Favynd 18 Microgram Inhalation Powder Hard Capsule
uipqiaSui 4|||||K
jaSuuqaog vjly
Lunjdojjop
0|nsde3 pjeq 'jap/wod uope|equ; uiej6oj3;ui si PuAapj
jssn sip joj uopeuuojui pegeej s6e>ped
Package leaflet: Information for the user
Favynd®
18 microgram inhalation powder, hard capsule
tiotropium

Boehringer
Ingelheim
Package leaflet: Information for the user_
Favynd® ®S£r
18 microgram inhalation powder, hard capsule
tiotropium
Read all of this leaflet carefully before you start using this medicine because it contains important information for you.
- Keep this leaflet. You may need to read it again.
- If you have any further questions, ask your doctor or pharmacist.
- This medicine has been prescribed for you only. Do not pass it on to others. It may harm them, even if their signs of illness are the same as yours.
- If you get any side effects, talk to your doctor or pharmacist. This includes any possible side effects not listed in this leaflet. See section 4.
What is in this leaflet:
1. What Favynd 18 microgram is and what it is used for
2. What you need to know before you use Favynd 18 microgram
3. Flow to use Favynd 18 microgram
4. Possible side effects
5. Flow to store Favynd 18 microgram
6. Contents of the pack and other information
1. What Favynd 18 microgram is and what it is used for
Favynd 18 microgram helps people who have chronic obstructive pulmonary disease (COPD) to breathe more easily. COPD is a chronic lung disease that causes shortness of breath and coughing. The term COPD is associated with the conditions chronic bronchitis and emphysema. As COPD is a chronic disease you should use Favynd 18 microgram every day and not only when you have breathing problems or other symptoms of COPD.
Favynd 18 microgram is a long-acting bronchodilatorthat helps to open your airways and makes it easier to get air in and out of the lungs. Regular use of Favynd 18 microgram can also help you when you have on-going shortness of breath related to your disease and will help you to minimise the effects of the disease on your everyday life. It also helps you to be active longer. Daily use of Favynd 18 microgram will also help to prevent sudden, short-term worsening of your COPD symptoms which may last for several days. The effect of this medicine lasts for 24 hours, so you only need to use it once a day.
2. What you need to know before you use Favynd 18 microgram
Do not use Favynd 18 microgram
- if you are allergic to tiotropium or any other ingredients of this medicine (listed in section 6).
- if you are allergic to atropine or substances related to it, e.g. ipratropium or oxitropium.
Warnings and precautions
Talk to your doctor or pharmacist before using Favynd
18 microgram
- Talk to your doctor if you suffer from narrow angle glaucoma, prostate problems or have difficulty passing urine.
- If you have problems with your kidneys, please consult your doctor.
- Favynd 18 microgram is indicated for maintenance treatment of your chronic obstructive pulmonary disease, it should not be used to treat a sudden attack of breathlessness or wheezing.
- Immediate allergic reactions such as rash, swelling, itching, wheezing or breathlessness may occur after administration of Favynd 18 microgram. If this occurs, please consult your doctor immediately.
- Inhaled medicines such as Favynd 18 microgram may cause tightness of the chest, coughing, wheezing or breathlessness immediately after inhalation. If this occurs, please consult your doctor immediately.
- Take care not to let the inhalation powder enter your eye as this may result in precipitation or worsening of narrow-angle glaucoma, which is a disease of the eyes. Eye pain or discomfort, blurred vision, seeing halos around lights or coloured images in association with red eyes may be signs of an acute attack of narrow-angle glaucoma. Eye symptoms may be accompanied by headache, nausea or vomiting. You should stop using tiotropium bromide and immediately consult your doctor, preferably an eye specialist, when signs and symptoms of narrow-angle glaucoma appear.
- Dry mouth, which has been observed with anti-cholinergic treatment, may in the long term be associated with dental caries. Therefore, please remember to pay attention to oral hygiene.
- In case you have suffered from a myocardial infarction during the last 6 months or from any unstable or life threatening irregular heart beat or severe heart failure within the past year, please, inform your doctor. This is important to decide if Favynd is the right medicine for you to take.
- Do not use Favynd 18 microgram more frequently than once daily.
Children and adolescents
Favynd 18 microgram is not recommended for children and adolescents under 18 years.
Other medicines and Favynd 18 microgram
Tell your doctor or pharmacist if you are taking, have recently taken or might take any other medicines, including medicines available without prescription.
Please tell your doctor or pharmacist if you are using/have used similar medicines for your lung disease, such as ipratropium or oxitropium.
No specific side effects have been reported when Favynd 18 microgram has been used together with other products used to treat COPD such as reliever inhalers, e.g. salbutamol, methylxanthines, e.g. theophylline and/or oral and inhaled steroids e.g. prednisolone.
Pregnancy and breast-feeding
If you are pregnant or breast-feeding, think you may be pregnant or are planning to have a baby, ask your doctor or pharmacist for advice before taking this medicine. You should not use this medicine unless specifically recommended by your doctor.
Driving and using machines
The occurrence of dizziness, blurred vision, or headache may influence the ability to drive and use machinery.
Favynd 18 microgram contains lactose monohydrate
When taken according to dosage recommendations, one capsule once a day, each dose supplies up to 5.5 mg lactose monohydrate. If you have been told by your doctor that you have an intolerance to some sugars, contact your doctor before taking this medicinal product.
3. How to use Favynd 18 microgram
Always use this medicine exactly as your doctor or pharmacist has told you. Check with your doctor or pharmacist if you are not sure.
The recommended dose is to inhale the contents of 1 capsule (18 micrograms of tiotropium) once a day. Do not use more than the recommended dose.
Favynd 18 microgram is not recommended for children and adolescents under 18 years.
You should try to use the capsule at the same time every day. This is important because Favynd 18 microgram is effective over 24 hours.
The capsules are only for inhalation and not for oral intake.
Do not swallow the capsules.
The HandiHaler device, which you should put the Favynd capsule into, makes holes in the capsule and allows you to breathe in the powder.
Make sure that you have an HandiHaler and that you can use it properly. The instructions for use of the HandiHaler are provided on the other side of this leaflet.
Make sure that you do not blow into the HandiHaler.
If you have any problems using the HandiHaler, ask your doctor, nurse or pharmacist to show you how it works.
You should clean your HandiHaler once a month. Cleaning instructions for the HandiHaler are provided on the other side ofthis leaflet.
When taking Favynd 18 microgram, take care not to let any of the powder enter your eyes. If any powder does get into your eyes you may get blurred vision, eye pain and/or red eyes, you should wash your eyes in warm water immediately. Then talk to your doctor immediately for further advice.
If you feel that your breathing is worsening, you should tell your doctor as soon as possible
Austria, Liechtenstein
Belgium
Bulgaria
Cyprus
Czech Republic
Denmark
Finland
France
Germany
Greece
Flungary
Iceland
Ireland, Malta, UK
Italy
Latvia
Lithuania
Luxembourg
Netherlands
Norway
Poland
Portugal
Romania
Slovakia
Slovenia
Sweden
If you use more Favynd 18 microgram than you should
If you inhale from more than 1 capsule of Favynd 18 microgram in a day, you should talkto your doctor immediately. You may be at a higher risk of experiencing a side effect such as dry mouth, constipation, difficulties passing urine, increased heart beat, or blurred vision.
If you forget to use Favynd 18 microgram
If you forget to take a dose, take one as soon as you remember but do not take two doses at the same time or on the same day. Then take your next dose as usual.
If you stop taking Favynd 18 microgram
Before you stop taking Favynd 18 microgram, you should talk to your doctor or your pharmacist. If you stop taking Favynd 18 microgram the signs and symptoms of COPD may worsen.
If you have any further questions on the use of this medicine, askyour doctor or pharmacist.
4. Possible side effects
Like all medicines, this medicine can cause side effects, although not everybody gets them.
The side effects described below have been experienced by people taking this medicine and they are listed according to frequency as either common, uncommon, rare or not known.
Common (may affect up to 1 in 10 people):
- dry mouth: this is usually mild
Uncommon (may affect up to 1 in 100 people):
- dizziness
- headache
- taste disorders
- blurred vision
- irregular heart beat (atrial fibrillation)
- inflammation of the throat (pharyngitis)
- hoarseness (dysphonia)
- cough
- heart burn (gastrooesophageal reflux disease)
- constipation
- fungal infections of the oral cavity and throat (oropharyngeal candidiasis)
- rash
- difficulties passing urine (urinary retention)
- painful urination (dysuria)
Rare (may affect up to 1 in 1.000 people):
- difficulty in sleeping (insomnia)
- seeing halos around lights or coloured images in association with red eyes (glaucoma)
- increase of the measured eye pressure
- irregular heart beat (supraventricular tachycardia)
- faster heart beat (tachycardia)
- feeling your heart beat (palpitations)
- tightness of the chest, associated with coughing, wheezing or breathlessness immediately after inhalation (bronchospasm)
- nosebleed (epistaxis)
- inflammation of the larynx (laryngitis)
- inflammation of the sinuses (sinusitis)
- blockage of intestines or absence of bowel movements (intestinal obstruction including ileus paralytic)
- inflammation of the gums (gingivitis)
- inflammation of the tongue (glossitis)
- difficulties swallowing (dysphagia)
- inflammation ofthe mouth (stomatitis)
- feeling sick (nausea)
- hypersensitivity, including immediate reactions
- serious allergic reaction which causes swelling ofthe face or throat (angioedema)
- nettle rash (urticaria)
- itching (pruritus)
- infections ofthe urinary tract
Not known (frequency cannot be estimated from the available data):
- depletion of body water (dehydration)
- dental caries
- severe allergic reaction (anaphylactic reaction)
- infections or ulcerations of the skin
- dryness of the skin
- swelling of joints
Serious side effects include allergic reactions which cause swelling of the face or throat (angioedema) or other hypersensitivity reactions (such as sudden reduction of your blood pressure or dizziness) may occur individually or as part of severe allergic reaction (anaphylactic reaction) after administration of Favynd 18 microgram. In addition, in common with all inhaled medicines, some patients may experience an unexpected tightness of the chest, coughing, wheezing or breathlessness immediately after inhalation (bronchospasm). If any of these occur, please consult your doctor immediately.
Reporting of side effects
If you get any side effects, talkto your doctor or pharmacist. This includes any possible side effects not listed in this leaflet. You can also report side effects directly via the Yellow Card Scheme at: www.mhra.gov.uk/yellowcard. By reporting side effects you can help provide more information on the safety of this medicine.
5. How to store Favynd 18 microgram
Keep this medicine out ofthe sight and reach of children.
Do not use this medicine after the expiry date which is stated on the carton and the blister foil. The expiry date refers to the last day of that month.
Do not store above 25°C.
Do not freeze.
Once you have taken your first capsule from the blister you must continue to take the capsules for the next 9 days, one capsule a day, from the same blister.
Do not throw away any medicines via wastewater or household waste. Askyour pharmacist how to throw away medicines you no longer use. These measures will help protect the environment.
6. Contents of the pack and other information What Favynd 18 microgram contains
- The active substance is tiotropium. Each capsule contains 18 micrograms ofthe active ingredient tiotropium (as bromide monohydrate). The delivered dose (the dose that leaves the mouthpiece ofthe HandiHaler device) is
10 micrograms tiotropium.
- The other ingredient is lactose monohydrate.
What Favynd 18 microgram looks like and contents of the pack
Favynd 18 microgram inhalation powder, hard capsule is a light green hard capsule containing the inhalation powder, with the product code Tl 01 and company logo printed on the capsule.
The product is available in the following packs:
A pack containing 30 capsules
A pack containing 60 capsules
A pack containing 90 capsules
A pack containing 10 capsules and 1 HandiHaler
A pack containing 30 capsules and 1 HandiHaler
Hospital pack: Bundle pack containing 5 cardboard boxes of
30 capsules plus HandiHaler device
Hospital pack: Bundle pack containing 5 cardboard boxes of 60 capsules
Marketing Authorisation Holder and Manufacturer
The marketing authorisation holder for Favynd 18 microgram is:
Boehringer Ingelheim International GmbFI Binger StraBe 173 D-55216 Ingelheim am Rhein Germany
The manufacturer for Favynd 18 microgram and the HandiHaler device is:
Boehringer Ingelheim Pharma GmbFI &Co. KG
Binger StraGe 173
D-55216 Ingelheim am Rhein
Germany
This medicinal product is authorised in the member States of the EEA under the following names:
Favynd 18 Mikrogramm
Flartkapseln mit Pulver zur Inhalation
Favynd - 18 meg poudre pour
inhalation en gelule
CDaBMHfl 18 MMKporpaMa
npax 3a MHxanapnfl, TBt>pflM Kancynn
Favynd Kovic; yia eianvoij,
oxAripo KarjjdKio 18mcg
Favynd 18 pg prasek k inhalaci
vtvrdych tobolkach
Favynd, inhalationspulver
i kapsler 18 mikrogram
Favynd 18 mikrog inhalaatiojaue,
kapseli, kova
Favynd 18 microgrammes, poudre pour inhalation en gelule Favynd 18 Mikrogramm Flartkapseln mit Pulver zur Inhalation Favynd Kovic; yia eionvorj, oxAripo KaipaKio INFIPD.CAP 18mcg/CAP Favynd 18 mikrogramm inhalacios por kemeny kapszulaban Favynd 18 mikrogromm, innondunarduft, hart hylki Favynd 18 microgram inhalation powder, hard capsule Favynd 18 microgrammi, polvere per inalazione, capsula rigida Favynd 18 mikrogrami, inhalacijas pulveris cietas kapsula Favynd 18 mikrogramp jkvepiamieji milteliai (kietosios kapsules)
Favynd GELULES 18 MCG Favynd 18 microgram, inhalatiepoeder in harde capsules Favynd inhalasjonspulver, hard kapsel 18 mikrog Favynd, 18 pg/dawk^ inhalacyjnq, proszek do inhalacji w kapsutkach twardych
Favynd, PO PARA INALAgAO,
CAPSULA DURA 0,018 mg Favynd 18 micrograme capsule cu pulbere de inhalat Favynd 18 mikrogramov, inhalacny prasok vtvrdej kapsule Favynd 18 mikrogramov prasek za inhaliranje, trde kapsule Favynd, inhalationspulver, hard kapsel 18 mikrog
This leaflet was last revised in 09/2015.
Not all pack sizes may be marketed.
HandiHaler® instructions for use
Dear Patient,
The HandiHaler enables you to inhale the medicine contained in the Favynd capsule - that your physician has prescribed for your breathing problems.
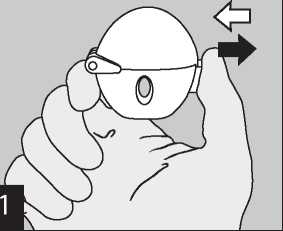
To release the dust cap press the piercing button completely in and let go.
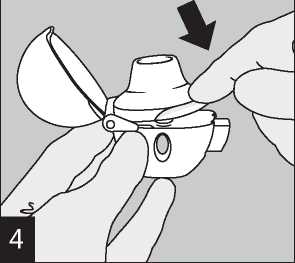
Close the mouthpiece firmly until you hear a click, leaving the dust cap open.
Remember to carefully follow your doctor's instructions for using Favynd. The HandiHaler is especially designed for Favynd. You must not use it to take any other medicine. You can use your HandiHaler for up to one year to take your medicine.
Open the dust cap completely by pulling it upwards. Then open the mouthpiece by pulling it upwards.
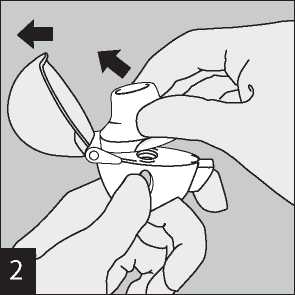
Hold the HandiHaler device with the mouthpiece upwards and press the piercing button completely in only once, and release. This makes holes in the capsule and allows the medicine to be released when you breathe in.
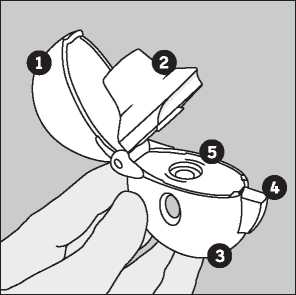
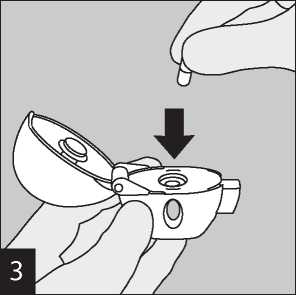
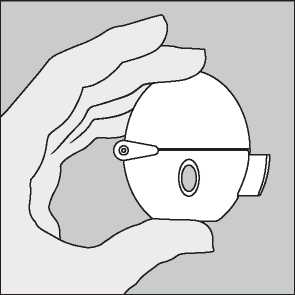
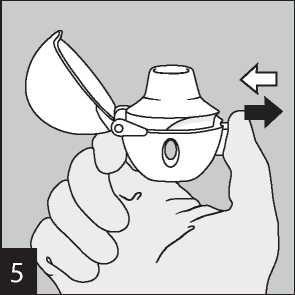
Remove a Favynd capsule from the blister (only immediately before use, see blister handling) and place it in the centre chamber (©), as illustrated. It does not matter which way the capsule is placed in the chamber.
Breathe out completely.
Important: Please avoid breathing into the mouthpiece at any time.

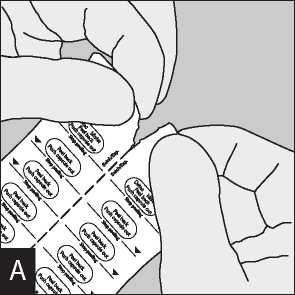
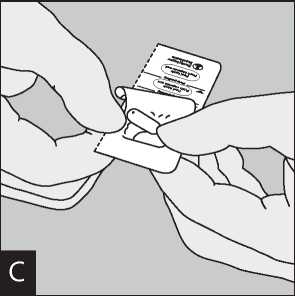
Separate the blister strips by tearing along the perforation.
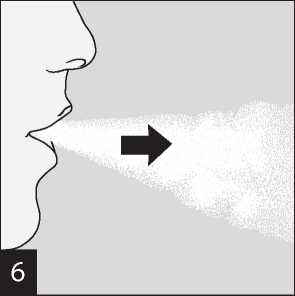
Raise the HandiHaler to your mouth and close your lips tightly around the mouthpiece. Keep your head in an upright position and breathe in slowly and deeply but at a rate sufficient to hear or feel the capsule vibrate. Breathe in until your lungs are full; then hold your breath as long as comfortable and at the same time take the HandiHaler out of your mouth.
Resume normal breathing. Repeat steps 6 and 7 once, in order to empty the capsule completely.
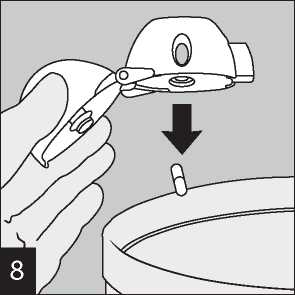
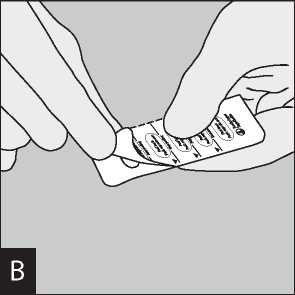
Open the mouthpiece again. Tip out the used capsule Peel back foil (only immediately before use) using and dispose. Close the mouthpiece and dust cap for the tab until one capsule is fully visible,
storage of your HandiHaler device.
In case a second capsule is exposed to air inadvertently this capsule has to be discarded.
Cleaning your HandiHaler
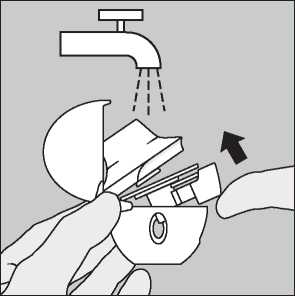
Clean the HandiHaler once a month. Open the dust cap and mouthpiece. Then open the base by lifting the piercing button. Rinse the complete inhaler with warm water to remove any powder. Dry the HandiHaler thoroughly by tipping excess of water out on a paper towel and air-dry afterwards, leaving the dust cap, mouthpiece and base open. It takes 24 hours to air dry, so clean it right after you have used it and it will be ready for your next dose. If needed, the outside of the mouthpiece may be cleaned with a moist but not wet tissue.
Remove capsule.
Favynd capsules contain only a small amount of powder so that the capsule is only partially filled.
Manufacturer of the HandiHaler device:
Boehringer Ingelheim Pharma GmbH & Co. KG D-55216 Ingelheim am Rhein Germany