Latanoprost 0.005% Eye Drops
Out of date information, search anotherXALATAN® 0.005% EYE DROPS / LATANOPROST 0.005% EYE DROPS
_(latanoprost)_
PATIENT INFORMATION LEAFLET
This product is available using the names Xalatan 0.005% Eye Drops / Latanoprost 0.005% Eye Drops but will be referred to as Xalatan throughout the leaflet.
Read all of this leaflet carefully before you start using this medicine. Even if you have already used Xalatan or a similar medicine before, we advise you to read this text carefully. The information may have been changed.
• Keep this leaflet. You may need to read it again.
• If you have any further questions, ask your doctor or the doctor treating your child or your pharmacist.
• This medicine has been prescribed for you or for your child. Do not pass it on to others. It may harm them, even if their symptoms are the same as yours.
• If any of the side effects gets serious, or if you notice any side effects not listed in this leaflet, please tell your doctor or the doctor treating your child or your pharmacist.
In this leaflet:
1. What Xalatan is and what it is used for
2. Before you use Xalatan
3. How to use Xalatan
4. Possible side effects
5. How to store Xalatan
6. Further information
1. WHAT XALATAN IS AND WHAT IT IS USED FOR
Xalatan belongs to a group of medicines known as prostaglandin analogues. It works by increasing the natural outflow of fluid from inside the eye into the bloodstream.
Xalatan is used to treat conditions known as open angle glaucoma and ocular hypertension in adults. Both of these conditions are linked with an increase in the pressure within your eye, eventually affecting your eye sight.
Xalatan is also used to treat increased eye pressure and glaucoma in all ages of children and babies.
2. BEFORE YOU USE XALATAN
Xalatan can be used in adult men and women (including the elderly) and in children from birth to 18 years of age. Xalatan has not been investigated in prematurely born infants (less than 36 weeks gestation).
Do not use Xalatan if you are
• Allergic (hypersensitive) to latanoprost or any of the other ingredients of Xalatan (see section 6 for the list of ingredients in your medicine)
• Pregnant or trying to become pregnant
• Breast-feeding
Take special care with Xalatan
Talk to your doctor or the doctor treating your child or your pharmacist before you take Xalatan or before you give this to your child if you think any of the following apply to you or your child:
• If you or your child are about to have or have had eye surgery (including cataract surgery)
• If you or your child suffer from eye problems (such as eye pain, irritation or inflammation, blurred
vision)
• If you or your child suffers from dry eyes
• If you or your child have severe asthma or the asthma is not well controlled
• If you or your child wear contact lenses. You can still use Xalatan, but follow the instruction for
contact lens wearers in Section 3
• If you have suffered or are currently suffering from a viral infection of the eye caused by the herpes simplex virus (HSV)
Taking other medicines
Xalatan may interact with other medicines. Please tell your doctor, the doctor treating your child or pharmacist if you or your child are taking or have taken any other medicines including those medicines (or eye drops) obtained without a prescription.
Pregnancy
Do not use Xalatan when you are pregnant. Tell your doctor immediately if you are pregnant, think you are pregnant, or are planning to become pregnant.
Breast-feeding
Do not use Xalatan when you are breast-feeding.
Driving and using machines
When you use Xalatan you might have blurred vision, for a short time. If this happens to you, do not drive or use any tools or machines until your vision becomes clear again.
Important information about some of the ingredients of Xalatan
Xalatan contains a preservative called benzalkonium chloride. This preservative may cause eye irritation or disruption to the surface of the eye. Benzalkonium chloride can be absorbed by contact lenses and is known to discolour soft contact lenses. Therefore, avoid contact with soft contact lenses.
If you or your child wear contact lenses, they should be removed before using Xalatan. After using Xalatan you should wait 15 minutes before putting the contact lenses back in. See the instructions for contact lens wearers in Section 3.
3. HOW TO USE XALATAN
Always use Xalatan exactly as your doctor or the doctor treating your child has told you. You should check with your doctor or the doctor treating your child or pharmacist if you are not sure.
The usual dosage for adults (including the elderly) and children is one drop once a day in the affected eye(s). The best time to do this is in the evening.
Do not use Xalatan more than once a day, because the effectiveness of the treatment can be reduced if you administer it more often.
Use Xalatan as instructed by your doctor or by the doctor treating your child until they tell you to stop.
Contact lens wearers
If you or your child wear contact lenses, they should be removed before using Xalatan. After using Xalatan you should wait 15 minutes before putting the contact lenses back into the eyes.
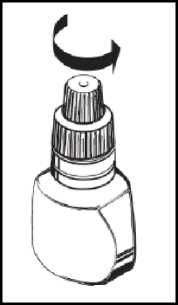
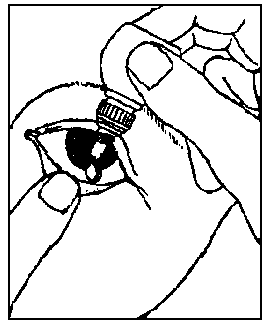
Instructions for use
1. Wash your hands and sit or stand comfortably.
2. Unscrew the protective cap. The protective cap should be retained.
3. Use your finger to gently pull down the lower eyelid of your affected eye.
4. Place the tip of the bottle close to, but not touching your eye.
5. Squeeze the bottle gently so that only one drop goes into your eye, then release the lower eyelid.
6. Press a finger against the corner of the affected eye by the nose. Hold for 1 minute whilst keeping the eye closed.
7. Repeat in your other eye if your doctor has told you to do this.
8. Put the protective cap back on the bottle.
If you use Xalatan with other eye drops
Wait at least 5 minutes between using Xalatan and taking other eye drops.
If you use more Xalatan than you should
If you put too many drops into the eye, it may lead to some minor irritation in the eye and the eyes
may water and turn red. This should pass, but if you are worried contact your doctor or the doctor
treating your child for advice.
Contact your doctor as soon as possible if you or your child swallows Xalatan accidentally.
If you forget to use Xalatan
Carry on with the usual dosage at the usual time. Do not take a double dose to make up for the dose
you have forgotten. If you are unsure about anything talk to your doctor or pharmacist.
If you stop using Xalatan
You should speak to your doctor or the doctor treating your child if you want to stop taking Xalatan.
4. POSSIBLE SIDE EFFECTS
Like all medicines, Xalatan can cause side effects, although not everybody gets them.
Very common effects (likely to affect more than 1 in 10 people):
• A gradual change in your eye colour by increasing the amount of brown pigment in the coloured part of the eye known as the iris. If you have mixed-colour eyes (blue-brown, grey-brown, yellow-brown or green-brown) you are more likely to see this change than if you have eyes of one colour (blue, grey, green or brown eyes). Any changes in your eye colour may take years to develop although it is normally seen within 8 months of treatment. The colour change may be permanent and may be more noticeable if you use Xalatan in only one eye. There appears to be no problems associated with the change in eye colour. The eye colour change does not continue after Xalatan treatment is stopped.
• Redness of the eye.
• Eye irritation (a feeling of burning, grittiness, itching, stinging or the sensation of a foreign body in the eye).
• A gradual change to eyelashes of the treated eye and the fine hairs around the treated eye, seen mostly in people of Japanese origin. These changes involve an increase of the colour (darkening), length, thickness and number of your eye lashes.
Common effects (likely to affect less than 1 in 10 people):
• Irritation or disruption to the surface of the eye, eyelid inflammation (blepharitis) and eye pain.
Uncommon effects (likely to affect less than 1 in every 100 people):
• Eyelid swelling, dryness of the eye, inflammation or irritation of the surface of the eye (keratitis), blurred vision and conjunctivitis.
• Skin rash.
Rare effects (likely to affect less than 1 in every 1000 people):
• Inflammation of the iris, the coloured part of the eye (iritis/uveitis); swelling of the retina (macular oedema), symptoms of swelling or scratching/damage to the surface of the eye, swelling around the eye (periorbital oedema), misdirected eyelashes or an extra row of eyelashes, light sensitivity (photophobia).
• Skin reactions on the eyelids, darkening of the skin of the eyelids.
• Asthma, worsening of asthma and shortness of breath (dyspnoea).
Very rare effects (likely to affect less than 1 in 10,000 people):
• Worsening of angina in patients who also have heart disease, chest pain, sunken eye appearance (eye sulcus deepening).
Patients have also reported the following side-effects: fluid filled area within the coloured part of the eye (iris cyst), headache, dizziness, palpitations, muscle pain, joint pain and developing a viral infection of the eye caused by the herpes simplex virus (HSV).
Side effects seen more often in children compared to adults are: runny itchy nose and fever.
If any of the side effects become serious, or if you notice any side effects not listed in this leaflet, please contact your doctor or pharmacist.
5. HOW TO STORE XALATAN
• Keep all medicines in a safe place, out of the sight and reach of children.
• Do not use after the expiry date which is stated on the carton and bottle. The expiry date refers to the last day of that month.
• Before Xalatan is first opened keep it in a fridge (between 2°C and 8°C / 36°F and 46°F), out of direct light.
• Once the bottle has been opened, it is not necessary to put it in the fridge; Xalatan can be kept at normal room temperature (below 25°C / 77°F).
• Each bottle should be thrown away four weeks after first opening.
• If your medicine appears to be discoloured, or shows any other signs of deterioration, take it back to your pharmacist who will advise you.
• If your doctor tells you to stop using your medicine, please take it back to the pharmacist for safe disposal. Only keep it if your doctor tells you to.
• Medicines should not be disposed of via wastewater or household waste. Ask your pharmacist how to dispose of medicines no longer required. These measures will help to protect the environment.
6. FURTHER INFORMATION
What Xalatan contains
The active substance is 50 microgrames/ml (0.005% w/v) latanoprost.
The other ingredients are: benzalkonium chloride Ph Eur, sodium chloride Ph Eur, sodium dihydrogen phosphate monohydrate, disodium phosphate anhydrous and water for injections Ph Eur.
What Xalatan looks like and contents of the pack
Xalatan is a clear, colourless liquid.
Each bottle contains 2.5ml of solution.
Manufacturer
This product is manufactured by: Pfizer Manufacturing Belgium NV, Rijksweg 12, 2870 Puurs, Belgium.
Procured from within the EU and repackaged by: Doncaster Pharmaceuticals Group Ltd., Kirk Sandall, Doncaster, DN3 1QR.
Product Licence holder: Doncaster Pharmaceuticals Group Ltd., Kirk Sandall, Doncaster, DN3 1QR.
POM
PL No: 04423/0349
Leaflet review and issue date (Ref): 26.03.13
Xalatan® is a registered trademark of Pfizer Ltd.
Page 2 of 2