the contents of the pack
Vancomycin powder is a freeze-dried, off-white powder. It is vacuum-packed in a glass vial with a rubber stopper and an aluminium seal with flip-offcap.
Pack sizes: 1 vial in a carton
Marketing Authorisation Holder:
Actavis Group PTC ehf., Reykjavikurvegur 76-78, 220 Hafnarfjordur, Iceland
Manufacturers:
Agila Specialties Polska Sp. z o.o.
10, Daniszewska Str.
03-230 Warsaw Poland
Actavis Group PTC ehf,
Reykjavikurvegur 76-78,
IS-220 Hafnarfjordur,
Iceland
This leaflet was last revised in October 2013.
If you would like a leaflet with larger text, please contact
actav/s
Continued top of next column Actavis, Barnstaple, EX32 8NS, UK
AAAG0244 101xxxx

01271 311257.
Please note: 101xxxx is a supplier code that will be updated by the supplier when printed.
Shelf-life of reconstituted concentrate:
The reconstituted concentrate should be diluted immediately after preparation.
Shelf-life of diluted product:
Chemical and physical in-use stability of the diluted product has been demonstrated for 48 hours at both 2-8 °C and 25°C when diluted with either 0.9% sodium chloride or 5% glucose.
From a microbiological point of view, the medicinal product should be used immediately unless reconstitution and dilution has taken place in controlled and validated aseptic conditions. If not used immediately, in-use storage times and conditions prior to use are the responsibility of the user and the product should be protected from light during storage.
In patients with impaired renal function the dose must be adjusted. Serum levels of vancomycin should be monitored regularly. For most patients with impaired renal function the following nomogramm can be used to determine the dose needed. The total daily dose of vancomycin (in mg) should be about 15 times the glomerular filtration rate (in ml/min). The starting dose should always be at least 15mg/kg. The nomogram is not valid for functionally anephric patients on dialysis.
AAAG0244
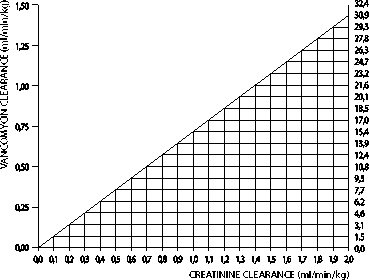
If the creatine clearance is not available, the following formula may be applied to calculate the creatinine clearance from the patient's age, sex and serum creatinine:
Men: Weight (kg) x 140 - age (years)
72 x serum creatinine (mg/100ml)
Women: 0.85 x value calculated by the above formula.
^actavis
Actavis, Barnstaple, EX32 8NS, UK