Zaditen 0.25Mg/Ml Eye Drops Solution
PACKAGE LEAFLET: INFORMATION FOR THE USER
ZADITEN
0. 25.mg/ml, eye drops, solution
Ketotifen
Read all of this leaflet carefully before you start using this medicine.
• Keep this leaflet. You may need to read it again.
• If you have any further questions, ask your doctor or pharmacist.
• This medicine has been prescribed for you. Do not pass it on to others. It may harm them, even if their symptoms are the same as yours.
• If any of the side effects gets serious, or if you notice any side effects not listed in this leaflet, please tell your doctor or pharmacist.
In this leaflet:
1. What Zaditen is and what it is used for
2. Before you use Zaditen
3. How to use Zaditen
4. Possible side effects
5. How to store Zaditen
6. Further information
1. WHAT ZADITEN IS AND WHAT IT IS USED FOR
Zaditen contains the active substance ketotifen, which is an anti-allergic substance. Zaditen is used to treat eye symptoms of hay fever.
2. BEFORE YOU USE ZADITEN
Do not use Zaditen
If you are allergic (hypersensitive) to ketotifen or any of the other ingredients of Zaditen. Using other medicines
If you need to apply any other medicinal products to your eyes together with Zaditen, wait at least 5 minutes between applying each product.
Please tell your doctor or pharmacist if you are taking or have recently taken any other medicines, including medicines obtained without a prescription. This is particularly important for medicines which are used to treat:
• depression
• allergy (e.g. antihistamines)
Using Zaditen with food and drink
Zaditen may increase the effect of alcohol.
Pregnancy and breast-feeding
If you are pregnant or think you might be, ask your doctor or pharmacist for advice before using Zaditen.
Zaditen can be used during breast-feeding.
*
Driving and using machines
Zaditen may cause blurred vision or drowsiness. If this happens to you, wait until this has cleared before driving or operating machinery.
Important information about some of the ingredients of Zaditen
Zaditen contains benzalkonium chloride and may cause eye irritation.
If you wear soft contact lenses you should remove them before using Zaditen as it can discolour your soft contact lenses. You should wait at least 15 minutes after using Zaditen before reinserting your contact lenses into your eyes.
3. HOW TO USE ZADITEN
Always use Zaditen exactly as your doctor has told you. You should check with your doctor or pharmacist if you are not sure.
The usual dose for adults, elderly and children (age 3 and older) is one drop into the affected eye(s) twice a day (in the morning and evening).
Instructions for use
1. Wash your hands.
2. Open the bottle. Do not touch the tip after opening the bottle.
3. Lean your head back (Fig. 1).
4. Pull down your lower eyelid with your finger and hold the bottle in your other hand. Squeeze the bottle so that one drop falls into the eye (Fig. 2).
5. Close your eyes and press the tip of one finger against the corner of the eye for around 1-2 minutes. This will prevent the drop running through the tear duct into your throat and most of the drop will remain in the eye (Fig. 3). If necessary repeat steps 3 to 5 with your other eye.
6. Close the bottle after use.
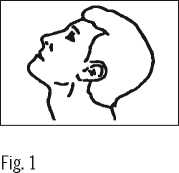


Fig. 3
If you have any further questions on the use of this product, ask your doctor or pharmacist.
If you use more Zaditen than you should
There is no danger if you have accidentally taken Zaditen by mouth or if you have used more than one drop in the eye. If you have any doubt contact your doctor for advice.
If you forget to use Zaditen
00000000
If you forget to use Zaditen you should treat your eyes as soon as you remember. Then continue with your normal routine.
Do not take a double dose to make up for a forgotten dose.
4. POSSIBLE SIDE EFFECTS
Like all medicines, Zaditen can cause side effects, although not everybody gets them. The following side effects have been reported.
Common (affecting less than 1 in every 10 patients)
• eye irritation or pain
• inflammation in the eye
• eye pain, blurred vision, abnormal intolerance to light
Uncommon (affecting less than 1 in every 100 patients)
• blurred vision when putting drops on the eye
• dry eye
• eyelid disorder
• conjunctivitis
• increased sensitivity of the eyes to light
• visible bleeding in white of eye
• headache
• drowsiness
• rash (which may also itch)
• eczema (itchy, red, burning rash)
• dry mouth
• allergic reaction (including swelling of the face and eyelids) and increase in severity of existing allergic conditions such as asthma and eczema
If any of the side effects gets serious, or if you notice any side effects not listed in this leaflet, please tell your doctor or pharmacist.
5. HOW TO STORE ZADITEN
Keep out of reach and sight of children.
Do not store above 25 °C.
The bottle is not sterile itself, but its contents are sterile until the bottle is opened.
After opening the bottle, the eye drops can only be stored for 4 weeks.
Do not use Zaditen after the expiry date, which is stated on the carton and bottle after EXP. The expiry date refers to the last day of that month.
Medicines should not be disposed of via wastewater or household waste. Ask your pharmacist how to dispose of medicines no longer required. These measures will help to protect the environment.
6. FURTHER INFORMATION
What Zaditen contains
The active substance is ketotifen (as fumarate). Each ml contains 0.345 mg ketotifen fumarate corresponding to 0.25 mg ketotifen.
The other ingredients are glycerol (E422), sodium hydroxide (E524), water for injections and benzalkonium chloride.
What Zaditen looks like and contents of the pack
Zaditen is a clear, colourless to faint yellow solution. The solution is available in a pack containing one bottle of 5 ml.
Marketing Authorisation Holder and Manufacturer
Marketing Authorisation Holder:
Laboratoires THEA
12, rue Louis Bleriot, 63017 Clermont-Ferrand Cedex 2, France.
The manufacturer responsible for release on to the market is :
EXCELVISION, rue de la Lombardiere, Annonay, France.
If you would like any more information, or would like the leaflet in a different format, please contact Medical Information at:
Spectum THEA Pharmaceuticals Ltd, telephone number 0845 521 1290.
Austria
Czech Republic
Denmark
Finland
France
German
Greece
Iceland
Ireland
Italy
Luxembourg
Norway
Portugal
Spain
Sweden
The Netherlands United Kingdom
Zaditen 0,025% - Augentropfen Zaditen 0,025%
Zaditen
Zaditen 0,25 mg/ml silmatipat, liuos Zaditen, 0,25 mg/ml, collyre en solution Zaditen ophtha 0,25 mg/ml Augentropfen Zaditen ofOaApiKEg stayoveg Zaditen
Zaditen 0.25mg/ml, eye drops, solution
Zaditen 0,25 mg/ml collirio soluzione, 1 flacone da 5 ml
Zaditen 0,25 mg/ml, collyre en solution
Zaditen
Zaditen 0,25 mg/ml collrio, solugao Zaditen colirio
Zaditen 0,25 mg/ml, ogondroppar, losning Zaditen 0,25 mg/ml, oogdruppels, oplossing Zaditen 0.25 mg/ml, eye drops, solution
Olhea
N2900C5ASP/0915
00000000
O 00000000