Wilate 500/Wilate 1000 Powder And Solvent For Solution For Injection
2015.03.25 / 200x490mm / B.181.006.UK_01 Color: Black, Black 20%
2015.03.25 / 200x490mm / B.181.006.UK_01 Color: Black, Black 20%
Read all of this leaflet carefully before you start using this
medicine.
• Please keep this leaflet. You may need to read it again.
• If you have further questions, please ask your doctor or your pharmacist.
• This medicine has been prescribed for you personally and you should not pass it on to others. It may harm them, even if their symptoms are the same as yours.
• If any of the side effects gets serious, or if you notice any side effects not listed in this leaflet, please tell your doctor or pharmacist.
1. What Wilate is and what it is used for
2. Before you use Wilate
3. How to use Wilate
4. Possible Side Effects
B.181.006.UK
PACKAGE LEAFLET: INFORMATION FOR THE USER
Wilate 500, 500 IU FVIII / 500 IU VWF, powder and solvent for solution for injection
Wilate 1000, 1000 IU FVIII / 1000 IU VWF, powder and solvent for solution for injection
Human coagulation factor VIII / human von Willebrand factor
In this leaflet:
1. What Wilate is and what it is used for
2. Before you use Wilate
3. How to use Wilate
4. Possible side effects
5. How to store Wilate
6. Further information
Wilate belongs to the pharmacotherapeutic group of medicines called clotting factors and contains human blood coagulation factor VIII (FVIII) and von Willebrand factor (VWF). Together these two proteins are involved in blood clotting.
Von Willebrand disease
Wilate is used to treat and prevent bleeding in patients with von Willebrand disease (VWD), which in fact is a family of related diseases. All types of VWD are inborn, where bleeding can go on for longer than expected. This is either due to a lack of VWF in the blood or due to VWF that does not work the way it should.
Haemophilia A
Wilate is used to treat and prevent bleeding in patients with haemophilia A. This is a condition in which bleeding can go on for longer than expected. It is due to an inborn lack of FVIII in the blood.
Do not use Wilate
• if you are allergic (hypersensitive) to human blood coagulation factor VIII, von Willebrand factor or any of the other ingredients of Wilate.
Take special care with Wilate
• Any medicine, such as Wilate, which is prepared from human blood (containing proteins) and which is injected into a vein (administered intravenously) can cause allergic reactions. Please pay attention to early signs of allergic reactions (hypersensitivity), such as hives, skin rash, tightness of the chest, wheezing, low blood pressure, or anaphylaxis (when any or all of the above symptoms develop rapidly and are intense)
If these symptoms occur, stop the injection immediately and contact your doctor.
• When medicines are made from human blood or plasma, certain measures are put in place to prevent infections being passed on to patients. These include careful selection of blood and plasma donors to make sure those at risk of carrying infections are excluded, and the testing of each donation and pools of plasma for signs of virus/infections. Manufacturers of these products also include steps in the processing of the blood or plasma that can inactivate or remove viruses. Despite these measures, when medicines prepared from human blood or plasma are administered, the possibility of passing on infection cannot be totally excluded. This also applies to any unknown or emerging viruses or other types of infections.
The measures taken are considered effective for enveloped viruses such as human immunodeficiency virus (HIV), hepatitis B (HBV) virus and hepatitis C virus (HCV), and for the non-enveloped hepatitis A virus (HAV). The measures taken may be of limited value against non-enveloped viruses such as parvovirus B19.
Parvovirus B19 infection may be serious for pregnant women (infection of the baby) and for individuals whose immune system is depressed or who have some types of anaemia (e.g. sickle cell disease or abnormal breakdown of red blood cells).
• It is strongly recommended that every time you receive a dose of Wilate the name and the batch number of the product are recorded in order to maintain a record of the batches used.
Your doctor may recommend that you consider vaccination against hepatitis A and B if your regularly/repeatedly receive human plasma-derived FVIII/VWF products.
Von Willebrand disease (VWD)
• Please see section 4. (Von Willebrand disease (VWD)) for side effects related to the treatment of VWD.
Haemophilia A
• Please see section 4. (Haemophilia A) for side effects related to the treatment of haemophilia A.
Taking other medicines
Although no influences on Wilate from other medicinal products are known, please tell your doctor or pharmacist if you are taking or have recently taken any other medicines (including medicines obtained without a prescription).
Please do not mix Wilate with any other medicines during the injection.
Pregnancy and breast-feeding
Ask your doctor or pharmacist for advice before using Wilate.
Important information about some of the ingredients of Wilate
This medicinal product contains up to 2.55 mmol sodium (58.7 mg) per dose for 500 IU FVIII and VWF/vial and up to 5.1 mmol sodium (117.3 mg) per dose for 1000 IU FVIII and VWF/vial. To be taken into consideration if you are on a controlled sodium diet.
Wilate should be injected into a vein (administered intravenously) after reconstitution with the supplied solvent. Treatment should be started under medical control.
Dosage
Your doctor will advise you about your individual dosage and the frequency with which you should use Wilate. Always use Wilate exactly as your doctor has told you. You should check with your doctor or pharmacist if you are not sure.
If you use more Wilate than you should
No symptoms of overdose with human FVIII or VWF have been reported. However, the recommended dosage should not be exceeded.
If you forget to take Wilate
Do not take a double dosage to make up for a forgotten dosage.
If you have any further questions on the use of this product, ask your doctor or pharmacist.
Like all medicines, Wilate can cause side effects, although not everybody gets them.
• Even though uncommon, hypersensitivity or allergic reactions have been observed. These may include:
burning and stinging at the infusion site, chills, flushing, headache, hives (uticaria), low blood pressure (hypotension), tiredness (lethargy), sickness (nausea), restlessness, increase of heart rate (tachycardia), tightness of the chest, feeling of pins and needles (tingling), vomiting, wheezing, sudden swellings in various parts of the body (angiooedema).
If you suffer from any of the above-mentioned symptoms, please inform your doctor.
You should stop using Wilate and see your doctor immediately, if you experience symptoms of angiooedema, such as:
o swollen face, tongue or throat (pharynx)
o difficulties to swallow
o hives and difficulties to breath
• On rare occasions, fever has been observed.
• In very rare cases, hypersensitivity may lead to a severe allergic reaction called anaphylaxis (when any or all of the above symptoms develop rapidly and are intense), which may include shock. In case of an anaphylactic shock, treatment using the current medical recommendations for shock is essential.
Von Willebrand disease (VWD)
• When using a FVIII-containing VWF product to treat VWD, the continued treatment may cause an excessive rise in FVIII in the blood. This may increase the risk that your blood flow will be disturbed (thrombosis).
If you are a patient with known clinical or laboratory risk factors, you have to be checked for early signs of thrombosis. Prevention (prophylaxis) of thrombotic events should be decided by your doctor, according to the current recommendations.
• Patients with VWD (especially type 3 patients) may develop inhibitors (neutralising antibodies) to VWF during the treatment with VWF. In these very rare cases inhibitors can stop Wilate working properly.
In case your bleeding continues, your blood has to be tested for these inhibitors.
Inhibitors may increase the risk of suffering severe allergic reactions (anaphylactic shock). If you suffer an allergic reaction, you should be tested for the presence of inhibitors.
Once inhibitors have been found in your blood, please contact a physician with experience in the care of patients with bleeding disorders. In patients with high amounts of inhibitors, another kind of treatment might be useful and should be considered.
Haemophilia A
• When using FVIII products to treat patients with haemophilia A, the formation of inhibitors (neutralising antibodies) to FVIII is a known complication. In these rare cases inhibitors can stop Wilate working properly and bleeding may continue. Please contact a specialised haemophilia centre if Wilate does not stop your bleeding. Regular blood tests will be performed during treatment to test for these inhibitors.
Inhibitors may increase the risk of suffering severe allergic reactions (anaphylactic shock). If you suffer an allergic reaction, you should be tested for the presence of inhibitors.
Uncommon: affects 1 to 10 users in 1,000 Rare: affects 1 to 10 users in 10,000 Very rare: affects less than 1 user in 10,000
5. How to store Wilate
There are insufficient data to recommend the use of Wilate in previously untreated patients.
The experience of treatment with Wilate in children less than 6 years of age is limited.
For information on viral safety see section 2. (Take special care with Wilate).
If any of the side effects gets serious, or if you notice any side effects not listed in this leaflet, please tell you doctor or pharmacist.
Instructions for Home Treatment
• Please read all the instructions and follow them carefully!
• Do not use Wilate after expiry date given on the label.
• During the procedure described below, sterility must be maintained!
• The solution in the syringe should be clear or slightly pearly shimmery. Do not inject solutions that are cloudy or have deposits.
• Use the prepared solution immediately, to prevent microbial contamination.
• Only use the injection set provided. The use of other injection/ infusion equipment can cause additional risks and treatment failure.
6. Further information

Keep out of the reach and sight of children.
Store powder and solvent vial in a refrigerator (2°C - 8°C).
Do not freeze.
Keep the vials in the outer carton to protect from light.
Do not use Wilate after the expiry date stated on the label.
Wilate can be stored at room temperature (max. +25°C) for 2 months. In this case the shelf-life expires 2 months after the product has been taken out of the refrigerator for the first time. The new shelf-life has to be noted on the outer carton by you.
The powder should be dissolved only directly before injection. The stability of the solution has been demonstrated for 12 hours at room temperature. Nevertheless, to prevent contamination, the solution should be used immediately and on one occasion only. Any unused product or waste must be disposed of correctly.
What Wilate contains
- The active substances are human coagulation factor VIII and human von Willebrand factor
- The other ingredients are sodium chloride, glycine, sucrose, sodium citrate and calcium chloride. Solvent: water for injections with
0.1% Polysorbate 80
What Wilate looks like and contents of the pack
Freeze-dried powder: white or pale yellow powder or crumbly solid Reconstituted solution: should be clear or slightly opalescent
Wilate is supplied as a powder and solvent for solution for injection. It comes in 2 pack sizes:
• Wilate 500, 500 IU FVIII and 500 IU VWF, powder and solvent for solution for injection, contains nominally 500 IU human coagulation factor VIII and 500 IU human von Willebrand factor per vial. The product contains approximately 100 IU/ml human coagulation factor VIII and 100 IU/ml human von Willebrand factor when reconstituted with 5 ml of Water for Injections with 0.1% Polysorbate 80 (Solvent).
• Wilate 1000, 1000 IU FVIII and 1000 IU VWF, powder and solvent for solution for injection, contains nominally 1000 IU human coagulation factor VIII and 1000 IU human von Willebrand factor per vial. The product contains approximately 100 IU/ml human coagulation factor VIII and 100 IU/ml human von Willebrand factor when reconstituted with 10 ml of Water for Injections with 0.1% Polysorbate 80 (Solvent).
Content of the package
1 vial with freeze-dried powder 1 vial with solvent
1 equipment pack with the following medical devices:
1 disposable syringe 1 transfer set (Mix2VialTM)
1 infusion set
2 alcohol swabs
Marketing Authorisation Holder
Octapharma Limited The Zenith Building 26 Spring Gardens Manchester M2 1AB United Kingdom
For any further information about this medicinal product, please contact the local representative of the Marketing Authorisation Holder:
Instructions for Preparing the Solution:
1. Do not use the product directly from the refrigerator. Allow the solvent and the powder in the closed vials to reach room temperature.
2. Remove the flip off caps from both vials and clean the rubber stoppers with one of the provided alcohol swabs.
3. The Mix2vial™ is depicted in Fig. 1. Place the solvent vial on an even surface and hold it firmly. Take the Mix2Vial™ and turn it upside down. Place the blue part of the Mix2Vial™ on top of the solvent vial and press firmly down until it snaps (Fig. 2 + 3).
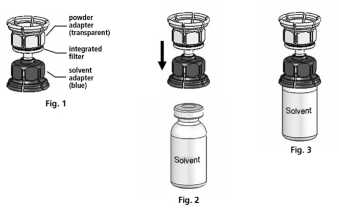
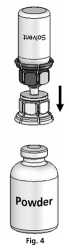
4. Place the powder vial on an even surface and hold it firmly. Take the solvent vial with the attached Mix2Vial™ and turn it upside down. Place the transparent part on top of the powder vial and press firmly down until it snaps (Fig. 4). The solvent flows automatically into the powder vial.
5. With both vials still attached, gently swirl the powder vial until the product is dissolved.
The dissolving is completed in less than 10 minutes at room temperature. Slight foaming might occur during preparation. Unscrew the Mix2Vial™ into two parts (Fig. 5). Foaming will disappear.
Dispose the empty solvent vial with the blue part of the Mix2Vial™.
Instructions for Injection:
As a precaution, your pulse rate should be taken before and during the injection. If a marked increase in your pulse rate occurs, reduce the injection speed or interrupt the administration for a short time.
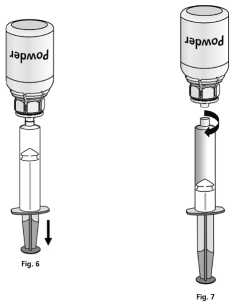
1. Attach the syringe to the transparent part of the Mix2Vial™. Turn the vial upside down and draw the solution into the syringe (Fig. 6).
The solution in the syringe should be clear or slightly pearly shimmery.
Once the solution has been transferred, firmly hold the plunger of the syringe (keeping it facing down) and remove the syringe from the Mix2Vial™ (Fig. 7). Dispose the Mix2Vial™ and the empty vial.
2. Clean the chosen injection site with one of the provided alcohol
Octapharma Limited
swabs
The Zenith Building 26 Spring Gardens
Manchester M2 1AB 3. Attach the provided injection needle to the syringe.
United Kingdom
Manufacturer
Octapharma Pharmazeutika Produktionsges.m.b.H. Oberlaaerstr. 235 A-1100 Vienna Austria
4. Insert the injection needle into the chosen vein. If you have used a tourniquet to make the vein easier to see, this tourniquet should be released before you start injecting Wilate.
No blood must flow into the syringe due to the risk of formation of fibrin clots.
5. Inject the solution into the vein at a slow speed, not faster than 2-3 ml per minute.
This leaflet was last approved in 03/2015.
This medicinal product is authorised in the Member States of the EEA under the following names:
Austria, Belgium, Bulgaria, Cyprus, Czech Republic, Estonia, Finland, Germany, Greece, Hungary, Ireland, Italia, Latvia, Lithuania, Luxembourg, Malta, Netherlands, Norway, Poland, Portugal, Romania, Slovenia, Slovak Republic, Spain, Sweden, United Kingdom: Wilate
Denmark: Wilnativ
If you use more than one vial of Wilate powder for one treatment, you may use the same injection needle and syringe again. The Mix2Vial™ is for single use only.
Any unused product or waste material should be disposed of in accordance with local requirements.
Wilate must not be mixed or injected (with the same infusion set) with other medicinal products.
Only use the injection set provided. The use of other injection/ infusion equipment can cause additional risks and treatment failure (FVIII/VWF adsorption to the internal surfaces of some infusion equipment).
B.181.006.UK
B.181.006.UK_GTP_01.indd 2 25.03.15 14:15