Phenylephrine 50 Micrograms/Ml Solution For Injection In Pre-Filled Syringe
The pre-filled syringe is for single patient only. Discard syringe after use. Do not reuse.
The content ofun-opened and un-damaged blister is sterile, and must not be openeduntil use.
The product should be inspected visually for particles and discoloration prior to administration. Only clear colourless solution free from particles or precipitates shouldbe used.
The product should not be used if the tamper evident seal on the syringe is broken.
The external surface of syringe is sterile until blister is opened.
When handledusing an aseptic method, Phenylephrine 50 micrograms/ml, solution for injection in pre-filled syringe can be placed > a sterile field.
1) Withdraw the pre-filled syringe from the sterile blister.
|
1 |
( \ |
|
_!_C |
i |
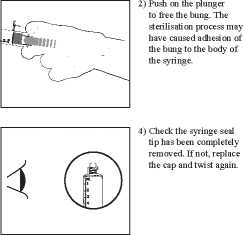
3) Itoist off the end cap to break the seal. Do not touch the exposed luer connection in order to avoid contamination.
5) Expel the air by gently pushing the plunger.
6) Connect the syringe to the IV access. Push the plunger slowly to inject the required volume.
7) Any unused medicinal product or waste material should be disposed ofin accordance with local requirements.
Package leaflet: Information for the patient
Phenylephrine 50 micrograms/ml, Solution for Injection in Pre-filled Syringe
(Referredto as “Phenylephrine Injection” inthis leaflet) Phenylephrine
Read all of this leaflet carefully before you are given this medicine because it contains important information for you.
- Keep this leaflet. You may need to read it again.
- Ifyou have any further questions, askyour doctor, pharmacist
- Ifyou get any side effects, talk toyour doctor, pharmacist or nurse. This includes any possible side effects not listed in this leaflet. See section 4.
What is in this leaflet
1. What Phenylephrine Injection is andwhat it is usedfor
2. What you need to know before you are given Phenylephrine Injection
3. How Phenylephrine Injection is given
4. Possible side effects
5. How to store Phenylephrine Injection
6. Contents of the pack and other information
1. What Phenylephrine Injection is and what it is used for
This medicine belongs to a group of adrenergic and dopaminergic agents.
It is used to treat low blood pressure during anaesthesia.
2. What you need to know before you are given Phenylephrine Injection
You will not receive Phenylephrine Injection:
- ifyou are allergic to phenylephrine hydrochloride or any of the other ingredients ofthis medicine (listed in section 6);
- ifyou suffer from severe high blood pressure, or peripheral vascular disease (poor blood circulation);
- ifyou are taking a Monoamine Oxydase inhibitor (MAO) (or within 2 weeks oftheir withdrawal), that is usedto treat depression (such as iproniazide, nialamide);
- ifyou suffer from severe overactive thyroidgland (hyperthyroidism).
Warnings and precautions
Ihlk to your doctor, pharmacist or nurse before you are given Phenylephrine Injection:
- ifyou are elderly;
- ifyou are diabetic;
- ifyou suffer from arterial hypertension;
- ifyou have an overactive thyroidgland (uncontrolled hyperthyroidism);
- ifyou have bloodvessel disease, such as arteriosclerosis (hardening andthickening ofthe walls ofthe bloodvessels);
- ifyou have a poor blood circulation in the brain;
- ifyou suffer from heart disease including chronic heart conditions, peripheral vascular insufficiency, heart rhythm disorders, tachycardia (high heart rate), bradycardia (low heart rate), partial heart block, angina pectoris;
- ifyou suffer from a closed angle glaucoma (a rare eye disease).
In patients with serious heart failure, phenylephrine may worsen the heart failure as a consequence ofbloodvessel constriction.
The bloodpressure in your arteries will be monitored during treatment. Ifyou have heart disease, additional monitoring of vital functions will be performed.
Children
This medicine is not recommended for use in children due to insufficient data on efficacy, safety and dosage recommendations. Other medicines and Phenylephrine Injection Tell your doctor ifyou are using, have recently used or might use any other medicines, such as:
- certain antidepressants (iproniazid, nialamide, moclobenide, toloxatone, imipramine, milnacipran or venlafaxine);
- medicine used to treat infections (linezolid);
- certain medicines used to treat migraine (dihydroergotamine, ergotamine, methylergometrine, methylsergide);
- certain medicines used to treat Parkinson disease (bromocriptine, lisuride, pergolide);
- medicine usedto inhibit the production of an hormone responsible oflactation (cabergoline);
- anaesthetics that are inhaled (desflurane, enflurane, halothane, isoflurane, methoxyflurane, sevoflurane);
- medicine used as an appetite suppressant (sibutramine);
- medicine usedto treat high bloodpressure (guanethidine);
- medicines used to treat heart failure and certain irregular heartbeats (cardiac glycosides);
- medicine used to treat abnormal heart rhythm (quinidine);
- medicine used during labour (oxytocin).
Pregnancy and breast-feeding
The safety of this medicine during pregnancy and breast-feeding has not been established, but the use ofPhenylephrine Injection is possible during pregnancy if necessary.
Use ofthis medicine during breast-feeding is not recommended. However, in the event of a single administration during childbirth, breast-feeding is possible.
Ifyou are pregnant or breast-feeding, think you may be pregnant or are planning to have a baby, askyour doctor, pharmacist or nurse for advice before being given this medicine.
Driving and using machines
Thlk toyour doctor ifyou want to drive and/or use machines after being given this medicine.
Phenylephrine Injection contains sodium This medicinal product contains 3.68 mg (0.160 mmol) of sodium per ml (a total of36.8 mg or 1.60 mmol sodium in a 10 ml syringe). This amount must be taken into consideration by patients on a salt-restricted diet.
3. How Phenylephrine Injection is given
The administration will be performed by a healthcare professional with appropriate training andrelevant experience, who will decide the correct dosage foryou andwhen and how the injection shouldbe administered.
The recommended doses are:
Use inAdults
Your doctor will determine the dose to be administered, and may repeat or adjust it until the desired effect is attained.
Use in patients with impaired renal function (kidneys not functioning well)
Lower doses ofphenylephrine may be neededin patients with impairedrenal function.
Use In patients with impaired liver function (liver not functioning well) Higher doses ofphenylephrinemaybeneeded inpatients with cirrhosis of the liver.
Use in older people
Tteatment in older people should be carried out with care.
Use in children
This medicine is not recommendedfor use in children due to insufficient data on efficacy, safety and dosage recommendations.
If you have been given more Phenylephrine Injection than you should:
You may have the following symptoms: palpitation, cardiac rhythm disorders (tachycardia, cardiac arrhythmias).
Ifyou have any further questions on the use ofthis medicine, ask your doctor, pharmacist, or nurse.
4. Possible side effects
Like all medicines, this medicine can cause side effects, although not everybody gets them.
Some side effects may be serious (frequencies are not known). Tell your doctor straight away if you get any of the following:
- chest pain or pain due to the angina,
- irregular heartbeat,
- feeling the heart pumping in the chest,
- bleeding in the brain (speech disorder, dizziness, paralysis of one side ofthe body),
- psychosis (loosing contact with reality).
Other side effects may include (frequencies are not known):
- reaction ofhypersensitivity (allergy),
- excessive dilation ofthe pupils,
- increased pressure in the eye (aggravation of glaucoma),
- excitability (excessive sensitivity of an organ or body part),
- agitation (restlessness),
- confusion,
- headache,
- nervousness,
- insomnia (difficulty falling or staying asleep),
- shaking (tremor),
- burning ofthe skin,
- prickling ofthe skin,
- itching or tingling skin sensation (paresthesia),
- slow or high heart rate,
- high blood pressure,
- difficulty in breathing,
- fluids in the lung,
- nausea,
- vomiting,
- sweating,
- pallor or skin blanching (pale colour ofthe skin),
- goose flesh,
- tissue damage at the site ofthe injection,
- muscle weakness,
- difficulty in passing urine or urine retention.
Reporting of side effects
Ifyou get any side effects, talk to your doctor, pharmacist or nurse. This includes any possible side effects not listed in this leaflet. You can also report side effects directly via Yellow Card Scheme - Website: www.mhra.gov.uk/yellowcard. By reporting side effectsyou can help provide more information on the safety of this
5. Howto store Phenylephrine Injection
Keep this medicine out ofthe sight andreach of children.
You shouldnot be given this medicine after the expiry date which is stated on the carton and syringe label. The expiry date refers to the last day of that month. Your doctor or nurse will check this.
Store the blister in the outer carton in order to protect from light. Keep the syringe in its unopened blister until use.
Do not use this medicine ifyou notice visible signs of deterioration. Any syringe, even partially used, should be discarded appropriately
Do not throw away any medicines via wastewater or household waste. Askyour pharmacist how to throw away medicines you no longer use. These measures will help protect the environment.
6. Contents of the pack and other information
What Phenylephrine Injection contains
The active ingredient is phenylephrine hydrochloride.
- Each ml of solution for injection contains phenylephrine hydrochloride, equivalent to 50 micrograms phenylephrine.
- Each 10 ml pre-filled syringe contains phenylephrine hydrochloride, equivalent to 500 micrograms phenylephrine.
- The other ingredients are sodium chloride, sodium citrate dihydrate, citric acid monohydrate, sodium hydroxide, and water for injections.
What Phenylephrine Injection looks like and contents of the
Phenylephrine Injection is a clear colourless solution, in a 10 ml polypropylene pre-filled syringe, individually packagedin a transparent blister pack.
The pre-filled syringes are available in boxes of 1 and 10 syringes. Not all pack sizes may be marketed.
Marketing Authorisation Holder and Manufacturer Marketing Authorisation Holder:
Jenson Pharmaceutical Services Ltd
Carradine House
237 Regents Park Road
London
N3 3LF
United Kingdom Manufacturer:
LABORATOIRE AGUETTANT 1, rue Alexander Fleming 69007 LYON France
For any information about this medicine, please contact the local representative ofthe Marketing Authorisation Holder:
Jenson Pharmaceutical Services Ltd
Carradine House
237 Regents Park Road
London
N3 3LF
United Kingdom
This leaflet was last revised in 09/2015.
Detailedinformation on this medicinal product is available on the website of the Medicines and Healthcare Products Regulatory Agency (MHRA).
TECHNICAL LEAFLET
PHENYLEPHRINE 50 micrograms/ml, Solution for injection in pre-filled syringe
The following information is intended for medical and healthcare professionals only:
This is an extract from the Summary ofProduct Characteristics to assist in the administration ofPHENYLEPHRINE 50 micrograms/ml, solution for injection in pre-filled syringe. When determining appropriateness of use in a particular patient, the prescriber shouldbe familiar with the Summary ofProduct Characteristics oftheproduct.
Qualitative and quantitative composition:
Each ml of solution for injection contains phenylephrine hydrochloride equivalent to 50 micrograms (0.05 mg) phenylephrine.
Each 10 ml pre-filled syringe contains phenylephrine hydrochloride equivalent to 500 micrograms (0.5 mg) phenylephrine.
Excipients with known effect: sodium.
Each ml of solution for injection contains 3.68 mg equivalent to 0.160 mmol of sodium.
Each 10 ml pre-filled syringe contains 36.8 mg equivalent to 1.60 mmol of sodium.
Other excipients: Sodium citrate dihydrate, Citric acidmonohydrate, Sodium hydroxide, water for injections.
Description of solution:
Clear colorless solution. pH: 4.7 - 5.3
Osmolality: 270-300 mOsm/Kg Posology and method of administration
Posologv
Adults
Intravenous bolu injection:
Normal dose is 50 to 100 micrograms, which can be repeated until the desired effect is attained. One bolus dose should not exceed 100 micrograms.
Continuotus infusion:
Initial dose is 25 to 50 micrograms/min. The doses may be increased or decreasedto maintain the systolic blood pressure close to the normal value. Doses between 25 and 100 micrograms/min have been assessed to be effective.
Renal impairment
Lower doses of phenylephrine may be needed in patients with impaired renal function.
Hepatic Impairment
Higher doses ofphenylephrine may be neededin patients with cirrhosis ofthe liver.
Older people
Tteatment of the elderly should be carried out with care.
Paediatric population
The safety and efficacy ofphenylephrine in children have not been established. No data are available.
Method of administration:
Parenteral administration. Intravenousbolusinjection orintravenous infusion.
Phenylephrine, 50 micrograms/ml, solution for injection should only be administeredby healthcare professionals with appropriate training and relevant experience.
Contraindications Phenylephrine should not be used:
- hypersensitivity to the active substance or to any ofthe excipients listed in section 6.1;
- in patients with severe hypertension or peripheral vascular disease due to the risk ofischemic gangrene or vascular thrombosis;
- in combination with non-selective monoamine oxidase inhibitors (MAOs) (or within 2 weeks of their withdrawal) due to risk of paroxysmal hypertension andpossibly fatal hyperthermia (see section 4.5);
- in patients with severe hyperthyroidism.
Special warning and precautions for use
The arterial bloodpressure should be monitored during treatment. Phenylephrine shouldbe administeredwith care to patients with:
- diabetes mellitus,
- arterial hypertension,
- uncontrolledhyperthyroidism,
- coronary heart disease and chronic heart conditions,
- non-severe peripheral vascular insufficiency,
- bradycardia,
- partial heart block,
- tachycardia,
- arrhythmias,
- angina pectoris (phenylephrine can precipitate or exacerbate angina in patients with coronary artery disease and history of angina),
- aneurysma,
- closed angle glaucoma.
Phenylephrine can induce a reduction in cardiac output. Therefore, care shouldbe exercisedin administering to patients with arteriosclerosis, the elderly andto patients with impaired cerebral or coronary circulation.
In patients with reduced cardiac output or coronary vascular disease, vital organ functions shouldbe closely monitored and dose reduction should be considered when systemic blood pressure is near the lower end of the target range.
In patients with serious heart failure or cardiogenic shock, phenylephrine may cause deterioration in the heart failure as a consequence of the induced vasoconstriction (increase in afterload). Particular attention should be paid to Phenylephrine injection to avoid extravasation, since this may cause tissue necrosis.
This medicinal product contains sodium. Each 10 ml pre-filled syringe contains 36.8 mg (equivalent to 1.60 mmol of sodium). To be taken into consideration by patients on a controlled sodium diet. Overdose:
Symptoms of overdose include headache, nausea, vomiting, paranoidpsychosis, hallucinations, hypertension andreflex bradycardia. Cardiac arrhythmia such as ventricular extrasystoles and short paroxysmal episodes ofventricular tachycardia may occur. Tteatment should consist of symptomatic and supportive measures. The hypertensive effects may be treated with an alpha-adrenoceptor blocking drug, such as phentolamine.
Pharmaceutical particulars:
Incompatibilities:
In the absence of compatibility studies, this medicinal product must not be mixedwith other medicinal products.
Storage:
Keep the syringe in its unopened blister until use. Store the blister in the outer carton in order to protect from light.