Kliovance
8-2901-01-053-9_v1-4:Layout 1 2015-08-25 10:13 AM Page 1
Code start — Code: 100% Direction Length: Max. 29 mm (100%)
llllllllllll

I
novo nordisk
Package leaflet:
Information for the user
Kliovance®
1 mg/0.5 mg film-coated tablets
Estradiol/norethisterone acetate
Read all of this leaflet carefully before you start taking this medicine because it contains important information for you.
- Keep this leaflet. You may need to read it again.
- If you have any further questions, ask your doctor or pharmacist.
- This medicine has been prescribed for you only. Do not pass it on to others. It may harm them, even if their signs of illness are the same as yours.
- If you get any side effects, talk to your doctor or pharmacist. This
8-2901-01-053-9 includes any possible side effects not listed in this leaflet. See section 4.
What is in this leaflet
1. What Kliovance® is and what it is used for
2. What you need to know before you take Kliovance®
3. How to take Kliovance®
4. Possible side effects
5. How to store Kliovance®
6. Contents of the pack and other information
1. What Kliovance® is and what it is used for
Kliovance® is a continuous combined Hormone Replacement Therapy (HRT). It contains two types of female hormones, an oestrogen and a progestagen. Kliovance® is used in postmenopausal women with at least 1 year since their last natural period.
Kliovance® is used for:
Relief of symptoms occurring after menopause
During the menopause, the amount of oestrogen produced by a woman's body drops. This can cause symptoms such as hot face, neck and chest ('hot flushes'). Kliovance® alleviates these symptoms after menopause. You will only be prescribed Kliovance® if your symptoms seriously hinder your daily life.
Prevention of osteoporosis
After the menopause some women may develop fragile bones (osteoporosis). You should discuss all available options with your doctor.
If you are at an increased risk of fractures due to osteoporosis and other medicines are not suitable for you, you can use Kliovance® to prevent osteoporosis after menopause.
Kliovance® is prescribed for women who have not had their womb removed, and whose periods stopped more than a year ago. There is only limited experience of treating women older than 65 years with Kliovance®.
2. What you need to know before you take Kliovance® Medical history and regular check-ups
The use of HRT carries risks which need to be considered when deciding whether to start taking it, or whether to carry on taking it.
The experience in treating women with a premature menopause (due to ovarian failure or surgery) is limited. If you have a premature menopause the risks of using HRT may be different. Please talk to your doctor.
Before you start (or restart) HRT, your doctor will ask about your own and your family's medical history. Your doctor may decide to perform a physical examination. This may include an examination of your breasts and/or an internal examination, if necessary.
Once you have started on Kliovance® you should see your doctor for regular check-ups (at least once a year). At these check-ups, discuss with your doctor the benefits and risks of continuing with Kliovance®. Go for regular breast screening, as recommended by your doctor.
Do not take Kliovance®
If any of the following applies to you. If you are not sure about any of the points below, talk to your doctor before taking Kliovance®.
Do not take Kliovance®:
• If you have, have had or suspect having breast cancer.
• If you have, or have had cancer which is sensitive to oestrogens, such as cancer of the womb lining (endometrium), or if you are suspected of having it.
• If you have any unexplained vaginal bleeding.
• If you have excessive thickening of the womb lining (endometrial hyperplasia) that is not being treated.
• If you have or have ever had a blood clot in a vein (thrombosis), such as in the legs (deep venous thrombosis) or the lungs (pulmonary embolism).
• If you have a blood clotting disorder (such as protein C, protein S or antithrombin deficiency).
• If you have or previously have had a disease caused by blood clots in the arteries, such as a heart attack, stroke or angina.
• If you have or have ever had a liver disease and your liver function tests have not returned to normal.
• If you have a rare blood problem called 'porphyria'
which is passed down in families (inherited).
• If you are allergic (hypersensitive) to estradiol, norethisterone acetate or any of the other ingredients of Kliovance® (listed in section 6 'Contents of the pack and other information').
If any of the above conditions appear for the first time while taking Kliovance®, stop taking it at once and consult your doctor immediately.
Warnings and precautions
Tell your doctor if you have ever had any of the following problems, before you start the treatment, as these may return or become worse during treatment with Kliovance®.
If so, you should see your doctor more often for check-ups:
• fibroids inside your womb
• growth of the womb lining outside your womb (endometriosis) or a history of excessive growth of the womb lining (endometrial hyperplasia)
• increased risk of developing blood clots (see 'Blood clots in a vein (thrombosis)')
• increased risk of getting
a oestrogen-sensitive cancer (such as having a mother, sister or grandmother who has had breast cancer)
• high blood pressure
• a liver disorder, such as a benign liver tumour
• diabetes
• gallstones
• migraine or severe headaches
• a disease of the immune system that affects many organs of the body (systemic lupus erythematosus, SLE)
• epilepsy
• asthma
• a disease affecting the eardrum and hearing (otosclerosis)
• a very high level of fat in your blood (triglycerides)
• fluid retention due to cardiac or kidney problems
• lactose intolerance.
Stop taking Kliovance® and see a doctor immediately
If you notice any of the following when taking HRT:
• any of the conditions mentioned in the 'Do not take Kliovance®' section
• yellowing of your skin or the whites of your eyes (jaundice). These may be signs of a liver disease
• a large rise in your blood pressure (symptoms may be headache, tiredness, dizziness)
• migraine-like headaches which happen for the first time
• if you become pregnant
• if you notice signs of a blood clot, such as:
- painful swelling and redness of the legs
- sudden chest pain
- difficulty in breathing.
For more information, see 'Blood clots in a vein (thrombosis)'.
Note: Kl iovance® is not a contraceptive. If it is less than 12 months since your last menstrual period or you are under 50 years old, you may still need to use additional contraception to prevent pregnancy. Speak to your doctor for advice.
HRT and cancer
Excessive thickening of the lining of the womb (endometrial hyperplasia) and cancer of the lining of the womb (endometrial cancer)
Taking oestrogen-only HRT will increase the risk of excessive thickening of the lining of the womb (endometrial hyperplasia) and cancer of the womb lining (endometrial cancer).
The progestagen in Kliovance® protects you from this extra risk.
Irregular bleeding
You may have irregular bleeding or drops of blood (spotting) during the first 3-6 months of taking Kliovance®. However, if the irregular bleeding:
• carries on for more than the first 6 months
• starts after you have been taking Kliovance® for more than
6 months
• carries on after you have stopped taking Kliovance®
see your doctor as soon as possible. Breast cancer
Evidence suggests that taking combined oestrogen-progestagen and possibly also oestrogen-only HRT increases the risk of breast cancer. The extra risk depends on how long you take HRT. The additional risk becomes clear within a few years. However, it returns to normal within a few years (at most 5) after stopping treatment.
Compare
Women aged 50 to 79 who are not taking HRT, on average, 9 to 17 in 1,000 will be diagnosed with breast cancer over a 5-year period. For women aged 50 to 79 who are taking oestrogen-progestagen HRT over 5 years, there will be 13 to 23 cases in 1,000 users (i.e. an extra 4 to 6 cases).
Regularly check your breasts. See your doctor if you notice any changes such as:
• dimpling of the skin
• changes in the nipple
• any lumps you can see or feel. Additionally, you are advised to join mammography screening programs when offered to you. For mammogram screening, it is important that you inform the nurse/healthcare professional who is actually taking the x-ray that you use HRT, as this medication may increase the density of your breasts which may affect the outcome of the mammogram. Where the density of the breast is increased, mammography may not detect all lumps.
Ovarian cancer
Ovarian cancer is rare. A slightly increased risk of ovarian cancer has been reported in women taking HRT for at least 5 to 10 years. Women aged 50 to 69 who are not taking HRT, on average, about 2 women in 1,000 will be diagnosed with ovarian cancer over a 5-year period.
For women who have been taking HRT for 5 years, there will be between 2 and 3 cases per 1,000 users (i.e. up to 1 extra case).
Effect of HRT on heart and
circulation
Blood clots in a vein
(thrombosis)
The risk of blood clots in the veins
is about 1.3 to 3 times higher in HRT users than in non-users, especially during the first year of taking it. Blood clots can be serious, and if one travels to the lungs, it can cause chest pain, breathlessness, fainting or even death.
You are more likely to get a blood clot in your veins as you get older and if any of the following applies to you. Inform your doctor if any of these situations applies to you:
• you are unable to walk for a long time because of major surgery, injury or illness (see also section 3, 'If you need to have surgery')
• you are seriously overweight (BMI >30 kg/m2)
• you have any blood clotting problem that needs long-term treatment with a medicine used to prevent blood clots
• if any of your close relatives has ever had a blood clot in the leg, lung or another organ
• you have systemic lupus erythematosus (SLE)
• you have cancer.
For signs of a blood clot, see 'Stop taking Kliovance® and see a doctor immediately'.
Compare
Looking at women in their 50s who are not taking HRT, on average, over a 5-year period,
4 to 7 in 1,000 would be expected to get a blood clot in a vein.
For women in their 50s who have been taking oestrogen-progestagen HRT for over 5 years, there will be 9 to 12 cases in 1,000 users (i.e. an extra 5 cases).
Heart disease (heart attack)
There is no evidence that HRT will prevent a heart attack. Women over the age of 60 years who use oestrogen-progestagen HRT are slightly more likely to develop heart
disease than those not taking any HRT.
Stroke
The risk of getting stroke is about 1.5 times higher in HRT users than in non-users. The number of extra cases of stroke due to use of HRT will increase with age.
Compare
Looking at women in their 50s who are not taking HRT, on average, 8 in 1,000 would be expected to have a stroke over a 5-year period.
For women in their 50s who are taking HRT, there will be 11 cases in 1,000 users over 5 years (i.e. an extra 3 cases).
Other conditions
HRT will not prevent memory loss. There is some evidence of a higher risk of memory loss in women who start using HRT after the age of 65. Speak to your doctor for advice.
Using other medicines
Some medicines may interfere with the effect of Kliovance®. This might lead to irregular bleeding. This applies to the following medicines:
• Medicines for epilepsy (such as phenobarbital, phenytoin and carbamazepine)
• Medicines for tuberculosis (such as rifampicin and rifabutin)
• Medicines for HIV infections (such as nevirapine, efavirenz, ritonavir and nelfinavir)
• Herbal remedies containing St John's Wort (Hypericum perforatum)
• Medicines for hepatitis C infections (such as telaprevir).
Other medicines may increase the effects of Kliovance®:
• Medicines containing ketoconazole (a fungicide).
Kliovance® may have an impact on a concomitant treatment with cyclosporine.
Please tell your doctor or pharmacist if you are taking or have recently taken any other medicines, including medicines obtained without a prescription, herbal medicines or other natural products.
Laboratory tests
If you need a blood test, tell your doctor or the laboratory staff that you are taking Kliovance®, because this medicine can affect the results of some tests.
Taking Kliovance® with food and drink
The tablets can be taken with or without food and drink.
Pregnancy and breast-feeding Pregnancy: Kliovance® is for use in postmenopausal women only. If you become pregnant, stop taking Kliovance® and contact your doctor.
Breast-feeding: You should not take Kliovance® if you are breastfeeding.
Driving and using machines
Kliovance® has no known effect on the ability to drive or use machines.
Important information about some of the ingredients in Kliovance®:
Kliovance® contains lactose monohydrate. If you have an intolerance to some sugars, contact your doctor before taking Kliovance®.
3. How to take Kliovance®
Always take this medicine exactly as your doctor has told you. Check with your doctor or pharmacist if you are unsure.
Take one tablet once a day, at about the same time each day.
Once you have finished all the 28 tablets in the pack, start a new pack continuing the treatment without interruption.
For further information on the use of the calendar pack, see 'User Instructions' at the end of the package leaflet.
You may start treatment with Kliovance® on any convenient day. However, if you are switching from an HRT product when you have monthly bleeding, start your treatment straight after the bleeding has ended.
Your doctor should aim to prescribe the lowest dose to treat your symptom for as short as necessary. Speak to your doctor if you think this dose is too strong or not strong enough.
If you take more Kliovance® than you should
If you have taken more Kliovance® than you should, talk to a doctor or pharmacist. An overdose of Kliovance® could make you feel sick or vomit.
If you forget to take Kliovance®
If you forget to take your tablet at the usual time, take it within the next 12 hours. If more than 12 hours have gone by, skip the missed dose and start again as
RA Labelling & IT Insert size: 999x100-006 Current: 2.0
Improved DI28MKII Colour: PMS 280C
Code start
Code: 100% Direction -*
Length: Max. 29 mm (100%)
8-2901-01-053-9_v1-4:Layout 1 2015-08-25
10:13 AM
Page
2

normal the next day. Do not take a double dose to make up for a forgotten tablet. Forgetting a dose may increase the likelihood of breakthrough bleeding and spotting if you still have your womb.
If you stop taking Kliovance®
If you would like to stop taking Kliovance®, talk to your doctor first. Your doctor will explain the effects of stopping treatment and discuss other possibilities with you.
If you have any further questions on the use of this medicine, ask your doctor or pharmacist.
If you need to have surgery
If you are going to have surgery, tell the surgeon that you are taking Kliovance®. You may need to stop taking Kliovance® about 4 to 6 weeks before the operation to reduce the risk of a blood clot (see section 2, 'Blood clots in a vein (thrombosis)'). Ask your doctor when you can start taking Kliovance® again.
4. Possible side effects
Like all medicines, this medicine can have side effects, although not everybody gets them.
The following diseases are reported more often in women using HRT compared to women not using HRT:
• breast cancer
• abnormal growth or cancer of the lining of the womb (endometrial hyperplasia or cancer)
• ovarian cancer
• blood clots in the veins of the legs or lungs (venous thromboembolism)
• heart disease
• stroke
• probable memory loss if HRT is started over the age of 65.
For more information about these side effects, see section 2, 'What you need to know before you take Kliovance®'.
Hypersensitivity/allergy
(uncommon side effect - affects 1 to 10 users in 1,000)
Though it is an uncommon event, hypersensitivity/allergy may occur. Signs of hypersensitivity/allergy may include one or more of the following symptoms: hives, itching, swelling, difficulty in breathing, low blood pressure (paleness and coldness of skin, rapid heart beat), feeling dizzy, sweating, which could be signs of anaphylactic reaction/shock. If one of the mentioned symptoms appears, stop taking Kliovance® and seek immediate medical help.
Very common side effects
(affects more than 1 user in 10)
• Breast pain or breast tenderness
• Vaginal bleeding.
Common side effects (affects 1 to 10 users in 100)
• Headache
• Weight gain caused by fluid retention
• Vaginal inflammation
• Migraine, new or worse than before
• Vaginal infection with a fungus
• Depression, new or worse than before
• Nausea
• Enlargement or swelling of the breasts (breast oedema)
• Back pain
• Uterine fibroid (benign tumour), aggravation, occurrence or reoccurrence
• Swelling of arms and legs (peripheral oedema)
• Weight increase.
Uncommon side effects (affects
1 to 10 users in 1,000)
• Bloating, abdominal pain, swelling, discomfort or flatulence
• Acne
• Hair loss (alopecia)
• Abnormal (male pattern) hair growth
• Itching or hives (urticaria)
• Inflammation of a vein (superficial thrombophlebitis)
• Leg cramps
• Drug ineffective
• Allergic reaction
• Nervousness.
Rare side effects (affects 1 to
10 users in 10,000)
• Blood clots in the blood vessels of the legs or the lungs (deep vein thrombosis, lung embolism).
Very rare side effects (affects less
than 1 user in 10,000)
• Cancer of the lining of the womb (endometrial cancer)
• Excessive thickening of the lining of the womb (endometrial hyperplasia)
• Increase in blood pressure or worsening of high blood pressure
• Gall bladder disease, gall stones occurrence/reoccurrence or aggravated
• Excessive secretion of sebum, skin eruption
• Acute or recurring attack of oedema (angioneurotic oedema)
• Insomnia, dizziness, anxiety
• Change in sexual desire
• Visual disturbances
• Weight decreased
• Vomiting
• Heartburn
• Vaginal and genital itching
• Heart attack and stroke.
Other side effects of combined
HRT
• gall bladder disease
• various skin disorders:
- discoloration of the skin especially of the face or neck known as 'pregnancy patches' (chloasma)
- painful reddish skin nodules (erythema nodosum)
- rash with target-shaped reddening or sores (erythema multiforme)
- red or purple discolorations of the skin and/or mucous membranes (vascular purpura)
Reporting of side effects
If you get any side effects, talk to
your doctor or pharmacist. This
includes any possible side effects not listed in this leaflet. You can also report side effects directly via the Yellow Card Scheme Website: www.mhra.gov.uk/yellowcard. By reporting side effects you can help provide more information on the safety of this medicine.
5. Howto store Kliovance®
Keep this medicine out of the sight and reach of children.
Do not use this medicine after the expiry date, which is stated on the label and outer carton after 'EXP'. The expiry date refers to the last day of that month.
Do not store above 25°C Do not refrigerate.
Keep the container in the outer carton in order to protect it from light.
Do not throw away any medicines via wastewater or household waste. Ask your pharmacist how to throw away medicines you no
longer use. These measures will help protect the environment.
6. Contents of the pack and other information What Kliovance® contains
- The active substances are estradiol 1 mg (as estradiol hemihydrate) and norethisterone acetate 0.5 mg.
- The other ingredients are: lactose monohydrate, maize starch, copovidone, talc and magnesium stearate.
- The film-coating contains: hypromellose, triacetin and talc.
What Kliovance® looks like and contents of the pack
The film-coated tablets are white, round with a diameter of 6 mm.
The tablets are engraved NOVO 288 on one side and the Novo Nordisk logo (an Apis bull) on the other side.
Pack sizes:
• 1 x28 film-coated tablets in a calendar pack
• 3x28 film-coated tablets in calendar packs
Not all pack sizes may be marketed.
Marketing Authorisation Holder and Manufacturer
Marketing Authorisation Holder:
Novo Nordisk Limited
3 City Place
Beehive Ring Road
Gatwick
West Sussex
RH6 0PA
Manufacturer:
Novo Nordisk A/S Novo Alle
DK-2880 Bagsrard Denmark
This medicinal product is authorised in the Member States of the EEA under the following names:
Member States of the EEA: Activelle® - except for United Kingdom: Kliovance®.
This leaflet was last revised in 09/2015
User Instructions
How to use the calendar pack
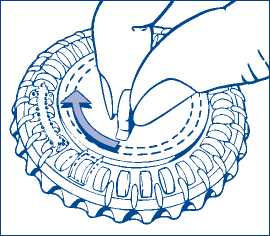
1. Set the day reminder
Turn the inner disc to set the day of the week opposite the little plastic tab.
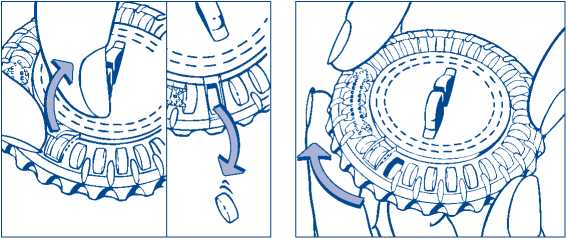
3. Move the dial every day
On the next day, simply move the transparent dial clockwise 1 space as indicated by the arrow. Tip out the next tablet. Remember to take only 1 tablet once a day.
You can only turn the transparent dial after the tablet in the opening has been removed.
Kliovance® is a trademark owned by Novo Nordisk Health Care AG, Switzerland
© 2015
Novo Nordisk A/S
novo nordisk