Artiss Solutions For Sealant Deep Frozen
Baxter
0001893
PACKAGE LEAFLET: INFORMATION FOR THE USER
ARTISS Solutions for Sealant m
Deep frozen
Active Substances: Human Fibrinogen, Human Thrombin, Aprotinin, Calcium Chloride.
000000
Read all of this leaflet carefully before you start using this medicine because it contains important information for you.
- Keep this leaflet. You may need to read it again.
- If you have any further questions, ask your doctor or pharmacist.
- If you get any side effects, talk to your doctor or pharmacist. This includes any possible side effects not listed in this leaflet. See section 4.
What is in this leaflet
1. What ARTISS is and what it is used for
2. What you need to know before you use ARTISS
3. How to use ARTISS
4. Possible side effects
5. How to store ARTISS
6. Contents of the pack and other information
1. What ARTISS is and what it is used for What ARTISS is
ARTISS is a two-component fibrin sealant, and it contains two of the proteins that make blood clot. These proteins are called fibrinogen and thrombin. When these proteins mix during application, they form a clot where the surgeon applies them. ARTISS is prepared as two solutions (Sealer Protein Solution and Thrombin Solution), which mix when applied.
What ARTISS is used for
ARTISS is a tissue sealant.
ARTISS is applied to glue soft tissues in plastic, reconstructive and burn surgery. For example, ARTISS may be used to glue skin grafts or skin flaps to burn wounds or to glue skin to the underlying tissue in plastic surgery. Also artificial skin may be glued to wounds with ARTISS.
The clot produced by ARTISS is very similar to a natural blood clot. This means that it will dissolve naturally and leave no residue. However, aprotinin (a protein, that delays dissolution of a clot) is added to increase the longevity of the clot and to prevent its premature dissolution.
2. What you need to know before you use ARTISS Do not use ARTISS:
• ARTISS must not be used for massive or brisk bleeding.
• ARTISS is not indicated to replace skin sutures intended to close a surgical wound.
• ARTISS is not indicated for use in neurosurgery and as a suture support for gastrointestinal anastomoses or vascular anastomoses as no data are available to support these indications.
• ARTISS MUST NOT be injected into blood vessels (veins or arteries), or into tissues. As ARTISS forms a clot where it is applied, injecting ARTISS may cause serious reactions (e.g. vessel occlusion). ARTISS should only be applied to the surface of tissues as a thin layer where it is needed.
• You must not receive ARTISS if you are allergic (hypersensitive) to the active substances, to bovine protein or any of the other ingredients (see section 6) of ARTISS. It may cause serious allergic reactions.
Please inform your doctor or surgeon if you know that you are allergic against aprotinin or any bovine protein.
Warnings and precautions
• ARTISS should not be used in laparoscopic surgery (keyhole surgery).
• Life-threatening/fatal air or gas embolism (air getting into the blood circulation which can be serious or life-threatening) has occurred with the use of spray devices employing pressure regulators to administer fibrin sealants. This appears to be related to the use of the spray device at higher than recommended pressures and/or in close proximity to the tissue surface. The risk appears to be higher when fibrin sealants are sprayed with air, as compared to CO2 and therefore cannot be excluded with ARTISS.
• When ARTISS is applied using a spray device, the pressure and spraying distance have to be within the range recommended by the manufacturer. ARTISS should be administered strictly according to the instructions and only with devices recommended for this product.
• When spraying ARTISS, changes in blood pressure, pulse, oxygen saturation and end tidal CO2 should be monitored for possible occurrence of gas embolism.
• ARTISS must not be used with the Easy Spray/Spray Set system in enclosed body areas.
• ARTISS should be applied only with CE marked application devices.
• If you have ever received ARTISS or aprotinin before, your body may have become sensitive to it. It is possible you may be allergic to this material, even if there was no reaction to the first application. If you think you have received either product in a previous operation, you have to inform your doctor about this.
• If there is any sign of an allergic reaction, your doctor will stop the use of ARTISS immediately and give appropriate treatment.
• Before administration of ARTISS parts of the body outside the designated application area have to be sufficiently protected/covered to prevent unwanted tissue adhesion.
• ARTISS is applied as a thin layer. Excessive clot thickness may negatively interfere with the product's efficacy and the wound healing process.
When medicines are made from human blood or plasma, certain measures are put in place to prevent infections being passed on to patients. These include:
• careful selection of blood and plasma donors to make sure those at risk of carrying infections are excluded,
• the testing of each donation and pools of plasma for signs of virus/infections,
• the inclusion of steps in the processing of the blood or plasma that can inactivate or remove viruses.
Despite these measures, when medicines prepared from human blood or plasma are administered, the possibility of passing on infection cannot be totally excluded. This also applies to any unknown or emerging viruses or other types of infections. The measures taken are considered effective for enveloped viruses such as human immunodeficiency virus (HIV), hepatitis B virus and hepatitis C virus, and for the non-enveloped hepatitis A virus. The measures taken may be of limited value against non-enveloped viruses such as parvovirus B19. Parvovirus B19 infection may be serious for pregnant women (fetal infection) and for individuals whose immune system is depressed or who have some types of anaemia (e.g. sickle cell disease or haemolytic anaemia).
It is strongly recommended that every time you receive a dose of ARTISS the name and batch number of the product are recorded in order to maintain a record of the batches used.
Other medicines and ARTISS
ARTISS can be used when you are receiving other medical products. There are no known interactions between ARTISS and other medicinal products. Your doctor will not use oxycellulose-containing preparations as carrier materials as they may reduce the efficacy of ARTISS.
Please tell your doctor or pharmacist if you are taking, have recently taken or might take any other medicines.
ARTISS with food and drink
Please ask your doctor. The doctor will decide if you are allowed to eat and drink before the application of ARTISS.
Pregnancy and breast-feeding
If you are pregnant or breastfeeding, think you may be pregnant or are planning to have a baby ask your doctor for advice. Your doctor will decide if you can use ARTISS during pregnancy or breast-feeding.
Driving and using machines
ARTISS will not affect your ability to drive or operate other types of machines.
3. How to use ARTISS
• ARTISS is only applied during a surgical operation. The use of ARTISS is restricted to experienced surgeons who have been trained in the use of ARTISS.
• The amount of ARTISS that will be used depends on a number of factors, including the type of surgery, the surface area of tissue to be treated during your operation and the way ARTISS is applied. The surgeon will decide how much is appropriate.
• During your operation, the surgeon will apply ARTISS onto the relevant tissue surface, using the special application device provided. This device ensures that equal amounts of both fibrin sealant components are applied at the same time -which is important for the optimal effect of ARTISS.
• Prior to applying ARTISS the surface area of the wound needs to be dried by standard techniques (e.g. intermittent application of compresses, swabs, use of suction devices).
• ARTISS must be sprayed only onto application sites that are visible.
• It is recommended that the initial application covers the entire intended application area.
When applying ARTISS using a spray device be sure to use a pressure and a distance from the tissue within the range recommended by the manufacturer as follows:
|
Recommended pressure, distance and devices for spray application of ARTISS | |||||
|
Spray set to be used |
Applicator tips to be used |
Pressure regulator to be used |
Recommended distance from target tissue |
Recommended spray pressure | |
|
Open wound surgery of subcutaneous tissue |
Tisseel/ Artiss Spray Set |
n.a. |
EasySpray |
10 - 15 cm |
1.5 - 2.0 bar (21.5 -28.5 psi) |
|
Tisseel/ Artiss Spray Set 10 pack |
n.a. |
EasySpray | |||
When spraying the ARTISS, changes in blood pressure, pulse, oxygen saturation and end tidal CO2 should be monitored because of the possibility of occurrence of air or gas embolism (see section 2).
If you use more ARTISS than you should
ARTISS is only applied during a surgical operation. It is applied by the surgeon and the amount of ARTISS is determined by the surgeon.
If you have any further questions on the use of this product, ask your doctor or pharmacist.
4. Possible side effects
Like all medicines, this medicine can cause side effects, although not everybody gets them. The following table explains what is meant with a certain frequency, as given in the following section:
|
very common: |
may affect more than 1 in 10 people |
|
common: |
may affect up to 1 in 10 people |
|
uncommon: |
may affect up to 1 in 100 people |
|
rare: |
may affect up to 1 in 1,000 people |
|
very rare: |
may affect up to 1 in 10,000 people |
|
not known: |
frequency cannot be estimated from the available data |
• There is a slight possibility that you might have an allergic reaction to one of the components of ARTISS (see section 6). This is more likely if you have been treated with ARTISS or aprotinin during a previous operation. Allergic reactions can be serious, and it is very important that you discuss this possibility in detail with your doctor.
• Allergic reactions of the anaphylactic/anaphylactoid type may occur, frequency of this is not known. Early symptoms of allergic reactions can be: flushing, a fall in blood pressure, increased or decreased pulse rate, nausea (feeling sick), hives, itching, difficulty breathing.
• The surgical team treating you will be aware of the risk of this type of reaction - if they see any symptoms, the application of ARTISS will be stopped immediately. Severe symptoms may require emergency treatment. The frequency for allergic reactions is not known.
• If ARTISS is injected into soft tissues, it can cause local tissue damage. Frequency is not known.
• If ARTISS is injected into blood vessels (veins or arteries), it can cause clots to form (thrombosis). Frequency is not known.
• As ARTISS is made from plasma from blood donations, the risk of infection cannot be totally excluded, but the manufacturer undertakes numerous measures to reduce the risk (see section 2).
Adverse reactions reported from clinical studies of ARTISS and from postmarketing experience with Baxter Fibrin Sealants are summarized in the following. Known frequencies of these adverse reactions are based on a controlled clinical study in 138 patients where skin grafts were fixed to excised burn wounds using ARTISS. None of the events observed in the clinical study were classified as serious.
|
Table 1 Adverse Reactions | |
|
Adverse Reaction |
Frequency |
|
Dermal cyst |
uncommon |
|
Itching |
common |
|
Skin graft failure |
common |
|
Gas bubbles in the vascular system (Air embolism)* |
not known |
* The introduction of air or gas bubbles in the blood stream (air embolism) has occurred when fibrin sealants are applied with devices using pressurized air or gas; this is believed to be caused by inappropriate use of the spray device (e.g. at higher than recommended pressure and in close proximity to the tissue surface).
The following adverse reactions have been reported for other fibrin sealants, frequencies of those cannot be provided: Allergy, severe allergic reaction, slow heart rate, fast heart rate, decrease in blood pressure, effusion of blood, shortness of breath, sickness, hives, flushing, impaired healing, swelling, fever, and accumulation of lymph and other clear body fluids under the skin near the surgical site.
Reporting of side effects
If you get any side effects, talk to your doctor or pharmacist. This includes any possible side effects not listed in this leaflet.
You can also report side effects directly via the national reporting system listed below.
United Kingdom
The Yellow Card Scheme at: www.mhra.gov.uk/yellowcard Ireland
HPRA Pharmacovigilance, Earlsfort Terrace, IRL - Dublin 2; Tel: +353 1 6764971; Fax: +353 1 6762517. Website: www.hpra.ie; e-mail: medsafety@hpra.ie By reporting side effects you can help provide more information on the safety of this medicine.
5. How to store ARTISS
Perforation
• Keep this medicine out of the sight and reach of children.
• Store and transport frozen (at <-20°C). The cold storage chain must not be interrupted until use.
• Keep the syringe in the original package in order to protect from light.
• Do not use ARTISS after the expiry date, which is stated on the label.
Storing after thawing:
Unopened pouches, thawed at room temperature, may be stored for up to 14 days at controlled room temperature (not exceeding +25°C).
After thawing, the solutions must not be refrozen or refrigerated!
ARTISS should not be disposed of via wastewater or household waste. Ask your pharmacist how to dispose of medicines no longer required. These measures will help to protect the environment.
6. Contents of the pack and other information What ARTISS contains
ARTISS contains two components:
Component 1 = Sealer Protein Solution:
The active substances contained in 1ml of the Sealer Protein Solution are:
Human Fibrinogen, 91 mg/ml; synthetic Aprotinin 3000 KlU/ml.
The excipients are Human Albumin, L-Histidine, Niacinamide, Polysorbate 80, Sodium Citrate Dihydrate and Water for Injections.
Component 2 = Thrombin Solution:
The active substances contained in 1 ml of the Thrombin Solution are:
Human Thrombin, 4 IU/ml; Calcium Chloride, 40 pmol/ml.
The excipients are Human Albumin, Sodium Chloride and Water for Injections.
|
After mixing |
1 ml |
2 ml |
4 ml |
10 ml |
|
Component 1: Sealer protein solution Human Fibrinogen (as clottable protein) Aprotinin (synthetic) |
45.5 mg 1,500 KIU |
91 mg 3,000 KIU |
182 mg 6,000 KIU |
455 mg 15,000 KIU |
|
Component 2: Thrombin Solution Human Thrombin Calcium Chloride |
2 IU 20 pmol |
4 IU 40 pmol |
8 IU 80 pmol |
20 IU 200 pmol |
ARTISS contains Human Factor XIII co-purified with Human Fibrinogen in a range of 0.6 - 5 IU/ml.
What ARTISS looks like and the contents of the pack
Frozen solutions for sealant (1 ml, 2 ml, or 5 ml of sealer protein solution and 1, 2 or 5 ml of Thrombin Solution in a single-use double-chamber syringe in a bag, and one device set with one double syringe plunger, 2 joining pieces and 4 application cannulae. Pack size of 1.).
The solution is colorless or pale yellow.
Not all pack sizes may be marketed.
Marketing Authorisation Holder and Manufacturer Marketing Authorisation Holder
Baxter Healthcare Ltd
Caxton Way
Thetford
Norfolk
IP24 3SE
United Kingdom
Tel 01635 206345
Fax 01635 206071
e-mail surecall@baxter.com
Manufacturer
Baxter AG IndustriestraBe 67 A-1221 Vienna Austria
This medicinal product is authorized in the Member States of the EEA under the following names:
ARTISS for the following countries: AT, BE, CZ, DE, EL, ES, FI, FR, IE, IT, LU, NL, NO, PL, PT, UK Artiss in DK, IS, SE APTMC (ARTISS) in BG
This leaflet was last revised in: July 2015
The following information is intended for medical or healthcare professionals only:
Fertility, pregnancy and lactation
The safety of fibrin sealants/haemostatics for use in human pregnancy or breastfeeding has not been established in controlled clinical trials. Animal studies have also not been performed.
Therefore, the product should be administered to pregnant and lactating women only if clearly needed.
The effects of ARTISS on fertility have not been established.
Posology and method of administration
ARTISS is intended for hospital use only. The use of ARTISS is restricted to experienced surgeons who have been trained in the use of ARTISS.
Posology
The amount of ARTISS to be applied and the frequency of application should always be oriented towards the underlying clinical needs of the patient.
The dose to be applied is governed by variables including, but not limited to, the type of surgical intervention, the size of the area and the mode of intended application, and the number of applications.
Application of the product must be individualized by the treating physician. In clinical trials, the individual dosages have typically ranged from 0.2 - 12 ml. For some procedures (e.g. the sealing of large burned surfaces), larger volumes may be required.
The initial amount of the product to be applied at a chosen anatomic site or target surface area should be sufficient to entirely cover the intended application area. The application can be repeated, if necessary, to any small areas that may not have been previously treated.
It is recommended that the initial application covers the entire intended application area.
As a guideline for the gluing of surfaces, 1 pack of ARTISS 2 ml (i.e., 1 ml Sealer Protein Solution pus 1 ml Thrombin Solution) will be sufficient for an area of at least 10 cm2.
The skin graft should be attached to the wound bed immediately after ARTISS has been applied. The surgeon has up to 60 seconds to manipulate and position the graft prior to polymerization. After the flap or graft has been positioned, hold in the desired position by gentle compression for at least 3 minutes to ensure ARTISS sets properly and the graft or flap adheres firmly to the underlying tissue.
The required amount of ARTISS depends on the size of the surface to be covered. The approximate surface areas covered by each pack size of ARTISS by spray application are:
|
Approximate area requiring tissue adherence |
Required pack size of ARTISS |
|
100 cm2 |
2 ml |
|
200 cm2 |
4 ml |
|
500 cm2 |
10 ml |
To avoid the formation of excess granulation tissue and to ensure gradual absorption of the solidified fibrin sealant, only a thin layer of the mixed Sealer Protein - Thrombin Solution, should be applied.
ARTISS has not been administered to patients > 65 years old in clinical trials. Paediatric Population
Currently available data are described in section 5.1 of the SmPC but no recommendation on a posology can be made.
Method of administration
For epilesional (topical) use. Do not inject.
For subcutaneous use only. ARTISS is not recommended for laparoscopic surgery. In order to ensure optimal safe use of ARTISS it should be sprayed using a pressure regulator device that delivers a maximum pressure of up to 2.0 bar (28.5 psi).
Prior to applying ARTISS the surface area of the wound needs to be dried by standard techniques (e.g. intermittent application of compresses, swabs, use of suction devices). Do not use pressurized air or gas for drying the site.
ARTISS must be sprayed only onto application sites that are visible.
ARTISS should only be reconstituted and administered according to the instructions and with the devices recommended for this product.
For spray application, see section Administration below.
Before administration of ARTISS care is to be taken that parts of the body outside the designated application area are sufficiently protected/covered to prevent tissue adhesion at undesired sites.
Special precautions for disposal and other handling General
To prevent ARTISS from adhering to gloves and instruments, wet these with sodium chloride solution before contact.
As a guideline for the gluing of surfaces, 1 pack of ARTISS 2 ml (i.e., 1 ml Sealer Protein Solution pus 1 ml Thrombin Solution) will be sufficient for an area of at least 10 cm2.
The required dose of ARTISS depends on the size of the surface to be covered. Handling and Preparation
The inner bag and its contents are sterile unless the integrity of the outside package is compromised.
It is recommended to thaw and warm the two sealant components using a sterile water bath at a temperature of 33 - 37°C. The water bath must not exceed a temperature of 37°C. (In order to control the specified temperature range, the water temperature should be monitored using a thermometer and the water should be changed as necessary. When using a sterile water bath for thawing and warming, the pre-filled double chamber syringe assembly should be removed from the aluminum-coated plastic bags.)
Perforation
The protective syringe cap should not be removed until thawing is complete and the joining piece is ready to be attached. Do not use ARTISS unless it is completely thawed and warmed (liquid consistency).
Thaw pre-filled syringes using one of the following options:
I. Room Temperature Thawing (not exceeding +25°C):
The product can be thawed at room temperature. Times given in Table 1 are minimum times for thawing at room temperature. The maximum time the product can be kept (in both aluminum-coated plastic bags) at room temperature is 14 days. When thawing at room temperature, the product must be additionally warmed to 33°C - 37°C in an incubator just before use. Respective warming times in the incubator are also given in Table 1.
Table 1: Thawing times at Room Temperature (= RT) followed by additional warming, prior to use, in an Incubator at 33°C to a maximum of 37°C
|
Pack Size |
Thawing Times at Room Temperature (Product in aluminum-coated plastic bags) |
Warming Times at 33 - 37°C in Incubator after Thawing at RT (Product in aluminum-coated plastic bags) |
|
2 ml |
60 minutes + 15 minutes | |
|
4 ml |
110 minutes + 25 minutes | |
|
10 ml |
160 minutes + 35 minutes | |
Once ARTISS has been warmed up to 33 - 37°C the product may be stored for up to 4 hours.
II. Quick Thawing:
Table 2: Thawing and Warming Times with Sterile Water Bath at 33°C to a maximum of 37°C
Transfer plunger and the inner pouch to the sterile field, remove prefilled syringe from inner pouch and place directly into sterile water bath. Ensure the contents of the prefilled syringe are completely immersed in water.
|
Pack Size |
Thawing and Warming Times (Product removed from aluminum-coated plastic bags) |
|
2 ml |
5 minutes |
|
4 ml |
5 minutes |
|
10 ml |
12 minutes |
A third alternative is to thaw the product off the sterile field using a non-sterile water bath. Maintain the prefilled syringe in both pouches and place into a water bath off the sterile field for an appropriate time (see Table 3). Ensure the pouches remain submerged throughout thawing. Remove from the water bath after thawing, dry external pouch and transfer inner pouch with prefilled syringe and plunger to the sterile field.
Table 3: Thawing and Warming times off the Sterile Field with Non-Sterile Water Bath at 33°C to a maximum of 37°C
|
Pack Size |
Thawing and Warming Times (Product in aluminum-coated plastic bags) |
|
2 ml |
30 minutes |
|
4 ml |
40 minutes |
|
10 ml |
80 minutes |
Alternatively, the sealant components may be thawed and warmed in an incubator between 33°C and 37°C. The thawing and warming times in the incubator are indicated in Table 4 below. The times refer to product in the aluminum-coated plastic bags.
Table 4: Thawing and Warming Times in Incubator at 33°C to a maximum of 37°C
|
Pack Size |
Thawing and Warming Times in Incubator (Product in aluminum-coated plastic bags) |
|
2 ml |
40 minutes |
|
4 ml |
85 minutes |
|
10 ml |
105 minutes |
Note: Do not thaw by holding product in your hands. Do not microwave.
After thawing do not refrigerate or refreeze.
After Quick Thawing (i.e. thawing at a temperature of 33 - 37°C) ARTISS may be stored at 33 - 37°C for a maximum of 4 hours.
To facilitate optimal blending of the two solutions, the two sealant components must be warmed to 33 - 37°C immediately before use. (The temperature of 37°C must, however, not be exceeded!)
The Sealer Protein and the Thrombin Solutions should be clear or slightly opalescent. Do not use solutions that are cloudy or have deposits. Thawed products should be inspected visually for particulate matter and discoloration prior to administration or any variation in physical appearance. In the event of either being observed, discard the solution.
The thawed Sealer Protein Solution should be a slightly viscous liquid. If the solution has the consistency of a solidified gel, it must be assumed to have become denatured (e.g., due to an interruption of the cold storage chain or by overheating during warming). In this case, ARTISS must not be used.
Unopened pouches, thawed at room temperature, may be stored for up to 14 days at controlled room temperature (not exceeding +25°C). If not used within 14 days after thawing, ARTISS has to be discarded.
The protective syringe cap should not be removed until thawing is complete and the joining piece is ready to be attached. Do not use ARTISS unless it is completely thawed and warmed (liquid consistency).
For further preparation instructions please refer to the responsible nurse or medical doctor.
Administration
For application, the double-chamber syringe with the Sealer Protein Solution and the Thrombin Solution has to be connected to a joining piece and an application cannula as provided in the accompanying set of devices. The common plunger of the double-chamber syringe ensures that equal volumes are fed through the joining piece before being mixed in the application cannula and ejected.
Operating Instructions
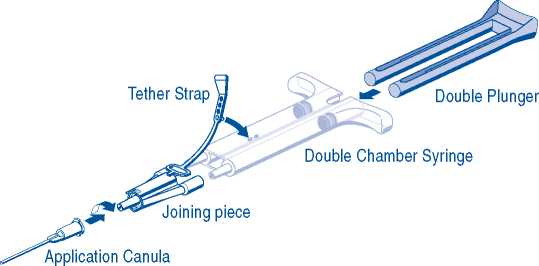
- Connect the nozzles of the double-chamber syringe to the joining piece ensuring that they are firmly fixed. Secure the joining piece by fastening the tether strap to the double-chamber syringe. If the tether strap tears, use the spare joining piece. If none is available, further use is still possible but tightness of the connection needs to be ensured to prevent any risk of leaking.
- Fit an application cannula onto the joining piece.
- Do not expel the air remaining inside the joining piece or application cannula until you start actual application as the aperture of the cannula may clog otherwise.
- Immediately before application expel and discard the first several drops from the application cannula to ensure adequately mixed product.
- Apply the mixed Sealer Protein - Thrombin Solution onto the recipient surface or surfaces of the parts to be sealed.
If application of the fibrin sealant components is interrupted, clogging may occur in the cannula. Replace the application cannula with a new one only immediately before application is resumed. If the apertures of the joining piece are clogged, use the spare joining piece provided in the package.
Spray application
When applying ARTISS using a spray device be sure to use a pressure and a distance from tissue within the ranges recommended by the manufacturer as follows:
|
Recommended pressure, distance and devices for spray application of ARTISS | |||||
|
Spray set to be used |
Applicator tips to be used |
Pressure regulator to be used |
Recommended distance from target tissue |
Recommended spray pressure | |
|
Open wound surgery of subcutaneous tissue |
Tisseel/ Artiss Spray Set |
n.a. |
EasySpray |
10 - 15 cm |
1.5 - 2.0 bar (21.5 -28.5 psi) |
|
Tisseel/ Artiss Spray Set 10 pack |
n.a. |
EasySpray | |||
When spraying the ARTISS, changes in blood pressure, pulse, oxygen saturation and end tidal CO? should be monitored because of the possibility of occurrence of air or gas embolism.
Application is also possible with other accessories supplied by BAXTER that are particularly suited for, e.g. minimally invasive surgery, application to large or difficult-to-access areas. When using these application devices, strictly follow the Instructions for Use of the devices.
After the two components have been applied, approximate the wound areas. The skin graft should be attached to the wound bed immediately after ARTISS has been applied. The surgeon has up to 60 seconds to manipulate and position the graft prior to polymerization. After the flap or graft has been positioned, hold in the desired position by gentle compression for at least 3 minutes to ensure ARTISS sets properly and the graft or flap adheres firmly to the underlying tissue.
Disposal
Any unused product or waste material should be disposed of in accordance with local requirements.