Zlatal 10 Mg Solution For Injection In Pre-Filled Syringe
Package leaflet: Information for the patient Zlatal 75 mg Solution for injection in pre-filled syringe Zlatal 10 mg Solution for injection in pre-filled syringe Zlatal 125 mg Solution for injection in pre-filled syringe Zlatal 15 mg Solution for injection in pre-filled syringe Zlatal 175 mg Solution for injection in pre-filled syringe Zlatal 20 mg Solution for injection in pre-filled syringe Zlatal 225 mg Solution for injection in pre-filled syringe Zlatal 25 mg Solution for injection in pre-filled syringe Methotrexate
Read all of this leaflet carefully before you start using this medicine because it contains important information for you.
- Keep this leaflet. You may need to read it again.
- If you have any further questions, ask your doctor or pharmacist.
- This medicine has been prescribed for you only. Do not pass it on to others. It may harm them, even if their signs of illness are the same as yours.
- If you get any side effects, talk to your doctor or pharmacist. This includes any possible side effects not listed in this leaflet. See section 4.
What is in this leaflet
1. What Zlatal is and what it is used for
2. What you need to know before you use Zlatal
3. How to use Zlatal
4. Possible side effects
5. How to store Zlatal
6. Contents of the pack and other information
1. What Zlatal is and what it is used for
Zlatal is a drug with the following properties:
- it interferes with the growth of certain cells in the body that reproduce quickly (anti-tumour agent)
- it reduces undesired reactions of the body’s own defence mechanism (immunosuppressant), and
- it has anti-inflammatory effects Zlatal is indicated for the treatment of:
- Active rheumatoid arthritis in adult patients,
- Polyarthritic forms of severe, active juvenile idiopathic arthritis, when the response to nonsteroidal anti- inflammatory drugs (NSAIDs) has been inadequate,
- Severe recalcitrant disabling psoriasis, which is not adequately responsive to other forms of therapy such as phototherapy, PUVA, and retinoids, and severe psoriatic affecting the joints (psoriac arthritis) in adult patients.
2. What you need to know before you use Zlatal Important warning with reference to the dosing of methotrexate: Methotrexate for the therapy of rheumatic diseases or diseases of the skin must only be used once weekly. Faulty dosing of methotrexate may lead to serious adverse effects including fatal course.
Please read section 3 of this PL very carefully.
Please ask your doctor or pharmacist before using Zlatal if you have any question.
Do not use Zlatal:
- If you are allergic (hypersensitive) to methotrexate or any of the other ingredients of this medicine (listed in section 6).
- If you have significant kidney disease (your doctor decides the severity of the disease)
- If you have significant liver disease (your doctor decides the severity of your disease)
- If you have disorders of the blood-forming system
- If your alcohol consumption is high
- If you have an impaired immune system
- If you have a severe or existing infection, e.g. tuberculosis and HIV
- If you have gastrointestinal ulcers
- If you are pregnant or breast-feeding (see section “Pregnancy, breast feeding and fertility”)
- If you receive vaccinations with live vaccines at the same time. Warnings and precautions
Talk to your doctor before using Zlatal:
- If you have diabetes mellitus treated with insulin
- If you have inactive, prolonged infections (e.g. tuberculosis, hepatitis B or C, shingles (herpes zoster)
- If you have/had any liver or kidney disease
- If you have problems with your lung function
- If you are severely overweight
- If you have abnormal accumulation of liquid in the abdomen or in the cavity between the lungs and chest wall (ascites, pleural effusions)
- If you are dehydrated or suffer from conditions leading to dehydration (vomiting, diarrhoea, stomatitis)
The therapy is to be administered once a week.
Incorrect intake of methotrexate can lead to severe, including potentially lethal, side effects. Read section 3 of this package leaflet carefully.
If you have experienced problems with your skin after radiation therapy (radiation induced dermatitis) and sun-bum these conditions can reappear under methotrexate therapy (recall-reaction).
Use in children, adolescents and elderly
Dose instructions depend on patient’s body weight. Use in children < 3 years of age is not recommended due to the insufficient experience in this age group.
Children and the elderly under Zlatal treatment should be kept under particularly close, medical surveillance, in order to identify possible side effects as early as possible.
Dose for elderly patients should be relatively low due to age-related reduced liver and kidney function and low folate reserves.
Special precautionary measures during treatment of Zlatal
Zlatal should only be prescribed by doctors with sufficient experience in
the Methotrexate treatment of the disease concerned.
Methotrexate temporarily affects sperm and egg production. You and your partner must avoid conception (becoming pregnant or fathering children) if currently receiving methotrexate and for at least six months after your treatment with methotrexate has stopped. See also section “Pregnancy, breast feeding and fertility”.
Skin changes caused by psoriasis can worsen during treatment with Zlatal if exposure to UV irradiation occurs at the same time.
Recommended follow-up examinations and precautions:
Even when Zlatal is used at low doses, serious side effects can occur. In order to recognise these in good time, your doctor must carry out checkups and laboratory tests.
Before the start of treatment:
Before treatment is started your doctor may carry out blood tests, and also to check how well your kidneys and liver are working. You may also have a chest X-ray. Further tests may also be done during and after treatment. Do not miss appointments for blood tests.
If the results of any of these tests are abnormal, treatment will only be resumed when all readings are back to normal.
Other medicines and Zlatal
Tell your doctor or pharmacist if you are taking, have recently taken or might take any other medicines, including medicines obtained without a prescription and herbal or natural medicinal products.
It is especially important to tell your doctor if you are using:
- other treatments for rheumatoid arthritis or psoriasis such as leflunomide, sulphasalazine (also used for ulcerative colitis), aspirin, phenylbutazone, or amidopyrine
- alcohol (should be avoided)
- live vaccinations
- azathioprine (used to prevent rejection after an organ transplant)
- retinoids (used to treat psoriasis and other skin disorders)
- anticonvulsant drugs (prevent fits)
- cancer treatments
- barbiturates (sleeping injection)
- tranquillisers
- oral contraceptives
- probenecid (against gout)
- antibiotics
- pyrimethamine (used to prevent and treat malaria)
- vitamin preparations, which contain folic acid
- proton-pump inhibitors (used to treat severe heartburn or ulcers)
- theophylline (used to treat asthma)
Zlatal with food, drink and alcohol
During treatment with Zlatal, you should not drink any alcohol and you should avoid excessive consumption of coffee, soft drinks containing caffeine and black tea.
Also make sure you drink plenty of liquids during treatment with Zlatal because dehydration (reduction in body water) can increase the toxicity of Zlatal.
Pregnancy, breast-feeding and fertility
If you are pregnant or breast-feeding or think you may be pregnant or are planning to have a baby, ask your doctor for advice before taking this medicine.
Pregnancy
Do not use Zlatal during pregnancy or if you are trying to become pregnant. Methotrexate can cause birth defects, harm unborn babies or cause miscarriages and so it is very important that it is not given to pregnant patients or patients planning to become pregnant. Therefore, in women of child-bearing age any possibility of pregnancy must be excluded with appropriate measures, e.g. a pregnancy test, before starting treatment. You must avoid becoming pregnant whilst taking methotrexate and for at least 6 months after treatment is stopped. Therefore you must ensure reliable contraception during this whole period (see also section “Warnings and precautions”).
If you do become pregnant during treatment, you should be offered advice regarding the risk of harmful effects on the child through treatment.
If you wish to become pregnant you should consult your doctor, who may refer you for specialist advice, before the planned start of treatment, because methotrexate may be genotoxic, which means that the medicine may cause genetic mutation.
Breast-feeding
Do not breast-feed during treatment, because methotrexate passes into breast milk. If your attending doctor considers treatment with methotrexate absolutely necessary during the lactation period, you must stop breast- feeding. Male fertility
Methotrexate may be genotoxic. This means that the medicine may cause genetic mutation. Methotrexate can affect sperm and egg production with the potential to cause birth defects. Therefore, you must avoid fathering a child whilst taking methotrexate and for at least 6 months after treatment is stopped. Since treatment with methotrexate may lead to infertility, it might be advisable for male patients to look into the possibility of sperm preservation before starting treatment (see also section “Warnings and precautions”).
Driving and using machines
Side effects affecting the central nervous system, such as tiredness and dizziness, may occur during treatment with Zlatal. In some cases, the ability to drive vehicles and/or use machines may therefore be impaired. If you feel tired or dizzy, you should not drive or use machines.
Zlatal contains sodium
This medicinal product contains less than 1 mmol sodium (23 mg) per dose, i.e. essentially “sodium-free”.
3. How to use Zlatal
Zlatal should only be prescribed by physicians who are familiar with the various characteristics of the medicinal products and its mode of action. Always use this medicine exactly as your doctor has told you. Check with your doctor or pharmacist if you are not sure.
Zlatal is administered once a week only. Together with your doctor you decide on a suitable weekday each week on which you receive your injection.
Incorrect intake of Zlatal can lead to severe, including potentially lethal, side effects. The usual dose is:
Dose in patients with rheumatoid arthritis
The recommended starting dose for methotrexate is 7.5 mg once a week. Zlatal is given in a single application as injection under the skin or into a muscle (see section “Method and duration of administration”).
In case of inadequate action and if tolerated well, Zlatal doses may be increased. The mean weekly dose is 15-20 mg. Generally, a weekly dose of 25 mg Zlatal should not be exceeded. Upon achieving desired therapeutic results, the dose should - if possible - be gradually reduced to the lowest possible effective maintenance dose.
Dose in children and adolescents below 16 years with polvarthritic forms of juvenile idiopathic arthritis
The recommended dose is 10-15 mg/m2 body surface area per week. In cases with inadequate response, the weekly dose may be increased up to 20 mg/m2 body surface area/week. However, regular check-ups should be done more often. It should only be given by subcutaneous (under the skin) or intramuscular (into the muscle) injection.
Use in children < 3 years of age is not recommended due to the insufficient experience in this age group.
Adults with severe forms of psoriasis vulgaris or psoriatic arthritis It is recommended to administer a single test dose of 5-10 mg, in order to assess possibly damaging effects. This dose can be administered subcutaneously (under the skin) or intramuscularly (into a muscle). If, one week later, no blood count changes are observed, therapy is continued with a dose of approximately
7.5 mg. The dose may be gradually increased (in steps of 5-7.5 mg per week and under blood count surveillance) until ideal therapeutic results are obtained. Generally, a weekly dose of 20 mg can be associated with significant increase in toxicity. A dose of 30 mg per week should not be exceeded. Upon achieving desired therapeutic results, the dose should be weekly reduced to the lowest possible effective maintenance dose for the individual patient.
Patients with a kidney disorder
Patients with a kidney disorder may need a reduced dose.
Method and duration of administration
The duration of the treatment is determined by the treating physician. Zlatal is injected once weekly! It is recommended to specify a certain day of the week as “day for injection”.
Zlatal is given as injection under the skin or into a muscle. Treatment of rheumatoid arthritis, juvenile idiopathic arthritis, psoriasis vulgaris and psoriatic arthritis with Zlatal is a long-term treatment.
Rheumatoid arthritis
Generally, improvement of the symptoms can be expected after 4-8 weeks of treatment. Symptoms may return after Zlatal discontinuation.
Severe types of psoriasis vulgaris and psoriatic arthritis /psoriasis arthro-paticai
Response to treatment can generally be expected after 2-6 weeks. Depending on the clinical picture and the changes of laboratory parameters, the therapy is then continued or discontinued.
At the start of your therapy, Zlatal may be injected by medical staff. However, your doctor may decide that it is right for you to learn how to inject Zlatal under the skin yourself. You will receive appropriate training for you to do this. Under no circumstances should you attempt to inject yourself unless you have been trained to do so.
How to give yourself an injection of Zlatal
If you have trouble handling the syringe, ask your doctor or pharmacist. Do not try to inject yourself if you have not been trained how to do so. If you are not sure what to do, talk to your doctor or nurse immediately. Before injecting yourself with Zlatal
- Check die expiry date on the medicine. Do not use if the date has passed.
- Check the syringe is not damaged and the medicine in it is a clear solution. If not, use another syringe.
- Check your last injection site to see if the last injection caused any redness, change in skin colour, swelling, oozing or is still painful, if so talk to your doctor or nurse.
- Decide where you are going to inject the medicine. Change the place where you inject each time.
Instructions on injecting yourself with Zlatal
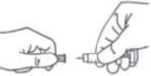
1) Wash your hands and wipe the area that you will inject with the alcohol swab provided.
2) Sit or lie in a comfortable position so you are relaxed. Make sure you can see the place you are going to inject. A lounge chair, recliner, or bed propped up with pillows is ideal.
3) Remember to change the place where you inject everytime.
4) Carefully pull off the needle cap from the Zlatal syringe. Throw away the cap. The syringe is pre-filled and ready to use.
Do not press on the plunger before injecting yourself to get rid of air bubbles. This can lead to a loss of the medicine. Once you have removed the cap, do not allow the needle to touch anything. This is to make sure the needle stays clean (sterile).
5) Hold the syringe in the hand you write with (like a pencil) and with your other hand, gently pinch the cleaned area between your forefinger and thumb to make a fold in the skin.
5208800/B
Customer: CENEXI-THISSEN Customer PO: MAILADG 12/02/16 Product description :METH 7,5 mg - 25 mg
Id Product customer: 5208800/B
Technical information
Dimension Leaflet: 150x630 Leaflet size folded: 150x30 Diecut internal reference:
Paperboard and grammage: Polor Bright-40gr
Bleed:
yes
/r?i
Printing information
Colors:
1 Black
2 Cad
Internal information
Operator: christel
Job number: 18245
Id record product: 50737
Date: 17-03-16 10:37 Proof number: 3
Information
Please signe here for approval:

GOLDPRINT
LGR - GoldPrint NV Vaucampslaan 32 1654 Huizingen Tel.: 02/363 89 79 FAX: 02/363 89 80
De kleuren van deze proefzijn slechts een digitale weergave. Beoordeel deze kleuren aan de hand van de vermelding en de PMS-waaier.
Les couleurs de cette ipreuve sont uniquement une reproduction digitale, jugez les couleurs d'apris I'lndication et l/venta il-PMS.
The colours are only a digital reproduction. Check the colours with a Pan tone PMS guide.
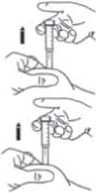
Make sure you hold the skin fold throughout the injection.
6) Hold the syringe so that the needle is pointing downwards (vertically at a 90° angle). Insert the full length of the needle into the skin fold.
7) Press down on the plunger with your finger until the syringe is empty. This will send the medication under the skin. Make sure you hold the skin fold throughout the injection.
8) Remove the needle by pulling it straight out.
A protective sleeve will automatically cover the needle.
You can now let go of the skin fold.
Note: the safety system allowing release of the protective sleeve, can only be activated when the syringe has been emptied by pressing the plunger all the way down. To avoid bruising, do not rub the injection site after you have injected yourself.
9) Drop the used syringe with its protective sleeve into the sharps bin provided. Close the container lid tightly and place the container out of reach of children. If you accidently get methotrexate on the surface of the skin or mucosa you must rinse with plenty of water.
When the container is full, give it to your doctor or home care nurse for disposal. Do not put it in the household rubbish.
If you use more Zlatal than you should
Follow the dose recommendations of your treating doctor. Do not change the dose of your own accord.
If you suspect that you (or someone else) have used too much Zlatal, please tell your doctor or contact the nearest hospital casualty department immediately. He/she will decide on any necessary treatment measures, depending on the severity of intoxication.
An overdose of methotrexate can lead to severe toxic reactions. Overdose symptoms may include easy bruising or bleeding, unusual weakness, mouth sores, nausea, vomiting, black or bloody stools, coughing up blood or vomit that looks like coffee grounds, and decreased urinating. See also section 4. Take your medicine package with you if you go to a doctor or hospital. The antidote in case of an overdose is calcium folinate.
If you forget to use Zlatal
Do not take a double dose to make up for a forgotten individual doses, but continue taking the ordered dose. Ask your doctor for advice.
If you stop taking Zlatal
You should not interrupt or discontinue Zlatal treatment, unless you have discussed this with your doctor. If you suspect severe side effects, contact your doctor immediately for advice.
If you have any further questions on the use of this medicine, ask your doctor or pharmacist.
4. Possible side effects
Like all medicines, this medicine can cause side effects, although not everybody gets them.
Tell your doctor straight away if you get any sudden wheeziness, difficulty in breathing, swelling of the eyelids, face or lips, rash or itching (especially affecting your whole body).
Serious side effects
If you develop any of the following side effects, contact your doctor immediately:
• lung complaints (symptoms may be general illness; dry, irritating cough; shortness of breath, breathlessness at rest, chest pain, or fever)
• Severe peeling or blistering of the skin
• Unusual bleeding (including vomiting blood) or bruising
• severe diarrhoea
• ulcers in mouth
• black or tarry stools
• blood in the urine or stools
• tiny red spots on the skin
• fever
• yellowing of the skin (jaundice)
• pain or difficulty in passing urine
• thirst and/or frequent urination
• fits (convulsions)
• loss of consciousness
• blurred or decreased vision
The following side effects have also been reported:
Very common: (affects more than 1 user in 101 loss of appetite, nausea (feeling sick), vomiting, tummy pain, inflammation and ulcers in the mouth and throat, and increase in liver enzymes Commomfaffects 1 to 10 users in 1001
Reduced blood cell formation with decrease in white and/or red blood cells and/or platelets (leukopenia, anaemia, thrombocytopenia), headache, tiredness, drowsiness, inflammation of the lungs (pneumonia) with dry, non-productive cough, shortness of breath and fever, diarrhoea, rash, reddening of the skin, itching.
Uncommon: (affects 1 to 10 users in 10001
Decrease in the number of blood cells and platelets, dizziness, confusion, depression, fits, inflammation of blood vessels, lung damage, ulcers and bleeding in the digestive tract, liver disorders, diabetes, decreased blood protein, nettle rash (alone), light sensitivity, brown skin, hair loss, increase of rheumatic nodules, shingles, painful psoriasis, joint or muscle pain, osteoporosis (reduction of bone mass), inflammation and ulcers of the bladder (possibly with blood in the urine), painful urination, severe allergic reactions, inflammation and ulcers of the vagina, slow wound healing. Rare: (affects 1 to 10 users in 10.0001
Inflammation of the sac around the heart, fluid in the sac around the heart, severe visual disturbance, mood fluctuation low blood pressure, blood clots, sore throat, interruption of breathing, asthma, inflammation of the digestive tract, bloody stools, inflamed gums, abnormal digestion, acute hepatitis (inflammation of the liver), changed colour of nails, acne, red or purple spots due to vessel bleeding, bone fracture, kidney failure, decrease or absence of urine, electrolyte disturbances, defective sperm formation, menstruation disorders.
Very rare: (affects less than 1 user in 10.0001
Infections, severe failure of the bone marrow, liver failure, swollen glands, sleeplessness, pain, muscle weakness, pins and needles, changes in sense of taste (metallic taste), inflammation of the lining of the brain causing paralysis or vomiting, red eyes, damage to the retina of the eye, fluid in the lungs, vomiting blood, cold sores, protein in the urine, fever, loss of sex drive, problems having an erection, infection around a fingernail, severe complication of the digestive tract, boils, small blood vessels in the skin, fungal infections, damage to the blood vessels of the skin, vaginal discharge, infertility, male breast enlargement (gynaecomastia).
When methotrexate is given by the intramuscular route, local undesirable effects (burning sensation) or damage (formation of sterile abscess, destruction of fatty tissue) at the site of injection can occur commonly. Subcutaneous application of methotrexate is locally well tolerated. Only mild local skin reactions were observed, decreasing during therapy. Methotrexate may cause a reduction in the number of white blood cells and your resistance to infection may be decreased. If you experience an infection with symptoms such as fever and serious deterioration of your general condition, or fever with local infection symptoms such as sore throat/sore pharynx/sore mouth or urinary problems you should see your doctor immediately. A blood test will be taken to check for possible reduction of white blood cells (agranulocytosis). It is important to inform your doctor about your medicine.
Methotrexate may cause serious (sometimes life-threatening) side effects.
Therefore, your doctor will do tests to check for abnormalities developing in the blood (e.g. low white blood cells, low platelets, lymphoma) and changes in the kidney and the liver.
Reporting of side effects
If you get any side effects, talk to your doctor, pharmacist or nurse. This includes any possible side effects not listed in this leaflet. You can also report side effects directly via the Yellow Card Scheme at: wwwjnhra.gov.uk/yellowcard
By reporting side effects you can help provide more information on the safety of this medicine.
5. How to store Zlatal
Keep this medicine out of the sight and reach of children.
Store below 25°C.
Keep the syringe in the outer carton in order to protect from light.
Do not freeze.
Do not use this medicine after the expiry date which is stated on the label of the pre-filled syringe and the carton after EXP. The expiry date refers to the last day of that month.
The product has to be used immediately after opening.
You must not use Zlatal, if the solution is not clear and contains particles. For single use only. Any used solution should be discarded!
Do not throw away any medicines via wastewater or household waste. Ask your pharmacist how to throw away medicines you no longer use. These measures will help protect the environment.
6. Contents of the pack and other information What Zlatal contains
- The active substance is methotrexate. 1 ml of solution contains methotrexate corresponding to 25 mg methotrexate.
- The other ingredients are sodium chloride, sodium hydroxide, water for injections.
What Zlatal looks like and contents of the pack
Zlatal pre-filled syringes contain a clear, yellow solution. The pre-filled syringes are attached with an injection needle and with a safety device to prevent needlestick injury and reuse.
The following pack sizes are available:
Pre-filled syringes of 0.3 ml contain 7.5 mg methotrexate.
Pre-filled syringes of 0.4 ml contain 10 mg methotrexate.
Pre-filled syringes of 0.5 ml contain 12.5 mg methotrexate.
Pre-filled syringes of 0.6 ml contain 15 mg methotrexate.
Pre-fiUed syringes of 0.7 ml contain 17.5 mg methotrexate.
Pre-filled syringes of 0.8 ml contain 20 mg methotrexate.
Pre-filled syringes of 0.9 ml contain 22.5 mg methotrexate.
Pre-filled syringes of 1.0 ml contain 25 mg methotrexate Solution for injection in packs of 1,4,6 and 24 pre-filled syringes.
Packs of 1,4,6 and 24 pre-filled syringes contain 2,8,12 and 48 alcohol
swabs respectively
Not all pack sizes may be marketed.
Marketing Authorisation Holder
Nordic Group BV
Siriusdreef 22
2132WT Hoofddorp
The Netherlands
Manufacturer
Cenexi - Laboratoires Thissen SA Rue de la Papyree 2-6, Braine-L’Alleud Brabant Wallon, B-1420 Belgium
Customer: CENEXI-THISSEN Customer PO: MAIL ADG 12/02/16 Product description :METH 7,5 mg - 25 mg
Id Product customer: 5208800/B
Technical information
Dimension Leaflet: 150x630 Leaflet size folded: 150x30 Diecut internal reference:
Paperboard and grammage: Polor Bright-40gr
Bleed:
yes
/ni
Printing information
Colors:
1 Black
2 Cad
This medicinal product is authorised in the Member States of the EEA under the following names:
Internal information
Zlatal 7.5 mg, solution for injection in pre-filled syringe Zlatal 10 mg, solution for injection in pre-filled syringe Zlatal 12.5 mg, solution for injection in pre-filled syringe Zlatal 15 mg, solution for injection in pre-filled syringe Zlatal 17.5 mg, solution for injection in pre-filled syringe Zlatal 20 mg, solution for injection in pre-filled syringe Zlatal 22.5 mg, solution for injection in pre-filled syringe Zlatal 25 mg, solution for injection in pre-filled syringe This leaflet was last revised in 02/16
Operator: christel
Job number: 18245
Id record product: 50737
Date: 17-03-16 10:36 Proof number: 3
Information
Please signe here for approval:

GOLDPRINT
LGR - GoldPrint NV Vaucampslaan 32 1654 Huizingen Tel.: 02/363 89 79 FAX: 02/363 89 80
5208800/B
De kleuren van deze proefzijn slechts een digitale weergave. Beoordeel deze kleuren aan de hand van de vermelding en de PMS-waaier.
Les couleurs de cette ipreuve sont uniquement une reproduction digitale, jugez les couleurs d'apris I'lndication et l/venta il-PMS.
The colours are only a digital reproduction. Check the colours with a Pantone PMS guide.